Physicochemical Properties
| Molecular Formula | C21H27N5O7 |
| Molecular Weight | 461.468384981155 |
| Exact Mass | 461.191 |
| CAS # | 1435784-14-7 |
| Related CAS # | Lometrexol;106400-81-1;Lometrexol disodium;120408-07-3;LY243246;106400-18-4 |
| PubChem CID | 136985357 |
| Appearance | White to off-white solid powder |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 33 |
| Complexity | 831 |
| Defined Atom Stereocenter Count | 2 |
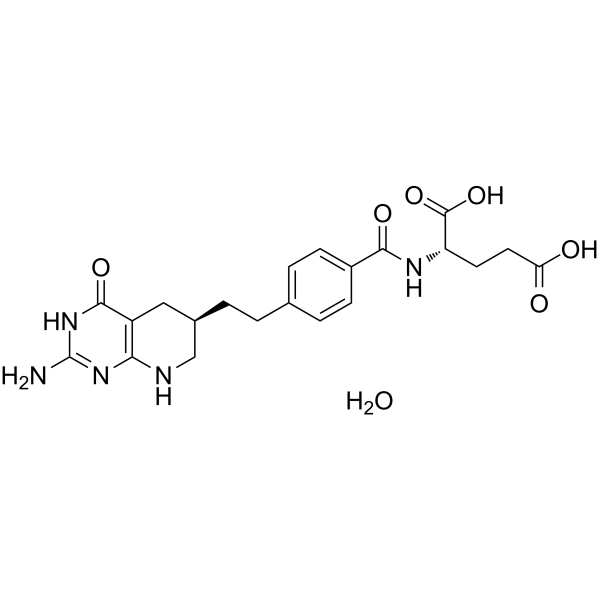
| SMILES | O=C1C2=C(N=C(N)N1)NC[C@H](CCC1C=CC(C(N[C@H](C(=O)O)CCC(=O)O)=O)=CC=1)C2.O |
| InChi Key | AEFQSKJUVDZANQ-YLCXCWDSSA-N |
| InChi Code | InChI=1S/C21H25N5O6.H2O/c22-21-25-17-14(19(30)26-21)9-12(10-23-17)2-1-11-3-5-13(6-4-11)18(29)24-15(20(31)32)7-8-16(27)28;/h3-6,12,15H,1-2,7-10H2,(H,24,29)(H,27,28)(H,31,32)(H4,22,23,25,26,30);1H2/t12-,15+;/m1./s1 |
| Chemical Name | (2S)-2-[[4-[2-[(6R)-2-amino-4-oxo-5,6,7,8-tetrahydro-3H-pyrido[2,3-d]pyrimidin-6-yl]ethyl]benzoyl]amino]pentanedioic acid;hydrate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | When hemethoxyl (DDATHF) hydration binds firmly to GART, intracellular purine ribose labels are rapidly and persistently depleted [3]. L1210 cells experience quick and total growth suppression when exposed to 1–30 μM of hydrate for 2–10 hours [3]. Hydro-induced chemotherapeutic cell cycle solution in L1210 cells using 1 μM of methorexol [3]. |
| ln Vivo | On day 7.5 of pregnancy, lometrexol (DDATHF; i.p. ; 15–60 mg/kg) is given. Hydrate works in a dose-dependent manner to enhance cellular resorption and development retardation while also causing neural tube abnormalities (NTDs) [1]. Glycinamide ribose plus formyl transferase (GARFT) activity is decreased by lomitrexol (ip; 40 mg/kg; on day 7.5 of gestation) hydrate, which also modifies the levels of ATP, GTP, dATP, and dGTP [1]. injection; 7.5 gestation days; 40 mg/kg aberrant edema and ischemia brought on by hydration in neural tube abnormalities (NTDs) [1]. |
| Cell Assay |
Cell Viability Assay[3] Cell Types: Mouse leukemia L1210 cells Tested Concentrations: 1, 30 μM Incubation Duration: 3]. 2, 4, 6, 8, 10 hrs (hours) Experimental Results: Induces rapid and complete growth inhibition. Cell cycle analysis [3] Cell Types: L1210 Cell Tested Concentrations: 1 μM Incubation Duration: 2, 4, 8, 12, 24 hrs (hours) Experimental Results: Caused rapid loss of G2/M phase cell population and accumulation of cells in early S phase for 8 hrs (hours). By 24 h, the S phase population appears to be slowly shifting to higher DNA content, thus transitioning from mid to late S phase to higher DNA content. |
| Animal Protocol |
Animal/Disease Models: C57BL/6 mice (7-8 weeks, 18-20 g)[1]: 15, 30, 35, 40, 45 and 60 mg/kg Route of Administration: intraperitoneal (ip) injection; gestation day 7.5 Experimental Results: The embryo resorption rate is increased and growth is retarded in a dose-dependent manner. Animal/Disease Models: C57BL/6 mice (7-8 weeks, 18-20g) [1] Doses: 40mg/kg Route of Administration: intraperitoneal (ip) injection; gestation day 7.5 Experimental Results: Inhibition of glycylamide ribonucleotide formyltransferase (GARFT) activity, and GARFT activity was maximally inhibited after 6 hrs (hrs (hours)). The levels of ATP, GTP, dATP and dGTP in NTDs embryonic brain tissue were Dramatically diminished at 6 hrs (hrs (hours)). Animal/Disease Models: C57BL/6 mice (7-8 weeks, 18-20g) [1] Doses: 40mg/kg Route of Administration: intraperitoneal (ip) injection; pregnancy day 7.5, lasting for 4 days Experimental Results: NTD group proliferation-related genes (Pcna , Foxg1 and Ptch1) diminished, and the expression of apoptosis-related genes (Bax, Casp8 and Casp9) increased. |
| References |
[1]. The effect of inhibiting glycinamide ribonucleotide formyl transferase on the development of neural tube in mice. Nutr Metab (Lond). 2016 Aug 23;13(1):56. [2]. Structural basis of inhibition of the human serine hydroxymethyltransferase SHMT2 by antifolate drugs. FEBS Lett. 2019 Jul;593(14):1863-1873. [3]. Antifolates targeting purine synthesis allow entry of tumor cells into S phase regardless of p53 function. Cancer Res. 2002 Sep 15;62(18):5236-41. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~40 mg/mL (~86.68 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1670 mL | 10.8349 mL | 21.6699 mL | |
| 5 mM | 0.4334 mL | 2.1670 mL | 4.3340 mL | |
| 10 mM | 0.2167 mL | 1.0835 mL | 2.1670 mL |