Physicochemical Properties
| Molecular Formula | C5H12NO5P |
| Molecular Weight | 195.1104 |
| Exact Mass | 197.045 |
| Elemental Analysis | C, 30.47; H, 6.14; N, 7.11; O, 40.58; P, 15.71 |
| CAS # | 157381-42-5 |
| PubChem CID | 1795545 |
| Appearance | Typically exists as solid at room temperature |
| Boiling Point | 472.1ºC at 760mmHg |
| Flash Point | 239.3ºC |
| Vapour Pressure | 3.25E-10mmHg at 25°C |
| Index of Refraction | 1.538 |
| LogP | 0.056 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 12 |
| Complexity | 224 |
| Defined Atom Stereocenter Count | 1 |
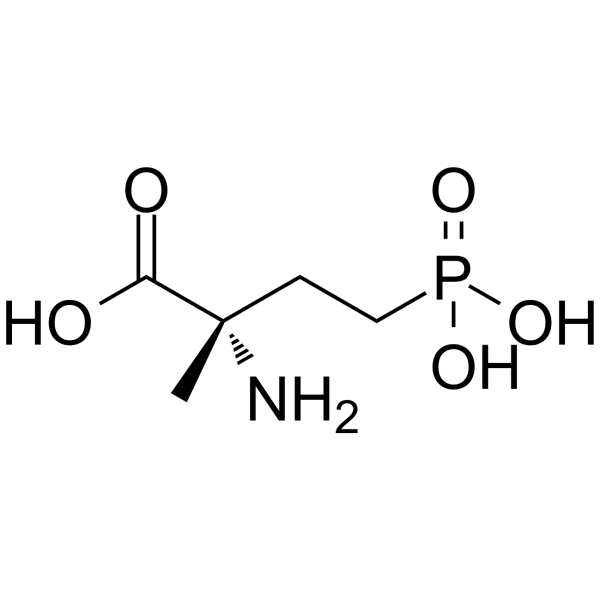
| SMILES | NC(C)(CCP(O)(O)=O)C(O)=O |
| InChi Key | HONKEGXLWUDTCF-YFKPBYRVSA-N |
| InChi Code | InChI=1S/C5H12NO5P/c1-5(6,4(7)8)2-3-12(9,10)11/h2-3,6H2,1H3,(H,7,8)(H2,9,10,11)/t5-/m0/s1 |
| Chemical Name | (2S)-2-amino-2-methyl-4-phosphonobutanoic acid |
| Synonyms | MAP 4; MAP-4; 157381-42-5; (S)-2-Amino-2-methyl-4-phosphonobutanoic acid; L-Isovaline, 4-phosphono-; (2S)-2-amino-2-methyl-4-phosphonobutanoic acid; MAP4 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | mGluR |
| ln Vivo | Antagonism of L-AP4 and CCG 1 by ~-methyl-L-AP4 and ~-methyl-CCG 1 Twenty neurons where L-AP4 and/or CCG1 produced a disinhibition were studied further with the novel antagonists MAP4 and MCCG. 23 In these cases, L-AP4 or CCGI were initially applied to determine ejection parameters which produced disinhibition. An antagonist was then applied before, during and after a subsequent application of the agonist at the same ejection parameters which previously produced disinhibition. In this way it was possible to assess whether the antagonists had any direct effect, before assessing their antagonism of agonist-evoked disinhibition. The disinhibitory action of L-AP4 was challenged with MAP4 (51 _+ 4.5 nA) on eight neurons, and in all of these cases an antagonism of the L-AP4 action was seen without any apparent direct effect of the antagonist (Figs 1, 3A). In order to verify the specificity of the antagonist effects, it was important to investigate whether CCG1 was also susceptible to antagonism by MAP4. This was possible in four of these eight experiments, and in these cases MAP4 did not antagonize the disinhibition produced by CCGI (Fig. 3B), even though the antagonist had been effective against L-AP4 on the same neurons, thus showing that MAP4 was selective as an antagonist of L-AP4 effects. The antagonism of CCGl by MCCG was investigated on 12 neurons. In all of these, the disinhibition produced by CCG1 was reduced by iontophoretically-applied MCCG (68 _+ 9,4 nA) (Figs 2, 3D), but it was also found that this antagonist had a small, direct disinhibitory action in its own right on four of these neurons. MCCG was also tested against the disinhibitory actions of L-AP4 on five of the 12 neurons, and it also reduced the effects of this agonist, although to a lesser extent than the antagonism of CCG1 (Fig. 3C), thus indicating that it had a degree of selectivity towards CCG1 [2]. |
| Animal Protocol | Experiments were carried out in Wistar rats anaesthetized with urethane (l.2g/kg, i.p.) as detailed previously. 31"32 Extracellular recordings of single VB neuron activity were made through the central barrel (filled with 4 M NaC1) of seven-barrel glass iontophoretic microelectrodes. In some experiments, intracellular recordings were made with a pipette which was cemented alongside an iontophoretic pipette so as to protrude by about 50 pm beyond the tip of the iontophoretic pipette. The intracellular pipettes were filled with 1.2 M potassium citrate, and had impedances of 80 100 M~ before entry into the tissue. Detailed procedures for the intracellular recording experiments have been published previously. ~3'2° In both types of experiment, each iontophoretic barrel contained one of the following substances: CCG1 [(2S,3S,4S)-~-(carboxycyclopropyl)- glycine], N-methyl-D-aspartate, MAP4 (~-methyl-L-AP4), MCCG (~-methyl-CCG1), (IS,3R)-l-amino-l,3-cyclopentane dicarboxylate (all 50raM in water, as Na + salts, pH 8.0 8.5), L-AP4 [(S)-2-amino-4-phosphonobutyrate] (25 mM in water, pH 8.0-8.5), 1 M NaC1 (for current balancing) and Pontamine Sky Blue dye (2.5% in 0.5 M NaC1/0.5 M sodium acetate). All drugs were ejected iontophoretically as anions, and prevented from diffusing out of the pipette by a retaining current (10 20 nA) of opposite polarity to the ejection current. Sensory stimulation was carried out using electronically gated air jets (10 ms duration), which could be directed at a single facial vibrissa. Previous intracellular recordings from this laboratory have shown that an air jet directed at the vibrissa(e) of the excitatory receptive field typically evokes an EPSP (excitatory postsynaptic potential), action potential, IPSP (inhibitory postsynaptic potential) sequence. 33 As an alternative means of evoking IPSPs in VB neurons, electrical stimulation of the somatosensory cortex was performed, as it is well known that this is able to activate inhibitory processes within the thalamus? ,s'"~ Two silver wires were placed over the vibrissal representation of the somatosensory cortex, 2 3 mm apart, and stimulation was carried out by passing single current pulses between these electrodes (current range- 1.0 3.0 mA: duration0.1 ms). [1] |
| References |
[1]. Salt TE, Eaton SA. Distinct presynaptic metabotropic receptors for L-AP4 and CCG1 on GABAergic terminals: pharmacological evidence using novel alpha-methyl derivative mGluR antagonists, MAP4 and MCCG, in the rat thalamus in vivo. Neuroscience. 1995 Mar;65. |
| Additional Infomation |
High molecular weight proteins found in the MICROTUBULES of the cytoskeletal system. Under certain conditions they are required for TUBULIN assembly into the microtubules and stabilize the assembled microtubules.
A variety of metabotropic excitatory amino acid receptors are present in the thalamus. We have investigated the possibility that some of these receptors may have presynaptic effects on GABAergic inhibitory transmission in the thalamus. Inhibitory responses in ventrobasal thalamic neurons of urethane-anaesthetized rats were evoked by either air-jet stimuli to the vibrissae or by electrical stimulation of the somatosensory cortex. Both intracellular and extracellular recording methods were used to reveal inhibitory responses, either as inhibitory postsynaptic potentials or inhibition of excitatory responses in a condition-test paradigm. The metabotropic glutamate receptor agonists (S)-2-amino-4-phosphonobutyrate (L-AP4) and (2S,3S,4S)-alpha-(carboxycyclopropyl)-glycine (CCG1), applied in the vicinity of the recording site by iontophoresis, were found to reduce the amplitudes of inhibitory postsynaptic potentials (to 76% and 63% of control amplitudes, respectively) and inhibitions revealed by the condition-test paradigm (to 33% and 28% of control inhibitions, respectively). As the inhibitory responses arise from the neurons of the nucleus reticularis thalami, some distance away from the site of recording and iontophoretic drug application, it is likely that the reduction of inhibition seen with L-AP4 and CCG1 is due to an action of these agonists on the terminals or axons of these inhibitory neurons. The novel antagonists of L-AP4 and CCG1, alpha-methyl-L-AP4 and alpha-methyl-CCG1, were found to block the disinhibitory actions of the agonists in a differential manner when applied iontophoretically. This suggests that there may be at least two types of receptor mediating the disinhibitory effects [2]. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.1253 mL | 25.6266 mL | 51.2531 mL | |
| 5 mM | 1.0251 mL | 5.1253 mL | 10.2506 mL | |
| 10 mM | 0.5125 mL | 2.5627 mL | 5.1253 mL |