Physicochemical Properties
| Molecular Formula | C20H26CLN3O2 |
| Molecular Weight | 375.8923 |
| Exact Mass | 375.171 |
| Elemental Analysis | C, 63.91; H, 6.97; Cl, 9.43; N, 11.18; O, 8.51 |
| CAS # | 119942-70-0 |
| PubChem CID | 189710 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 26 |
| Complexity | 555 |
| Defined Atom Stereocenter Count | 2 |
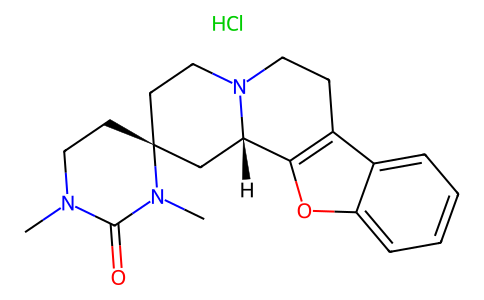
| SMILES | CN1CC[C@]2(CCN3CCC4=C([C@@H]3C2)OC5=CC=CC=C45)N(C)C1=O.Cl |
| InChi Key | ALYCEQJIRFYVGE-VASSOYJASA-N |
| InChi Code | InChI=1S/C20H25N3O2.ClH/c1-21-11-8-20(22(2)19(21)24)9-12-23-10-7-15-14-5-3-4-6-17(14)25-18(15)16(23)13-20;/h3-6,16H,7-13H2,1-2H3;1H/t16-,20+;/m0./s1 |
| Chemical Name | (2S,12bS)-1',3'-dimethylspiro[1,3,4,6,7,12b-hexahydro-[1]benzofuro[2,3-a]quinolizine-2,4'-1,3-diazinane]-2'-one;hydrochloride |
| Synonyms | MK-912; MK 912; 119942-70-0; MK-912 hydrochloride; MK-912 hydrate; MK912; MK 912; 4A6522ZKT5; L 657743-002W; MK912 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | alpha 2-adrenergic receptor |
| ln Vitro |
MK‐912 is the highest‐affinity, most α2C‐selective antagonist (0.15 nM α2C‐affinity) although its α2C‐selectivity is only 13‐fold greater than at α2A. [1] Figure 4 shows the affinity (log KD values) of the most selective ligand at each adrenoceptor subtype (i.e., BRL44408 for α2A, S32212 for α2B and MK‐912 for α2C) along with the single most selective antagonists at the other adrenoceptors and demonstrates that the α2‐adrenoceptors fall behind α1 and β with regards to availability of highly subtype‐selective ligands.[1] |
| ln Vivo | MK-912, a selective α2C-antagonist, and Y-27632, a Rho-kinase inhibitor, significantly inhibited the PSBF reduction induced by cooling to 15 °C in both mice and the inhibitory effects were comparable between these mice[2]. |
| References |
[1]. The affinity and selectivity of α-adrenoceptor antagonists, antidepressants and antipsychotics for the human α2A, α2B, and α2C-adrenoceptors and comparison with human α1 and β-adrenoceptors. Pharmacol Res Perspect. 2022 Apr;10(2):e00936. [2]. Involvement of nitric oxide production in the impairment of skin blood flow response to local cooling in diabetic db/db mice. Eur J Pharmacol. 2013 Nov 15;720(1-3):174-9. |
| Additional Infomation |
α2-Adrenoceptors, subdivided into α2A, α2B, and α2C subtypes and expressed in heart, blood vessels, kidney, platelets and brain, are important for blood pressure, sedation, analgesia, and platelet aggregation. Brain α2C-adrenoceptor blockade has also been suggested to be beneficial for antipsychotic action. However, comparing α2-adrenoceptor subtype affinity is difficult due to significant species and methodology differences in published studies. Here, 3 H-rauwolscine whole cell binding was used to determine the affinity and selectivity of 99 α-antagonists (including antidepressants and antipsychotics) in CHO cells expressing human α2A, α2B, or α2C-adrenoceptors, using an identical method to β and α1-adrenoceptor measurements, thus allowing direct human receptor comparisons. Yohimbine, RX821002, RS79948, and atipamezole are high affinity non-selective α2-antagonists. BRL44408 was the most α2A-selective antagonist, although its α1A-affinity (81 nM) is only 9-fold greater than its α2C-affinity. MK-912 is the highest-affinity, most α2C-selective antagonist (0.15 nM α2C-affinity) although its α2C-selectivity is only 13-fold greater than at α2A. There are no truely α2B-selective antagonists. A few α-ligands with significant β-affinity were detected, for example, naftopidil where its clinical α1A-affinity is only 3-fold greater than off-target β2-affinity. Antidepressants (except mirtazapine) and first-generation antipsychotics have higher α1A than α2-adrenoceptor affinity but poor β-affinity. Second-generation antipsychotics varied widely in their α2-adrenoceptor affinity. Risperidone (9 nM) and paliperidone (14 nM) have the highest α2C-adrenoceptor affinity however this is only 5-fold selective over α2A, and both have a higher affinity for α1A (2 nM and 4 nM, respectively). So, despite a century of yohimbine use, and decades of α2-subtype studies, there remains plenty of scope to develop α2-subtype selective antagonists.[1] An enhanced vasocontrictor activity of cutaneous vessels participates in the reduction of skin blood flow induced by cooling. The present study investigated changes in the local response to cooling in hyperglycemic conditions. Male diabetic db/db and control C57BL/6J mice, anaesthetized with pentobarbitone, were treated with tetrodotoxin for eliminating the sympathetic nerve tone and artificially ventilated. The plantar skin blood flow (PSBF) was measured by laser Doppler flowmetry. Cooling the air temperature around the foot reduced PSBF in a temperature-dependent manner in control and db/db mice. The PSBF reduction was significantly smaller in db/db mice than in control mice. Phentolamine, a non-selective α-antagonist, bunazosin, a selective α1-antagonist, MK-912, a selective α2C-antagonist, and Y-27632, a Rho-kinase inhibitor, significantly inhibited the PSBF reduction induced by cooling to 15 °C in both mice and the inhibitory effects were comparable between these mice. The cooling-induced PSBF reduction was also significantly inhibited by N(ω)-nitro-L-arginine, an inhibitor of nitric oxide synthase, in control mice; however, the inhibitory effect of N(ω)-nitro-L-arginine was not observed in db/db mice. The reduction of PSBF induced by the intraarterial administration of adrenaline was comparable between control and db/db mice both before and after the treatment with N(ω)-nitro-L-arginine. It is thus likely that the reduction of skin blood flow induced by local cooling might be partly mediated by a decrease in endothelium-derived nitric oxide production, and that an impairment of the nitric oxide production might be related to reduced vasocontrictor response to cooling in db/db mice.[2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6604 mL | 13.3018 mL | 26.6035 mL | |
| 5 mM | 0.5321 mL | 2.6604 mL | 5.3207 mL | |
| 10 mM | 0.2660 mL | 1.3302 mL | 2.6604 mL |