Physicochemical Properties
| Molecular Formula | C18H17N3O5S2 |
| Molecular Weight | 419.4747 |
| Exact Mass | 419.06 |
| CAS # | 1359431-16-5 |
| PubChem CID | 54733438 |
| Appearance | White to off-white solid powder |
| LogP | 2.1 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 28 |
| Complexity | 799 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | S1C2=C(C(=C(C(N([H])C3C([H])=C([H])C(=C([H])C=3[H])S(N([H])[H])(=O)=O)=O)C(N2[H])=O)O[H])C2=C1C([H])([H])C([H])([H])C([H])([H])C2([H])[H] |
| InChi Key | MVQJFYRZMPKNIV-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H17N3O5S2/c19-28(25,26)10-7-5-9(6-8-10)20-16(23)14-15(22)13-11-3-1-2-4-12(11)27-18(13)21-17(14)24/h5-8H,1-4H2,(H,20,23)(H2,19,25,26)(H2,21,22,24) |
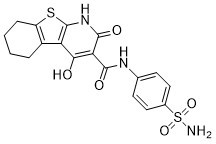
| Chemical Name | 4-hydroxy-2-oxo-N-(4-sulfamoylphenyl)-5,6,7,8-tetrahydro-1H-[1]benzothiolo[2,3-b]pyridine-3-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | HGC27, AGS, and MKN45 cell growth is considerably inhibited by M435-1279 (0, 2, 4, 8, 16, and 31 μM; 48 hours) [1]. M435-1279 (0, 4, 8, 12, 16, and 20 μM) reduces the viability of cells in GES-1, HGC27, MKN45, and AGS cells, respectively, with IC50 values of 16.8, 11.88, 6.93, and 7.76 μM [1]. M435-1279 (31 nM to 500 μM) binds to UBE2T with a KD value of 50.5 μM. In 48 hours, M435-1279 (11.88 μM) prevents excessive activation of Wnt/β-catenin staining and RACK1 ubiquitination [1]. |
| ln Vivo | M435-1279 (5 mg/kg/day; intratumoral injection for 18 days) suppresses tumor growth [1]. M435-1279 causes increased RACK1 protein expression and direct Ki-67 and β-catenin protein expression in intratumoral tumors |
| Cell Assay |
Cell viability assay [1] Cell Types: HGC27, AGS and MKN45 Cell Tested Concentrations: 0, 4, 8, 12, 16, 20 μM Incubation Duration: Experimental Results: Inhibited cell viability, IC50 was 16.8, 11.88, 6.93, 7.76 μM respectively. in GES-1, HGC27, MKN45, and AGS cells. |
| Animal Protocol |
Animal/Disease Models: BALB/C nude mouse CDX model (MKN45 tumor size: 75–100 mm3) [1 ] Doses: 5 mg/kg/day Route of Administration: Intratumoral injection for 18 days Experimental Results: Slowed tumor growth. Induced higher RACK1 protein expression and diminished Ki-67 and β-catenin protein expression in intratumoral tumors. |
| References |
[1]. A novel UBE2T inhibitor suppresses Wnt/β-catenin signaling hyperactivation and gastric cancer progression by blocking RACK1 ubiquitination. Oncogene. 2021 Feb;40(5):1027-1042. doi: 10.1038/s41388-020-01572-w. Epub 2020 Dec 15. Erratum in: Oncogene. 2021 Apr;40(14):2622-2623. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~119.20 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.96 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3840 mL | 11.9198 mL | 23.8396 mL | |
| 5 mM | 0.4768 mL | 2.3840 mL | 4.7679 mL | |
| 10 mM | 0.2384 mL | 1.1920 mL | 2.3840 mL |