Physicochemical Properties
| Molecular Formula | C21H18O12 |
| Molecular Weight | 462.3604 |
| Exact Mass | 462.079 |
| Elemental Analysis | Because of the widespread use of botanicals, it has become crucial for health professionals to improve their knowledge about safety problems. Several herbal medicines contain chemicals with allergenic properties responsible for contact dermatitis. Among these, one is Rosmarinus officinalis L. (rosemary), a plant used since ancient times in folk medicine; at the present time it is used worldwide as a spice and flavouring agent, as a preservative and for medicinal and cosmetic purposes. The present article aims to revise and summarise scientific literature reporting cases of contact dermatitis caused by the use of R. officinalis as a raw material or as herbal preparations. Published case reports were researched on the following databases and search engines: PUBMED, MEDLINE, EMBASE, Google Scholar, Scopus. The used keywords were: R. officinalis and rosemary each alone or combined with the words allergy, contact dermatitis, allergic contact dermatitis, sensitisation and occupational dermatitis. The published case reports show that both rosemary extracts and raw material can be responsible for allergic contact dermatitis. Two cases related to contact dermatitis caused by cross-reactivity between rosemary and thyme were also commented. The diterpene carnosol, a chemical constituent of this plant, has been imputed as a common cause for this reaction. The incidence of contact dermatitis caused by rosemary is not common, but it could be more frequent with respect to the supposed occurrence. It seems plausible that cases of contact dermatitis caused by rosemary are more frequent with respect to the supposed occurrence, because they could be misdiagnosed. For this reason, this possibility should be carefully considered in dermatitis differential diagnosis.https://pubmed.ncbi.nlm.nih.gov/23827646/ |
| CAS # | 53527-42-7 |
| PubChem CID | 10253785 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.8±0.1 g/cm3 |
| Boiling Point | 865.4±65.0 °C at 760 mmHg |
| Flash Point | 305.9±27.8 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.764 |
| LogP | -1.16 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 33 |
| Complexity | 785 |
| Defined Atom Stereocenter Count | 5 |
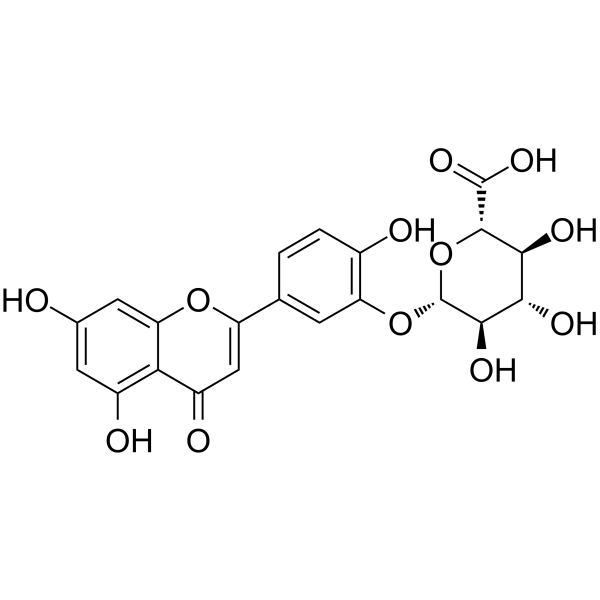
| SMILES | C1=CC(=C(C=C1C2=CC(=O)C3=C(C=C(C=C3O2)O)O)O[C@H]4[C@@H]([C@H]([C@@H]([C@H](O4)C(=O)O)O)O)O)O |
| InChi Key | JDOFZOKGCYYUER-ZFORQUDYSA-N |
| InChi Code | InChI=1S/C21H18O12/c22-8-4-10(24)15-11(25)6-12(31-14(15)5-8)7-1-2-9(23)13(3-7)32-21-18(28)16(26)17(27)19(33-21)20(29)30/h1-6,16-19,21-24,26-28H,(H,29,30)/t16-,17-,18+,19-,21+/m0/s1 |
| Chemical Name | (2S,3S,4S,5R,6S)-6-[5-(5,7-dihydroxy-4-oxochromen-2-yl)-2-hydroxyphenoxy]-3,4,5-trihydroxyoxane-2-carboxylic acid |
| Synonyms | Luteolin 3'-o-glucuronide; Luteolin-3-O-beta-D-glucuronide; Luteolin-3'-D-glucuronide; Luteolin 3'-glucuronide; Luteolin 3'-o-beta-D-glucuronide; UNII-1T6AU6J856; 1T6AU6J856; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural flavone; anti-inflammatory, anti-tumor, anti-oxidant, neuroprotective, anti-fungal activities |
| ln Vitro | Luteolin 3'-O-glucuronide is a luteolin glucosiduronic acid consisting of luteolin having a beta-D-glucosiduronic acid residue attached at the 3'-position. It has a role as a metabolite. It is a luteolin O-glucuronoside and a trihydroxyflavone. Luteolin 3'-o-glucuronide has been reported in Salvia officinalis, Melissa officinalis, and other organisms |
| References |
[1]. Rosmarinus officinalis L. as cause of contact dermatitis. Allergol Immunopathol (Madr). Nov-Dec 2014;42(6):616-9. |
| Additional Infomation |
Luteolin 3'-O-glucuronide is a luteolin glucosiduronic acid consisting of luteolin having a beta-D-glucosiduronic acid residue attached at the 3'-position. It has a role as a metabolite. It is a luteolin O-glucuronoside and a trihydroxyflavone. Luteolin 3'-o-glucuronide has been reported in Salvia officinalis, Melissa officinalis, and other organisms with data available. See also: Rosemary (part of). |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~216.28 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.50 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.50 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.50 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1628 mL | 10.8141 mL | 21.6282 mL | |
| 5 mM | 0.4326 mL | 2.1628 mL | 4.3256 mL | |
| 10 mM | 0.2163 mL | 1.0814 mL | 2.1628 mL |