MDL-29951 is a potent glycine antagonist of NMDA (N-methyl-D-aspartate) receptor activation with Ki value of 0.14 μM in an in vitro and in vivo [3H]glycine binding assay. MDL 100,748 and MDL 29,951 are approximately 2000-fold selective for the glycine binding site relative to the glutamate recognition sites.The MDL29951-activated receptor exhibits other activities associated with GPCR-mediated signaling, including G protein-dependent activation of extracellular signal-regulated kinase 1 and 2 (ERK1/2) and recruitment of β-arrestin. MDL29951 did not activate any of the known uracil or adenine nucleotide-activated P2Y receptors or cysteinyl leukotriene receptors. Gαi- and Gαq-dependent signaling responses also were observed in primary rat oligodendrocytes in the presence of MDL29951. Moreover, MDL29951 diminished myelination in primary oligodendrocytes isolated from heterozygous mice but had no effect on myelination in oligodendrocytes from GPR17 knockout mice. Effects of a small-molecule GPR17 agonist observed during oligodendrocyte differentiation support the idea that development of antagonists of GPR17 is a rational goal for elaboration of pharmacotherapies in demyelinating diseases.
Physicochemical Properties
| Molecular Formula | C12H9CL2NO4 | |
| Molecular Weight | 302.11 | |
| Exact Mass | 300.99 | |
| CAS # | 130798-51-5 | |
| Related CAS # |
|
|
| PubChem CID | 446916 | |
| Appearance | White to off-white solid powder | |
| Density | 1.7±0.1 g/cm3 | |
| Boiling Point | 582.5±45.0 °C at 760 mmHg | |
| Flash Point | 306.1±28.7 °C | |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C | |
| Index of Refraction | 1.707 | |
| LogP | 3.13 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 4 | |
| Heavy Atom Count | 19 | |
| Complexity | 379 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | KNBSYZNKEAWABY-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C12H9Cl2NO4/c13-5-3-7(14)10-6(1-2-9(16)17)11(12(18)19)15-8(10)4-5/h3-4,15H,1-2H2,(H,16,17)(H,18,19) | |
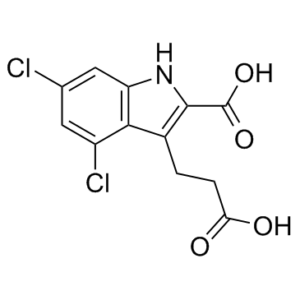
| Chemical Name | 3-(2-carboxyethyl)-4,6-dichloro-1H-indole-2-carboxylic acid | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
N-methyl-D-aspartate (NMDA) receptor (strychnine-insensitive glycine binding site, Ki = 0.14 μM; ~2000-fold selective for glycine binding site relative to glutamate recognition sites) [1] Fructose 1,6-bisphosphatase (AMP regulatory site, allosteric inhibitor) [2] GPR17 (G protein-coupled receptor, activator of Gαi- and Gαq-promoted signaling pathways) [3] |
| ln Vitro |
The glycine binding site is about 2000 times more selective than the glutamate recognition site for MDL 100,748 and MDL 29,951 [1]. MDL-29951 has an IC50 value of 2.5 μM, which inhibits human F16Bpase. MDL-29951 is less effective against the rat liver isoform (IC50=11 μM) but inhibits the enzyme isoforms of the human liver (IC50=2.5 μM), pig kidney (IC50=1.0 μM), and rabbit liver (IC50=0.21 μM)[2]. In addition to these GPCR-mediated signaling functions, MDL29951-activated receptors also recruit beta-arrestin and activate extracellular signal-regulated kinases 1 and 2 (ERK1/2) in a G protein-dependent manner. Similar to the recombinant cell system, MDL29951 stimulates rat oligodendrocyte precursor cells to accumulate cyclic adenosine monophosphate (cAMP) and inhibit Ca2+ signaling responses during the peak of GPR17 abundance. In cells with low GPR17 abundance, MDL29951's effect was significantly reduced, and pranlukast blocked it [3]. 1. For NMDA receptor-related activity: MDL-29951 potently inhibited [3H]glycine binding to the strychnine-insensitive glycine binding site of NMDA receptor with a Ki value of 0.14 μM; it completely inhibited the use-dependent binding of [3H]N-[1-(2-thienyl)cyclohexyl]-piperidine to NMDA receptor; it acted as a noncompetitive, glycine-reversible inhibitor of NMDA-induced biochemical and electrophysiological responses in brain slice preparations; in cultured neuronal preparations, it produced parallel rightward shifts in the glycine requirement for NMDA-stimulated elevations in cytosolic calcium, indicating a competitive interaction with the glycine binding site [1] 2. For fructose 1,6-bisphosphatase inhibition: MDL-29951 was identified as an allosteric inhibitor of fructose 1,6-bisphosphatase, and X-ray crystallography confirmed that it binds to the AMP regulatory site of this enzyme [2] 3. For GPR17 activation: MDL-29951 induced Gαi- and Gαq-dependent signaling responses in cell lines expressing recombinant GPR17; it also triggered Gαi- and Gαq-dependent signaling responses in primary rat oligodendrocytes; it did not activate known uracil/adenine nucleotide-activated P2Y receptors or cysteinyl leukotriene receptors [3] |
| ln Vivo |
1. Anticonvulsant activity: Intracerebroventricular (i.c.v.) administration of MDL-29951 to audiogenic seizure-susceptible DBA/2J mice exhibited potent anticonvulsant effects [1] 2. Effect on myelination: MDL-29951 diminished myelination in primary oligodendrocytes isolated from heterozygous mice, while it had no effect on myelination in oligodendrocytes from GPR17 knockout mice [3] |
| Enzyme Assay |
1. [3H]glycine binding assay for NMDA receptor: The assay was conducted to measure the inhibitory effect of MDL-29951 on glycine binding to NMDA receptor; samples containing the receptor preparation, [3H]glycine, and different concentrations of MDL-29951 were incubated under specific conditions, followed by separation of bound and free radioligand to determine the Ki value of MDL-29951 for the glycine binding site [1] 2. Fructose 1,6-bisphosphatase activity assay and X-ray crystallography: Enzyme activity assays were performed to evaluate the inhibitory effect of MDL-29951 on fructose 1,6-bisphosphatase; X-ray crystallography was used to determine the binding site of MDL-29951 on the enzyme, confirming its binding to the AMP regulatory site as an allosteric inhibitor [2] 3. GPR17 signaling assay: Cell lines expressing recombinant GPR17 were treated with MDL-29951, and the activation of Gαi- and Gαq-promoted signaling pathways was detected through relevant signaling readouts; primary rat oligodendrocytes were also treated with MDL-29951 to assess Gαi- and Gαq-dependent signaling responses [3] |
| Cell Assay |
1. Cultured neuronal cells assay: Cultured neuronal preparations were exposed to MDL-29951 and NMDA, and the cytosolic calcium levels were measured to evaluate the effect of MDL-29951 on NMDA-stimulated calcium elevation; the experiment demonstrated that MDL-29951 caused parallel rightward shifts in the glycine requirement for such calcium elevation [1] 2. Recombinant GPR17-expressing cell lines assay: These cell lines were incubated with MDL-29951 (and control agonists including uracil nucleotides and cysteinyl leukotrienes), and the activation of Gαi- and Gαq signaling pathways was assessed using appropriate detection methods; the results showed that MDL-29951 activated Gαi- and Gαq signaling, while control agonists had no effect [3] 3. Primary rat oligodendrocytes assay: Primary rat oligodendrocytes were treated with MDL-29951, and Gαi- and Gαq-dependent signaling responses were measured; additionally, oligodendrocytes from heterozygous and GPR17 knockout mice were treated with MDL-29951 to assess the impact on myelination [3] |
| Animal Protocol |
1. Audiogenic seizure model in DBA/2J mice: MDL-29951 was administered intracerebroventricularly (i.c.v.) to audiogenic seizure-susceptible DBA/2J mice; the anticonvulsant effect of the drug was evaluated by observing the seizure response of the mice after acoustic stimulation [1] 2. Oligodendrocyte myelination assay in mice: Primary oligodendrocytes were isolated from heterozygous mice and GPR17 knockout mice; these oligodendrocytes were treated with MDL-29951 in vitro, and the myelination status was assessed to determine the drug's effect [3] |
| References |
[1]. Potent indole- and quinoline-containing N-methyl-D-aspartate antagonists acting at the strychnine-insensitive glycine binding site. J Pharmacol Exp Ther. 1992 Sep;262(3):947-56. [2]. 3-(2-carboxyethyl)-4,6-dichloro-1H-indole-2-carboxylic acid: an allosteric inhibitor of fructose-1,6-bisphosphatase at the AMP site. Bioorg Med Chem Lett. 2003 Jun 16;13(12):2055-8. [3]. Harden TK. Enigmatic GPCR finds a stimulating drug. Sci Signal. 2013 Oct 22;6(298):pe34. |
| Additional Infomation |
1. MDL-29951 is an indole-containing compound, and together with MDL 100,748 (a quinoline-containing compound), it is a novel glycine antagonist of NMDA receptor activation; the results of relevant studies indicate that glycine is required for NMDA receptor activation, and bioavailable glycine antagonists like MDL-29951 may form the basis of a novel therapy for epilepsy [1] 2. MDL-29951 represents a new approach to inhibition of fructose 1,6-bisphosphatase and can serve as a lead compound for further drug design targeting this enzyme [2] 3. GPR17 is an orphan G protein-coupled receptor involved in oligodendrocyte differentiation and myelination in the central nervous system; the activation of GPR17 by MDL-29951 supports the development of GPR17 antagonists as a rational goal for pharmacotherapies in demyelinating diseases [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.28 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.28 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (8.28 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3101 mL | 16.5503 mL | 33.1005 mL | |
| 5 mM | 0.6620 mL | 3.3101 mL | 6.6201 mL | |
| 10 mM | 0.3310 mL | 1.6550 mL | 3.3101 mL |