Physicochemical Properties
| Molecular Formula | C20H13CL2N5O2 |
| Molecular Weight | 426.257 |
| Exact Mass | 425.045 |
| Elemental Analysis | C, 56.36; H, 3.07; Cl, 16.63; N, 16.43; O, 7.51 |
| CAS # | 920035-77-4 |
| PubChem CID | 16045418 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 5.019 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 29 |
| Complexity | 605 |
| Defined Atom Stereocenter Count | 0 |
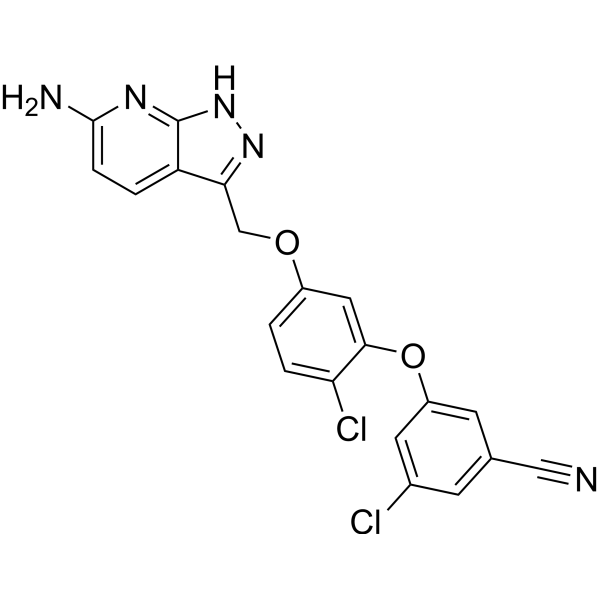
| SMILES | N#CC1=CC(OC2=CC(OCC3=NNC4=C3C=CC(N)=N4)=CC=C2Cl)=CC(Cl)=C1 |
| InChi Key | KXDIHAQCVNNLIB-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C20H13Cl2N5O2/c21-12-5-11(9-23)6-14(7-12)29-18-8-13(1-3-16(18)22)28-10-17-15-2-4-19(24)25-20(15)27-26-17/h1-8H,10H2,(H3,24,25,26,27) |
| Chemical Name | 3-[5-[(6-amino-2H-pyrazolo[3,4-b]pyridin-3-yl)methoxy]-2-chlorophenoxy]-5-chlorobenzonitrile |
| Synonyms | MK-4965; MK 4965; MK-4965; 920035-77-4; 3-(5-((6-Amino-1H-pyrazolo[3,4-b]pyridin-3-yl)methoxy)-2-chlorophenoxy)-5-chlorobenzonitrile; UNII-Z8856WDK9S; Z8856WDK9S; CHEMBL489586; 3-{5-[(6-amino-1H-pyrazolo[3,4-b]pyridin-3-yl)methoxy]-2-chlorophenoxy}-5-chlorobenzonitrile; Benzonitrile, 3-(5-((6-amino-1H-pyrazolo(3,4-b)pyridin-3-yl)methoxy)-2-chlorophenoxy)-5-chloro-; MK4965 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | NNRTI (nonnucleoside reverse transcriptase inhibitor); HIV-1 (IC50 = 0.23 nM for WT); 0.39 nM for Y181C; 0.40 nM for K103N |
| ln Vitro | Nonnucleoside reverse transcriptase inhibitors (NNRTIs) are the mainstays of therapy for the treatment of human immunodeficiency virus type 1 (HIV-1) infections. However, the effectiveness of NNRTIs can be hampered by the development of resistance mutations which confer cross-resistance to drugs in the same class. Extensive efforts have been made to identify new NNRTIs that can suppress the replication of the prevalent NNRTI-resistant viruses. MK-4965 is a novel NNRTI that possesses both diaryl ether and indazole moieties. The compound displays potency at subnanomolar concentrations against wild-type (WT), K103N, and Y181C reverse transcriptase (RT) in biochemical assays. MK-4965 is also highly potent against the WT virus and two most prevalent NNRTI-resistant viruses (viruses that harbor the K103N or the Y181C mutation), against which it had 95% effective concentrations (EC(95)s) of <30 nM in the presence of 10% fetal bovine serum. The antiviral EC(95) of MK-4965 was reduced approximately four- to sixfold when it was tested in 50% human serum. Moreover, MK-4965 was evaluated with a panel of 15 viruses with NNRTI resistance-associated mutations and showed a superior mutant profile to that of efavirenz but not to that of etravirine. MK-4965 was similarly effective against various HIV-1 subtypes and viruses containing nucleoside reverse transcriptase inhibitor or protease inhibitor resistance-conferring mutations. A two-drug combination study showed that the antiviral activity of MK-4965 was nonantagonistic with each of the 18 FDA-licensed drugs tested vice versa in the present study. Taken together, these in vitro data show that MK-4965 possesses the desired properties for further development as a new NNRTI for the treatment of HIV-1 infection. [1] |
| Enzyme Assay |
HIV-1 RT biochemical assay. [1] The ECL RT biochemical assay was performed on the basis of a previously described protocol. Briefly, HIV-1 RT enzyme (10 pM) was combined with an inhibitor or dimethyl sulfoxide (10%) in assay buffer (50 mM Tris-HCl, pH 7.8), and then the mixture was incubated at room temperature for 30 min. The polymerization reaction was initiated by the addition of a biotinylated template-primer substrate (5 nM) and deoxynucleoside triphosphates (0.6 μM of dATP, dGTP, and dCTP; 20 nM ruthenylated dUTP). The reactions were continued for 90 min at 37°C, followed by addition of 10 μl of 1 N NaOH to terminate the reaction. The resulting solution was incubated at room temperature for an additional 30 min and was then neutralized with 10 μl of 1 N HCl. Streptavidin-coated magnetic beads (80 μg/ml) were added to capture the product and the unreacted substrate. Quantification of the product was determined on the basis of the ECL signal measured with the ECL M-384 instrument. |
| Cell Assay |
HIV multiple-cycle replication assay.[1] MK-4965 was tested in cell culture multiple-cycle replication assays with genetically defined WT HIV-1 and three laboratory viruses with mutations (K103N, Y181C, and both K103N and Y181C). Antiviral activity assays were performed with a cell culture medium supplemented with 10% FBS or 50% human serum (HS). MK-4965 was diluted into an appropriate culture medium for each assay and was tested at concentrations ranging from 833 nM to 0.32 nM. Antiviral assays were performed with variants of a laboratory HIV-1 isolate, R8, and MT-4 human T-lymphoid cells. Cells (2.5 × 105 cells/well) were infected with viral stocks at a low multiplicity of infection in the absence of inhibitor and were incubated at 37°C in a 5% CO2 atmosphere. Twenty-four hours later, the infected cells were washed, resuspended in fresh culture medium (RPMI 1640 with 10% FBS or RPMI 1640 with 50% type AB HS), and seeded into a 96-well cell culture plate at 5 × 104 cells per 125 μl. MK-4965 was added to the wells in serial threefold dilutions. The NNRTIs EFV, NVP, and ETV were used as comparators in the assay. The cells were incubated for an additional 72 h, and virus production in the individual culture wells was assessed by measuring the level of production of viral core p24 antigen in the culture medium with an Alphascreen kit from Perkin-Elmer. The 95% effective concentration (EC95) for virus inhibition was defined as the lowest concentration of test compound that inhibited virus p24 antigen production by at least 95% relative to the level of production by the untreated control cultures. The cytotoxicities of MK-4965 and the comparator drugs at the concentrations tested (see above) were assessed by using Alamar blue, as recommended by the manufacturer, after incubation at 37°C in 5% CO2 for 24, 48, or 72 h. MK-4965 did not show any cytotoxicity even at concentrations much higher than its EC95 (EC50 for cytotoxicity, 2.8 μM). HIV-1 single-cycle replication assay with viruses containing RT sequences from patient isolates.[1] Monogram Biosciences performed a single-cycle replication PhenoScreen assay with MK-4965 and EFV against a broader array of clinical NNRTI-resistant HIV-1 isolates using a variation of its clinical diagnostic assays. In addition, nine HIV-1 sybtypes and circulating recombinant forms (CRFs) were also evaluated for their susceptibilities to NNRTIs. Clade B viruses containing PI or NRTI resistance-conferring mutations were tested to determine whether the mutations confer any resistance to NNRTIs. RT inhibitor activity was evaluated with nine serial drug concentrations. Combination study. (i) General information.[1] The combination study was conducted by SRI in a two-drug combination fashion with MK-4965 and each of the 18 FDA-approved antiviral agents described above, including RAL and ETV. RBV and d4T were used as the positive antagonism controls because the antiviral activity of d4T is antagonized by RBV. The cells were quantified with a hemacytometer, and cell viability was determined by trypan blue exclusion. Cell viability was greater than 95% under the conditions utilized in the assay. For the study of each combination, MK-4965 was prepared by starting with 100 nM and then obtaining seven additional serial half-log dilutions. Each dilution of MK-4965 was tested in combination with five dilutions of a second anti-HIV drug starting from 10 times the EC50, followed by testing with four additional half-log dilutions. The dimethyl sulfoxide concentration was <0.25%, which has previously been shown to have no effect in the assays described. |
| References |
[1]. Antiviral activity of MK-4965, a novel nonnucleoside reverse transcriptase inhibitor. Antimicrob Agents Chemother. 2009 Jun;53(6):2424-31. |
| Additional Infomation |
MK-4965 is an HIV-1 reverse transcriptase inhibitor. In summary, MK-4965 is a novel NNRTI containing diaryl ether and indazole moieties. It exhibits excellent activity against RT enzymes containing K103N or Y181C mutations and also against viruses containing the same RT mutations. All HIV subtypes tested in this study had similar susceptibilities to MK-4965, and PI and NRTI resistance-conferring mutations did not compromise the antiviral activity of MK-4965. Given the absence of antagonistic effects with the other antiviral agents tested in the combination study, MK-4965 has in vitro properties consistent with the high potential to be a new NNRTI for treatment of HIV-1 infection. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3460 mL | 11.7299 mL | 23.4599 mL | |
| 5 mM | 0.4692 mL | 2.3460 mL | 4.6920 mL | |
| 10 mM | 0.2346 mL | 1.1730 mL | 2.3460 mL |