Physicochemical Properties
| Molecular Formula | C25H37NO5S |
| Molecular Weight | 463.63 |
| Exact Mass | 463.239 |
| CAS # | 1784701-63-8 |
| PubChem CID | 122368873 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 693.1±55.0 °C at 760 mmHg |
| Flash Point | 372.9±31.5 °C |
| Vapour Pressure | 0.0±4.9 mmHg at 25°C |
| Index of Refraction | 1.571 |
| LogP | 5.99 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 18 |
| Heavy Atom Count | 32 |
| Complexity | 694 |
| Defined Atom Stereocenter Count | 3 |
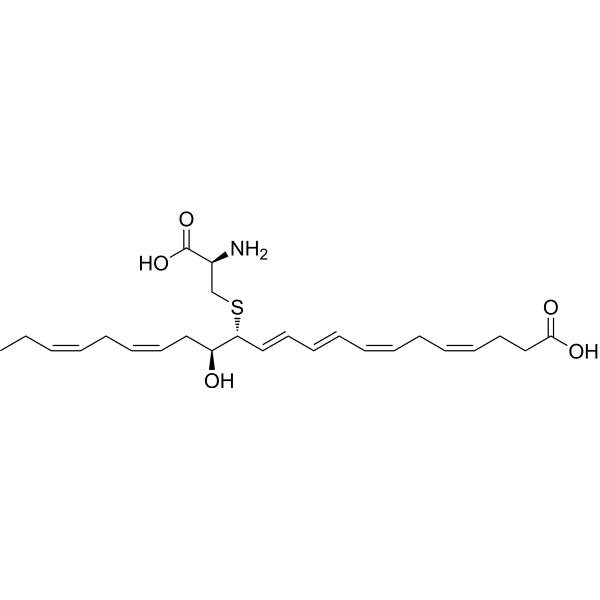
| SMILES | CC/C=C\C/C=C\C[C@@H]([C@@H](/C=C/C=C/C=C\C/C=C\CCC(=O)O)SC[C@@H](C(=O)O)N)O |
| InChi Key | GIIVKOKEBBFSDI-AIYHDIOKSA-N |
| InChi Code | InChI=1S/C25H37NO5S/c1-2-3-4-5-11-14-17-22(27)23(32-20-21(26)25(30)31)18-15-12-9-7-6-8-10-13-16-19-24(28)29/h3-4,6-7,9-15,18,21-23,27H,2,5,8,16-17,19-20,26H2,1H3,(H,28,29)(H,30,31)/b4-3-,7-6-,12-9+,13-10-,14-11-,18-15+/t21-,22-,23+/m0/s1 |
| Chemical Name | (4Z,7Z,9E,11E,13R,14S,16Z,19Z)-13-[(2R)-2-amino-2-carboxyethyl]sulfanyl-14-hydroxydocosa-4,7,9,11,16,19-hexaenoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | MCTR3 (1 nM) alleviates LPS-induced cell damage and oxidative stress in MLE12 cells through the ALX/PINK1 pathway and reduces the production of apoptosis- and autophagy-related proteins[1]. MCTR3 (1 nM) enhances endocytosis and bacterial phagocytosis of infiltrated leukocytes and reduces neutrophil infiltration and eicosanoid levels[2]. |
| ln Vivo | MCTR3 (2 ng/g, intraperitoneal injection, single dose) alleviates LPS-induced acute lung injury in C57BL/6 mice, improves inflammatory and oxidative responses, and reduces apoptosis[1]. MCTR3 (1-100 nM, in cold water) stimulates surgically injured planarian tissue regeneration in a dose-dependent manner, with 50% tissue regeneration (TRI50) taking 2.5 days[2]. |
| Cell Assay |
Immunofluorescence[1] Cell Types: MLE12 Concentration: 1 nM Incubation Duration: 24 h Experimental Results: Decreased LPS-stimulated expressions of PINK1 and Parkin. Western Blot Analysis[1] Cell Types: MLE12 Concentration: 1 nM Incubation Duration: 24 h Experimental Results: Decreased levels of Bax, Bak, Cyto C and LC3BII/I, increased levels of BCL2 and Bcl-xl. |
| Animal Protocol |
Animal/Disease Models:Lipopolysaccharide induced acute lung injury in C57BL6 mice[1] Doses: 2 ng/g Route of Administration: i.p., single dose Experimental Results: Reduced inflammatory cell infiltration and protein accumulation in bronchoalveolar lavage fluid (BALF), reduced MDA levels and increased SOD activity in lung tissue. Decreased levels of pro-inflammatory cytokine in serum. |
| References |
[1]. MCTR3 reduces LPS-induced acute lung injury in mice via the ALX/PINK1 signaling pathway. Int Immunopharmacol. 2021 Jan;90:107142. [2]. Identification and Actions of a Novel Third Maresin Conjugate in Tissue Regeneration: MCTR3. PLoS One. 2016 Feb 16;11(2):e0149319. [3]. , MCTR3 reprograms arthritic monocytes to upregulate Arginase-1 and exert pro-resolving and tissue-protective functions in experimental arthritis. EBioMedicine. 2022 May;79:103974. |
| Additional Infomation | (13R)-S-cysteinyl-(14S)-hydroxy-(4Z,7Z,9E,11E,16Z,19Z)-docosahexaenoic acid is a docosanoid that is (14S)-hydroxy-(4Z,7Z,9E,11E,16Z,19Z)-docosahexaenoic acid in which a cysteinyl group is attached at position 13R via a sulfide linkage. It has a role as a human metabolite. It is a dicarboxylic acid, a docosanoid, an organic sulfide, a secondary alcohol and a S-substituted L-cysteine. It is a conjugate acid of a (13R)-S-cysteinyl-(14S)-hydroxy-(4Z,7Z,9E,11E,16Z,19Z)-docosahexaenoate(1-). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1569 mL | 10.7845 mL | 21.5689 mL | |
| 5 mM | 0.4314 mL | 2.1569 mL | 4.3138 mL | |
| 10 mM | 0.2157 mL | 1.0784 mL | 2.1569 mL |