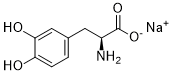
Levodopa sodium (Atamet, Stalevo, Madopar, L-DOPA, Dopar, Sinemet, Pharmacopa, Prolopa), the sodium salt of L-DOPA, is the L-isomer of DOPA and the precursor to the neurotransmitters dopamine, norepinephrine (noradrenaline), and epinephrine (adrenaline) with anti-Parkinson's disease activity. It has been used for the treatment of Parkinson's symptoms. Levodopa is a chemical that is made and used as part of the normal biology of humans, some animals and plants. Some animals and humans make it via biosynthesis from the amino acid L-tyrosine. Furthermore, L-DOPA itself mediates neurotrophic factor release by the brain and CNS.
Physicochemical Properties
| Exact Mass | 220.05857 |
| CAS # | 63302-01-2 |
| Related CAS # | L-DOPA;59-92-7 |
| PubChem CID | 138683040 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 15 |
| Complexity | 209 |
| Defined Atom Stereocenter Count | 1 |
| InChi Key | BDARLFNIXLYGPC-RGMNGODLSA-N |
| InChi Code | InChI=1S/C9H11NO4.Na/c10-6(9(13)14)3-5-1-2-7(11)8(12)4-5;/h1-2,4,6,11-12H,3,10H2,(H,13,14);/t6-;/m0./s1 |
| Synonyms | Levodopa sodium; Levodopa (sodium);3,4-Dihydroxyphenylalanine (sodium); SCHEMBL21055745; AKOS040752531 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Dopamine receptor |
| ln Vitro | Aromatic L-amino acid decarboxylase (EC. 4.1.1.28) deficiency is a newly described inborn error of metabolism that affects serotonin and dopamine biosynthesis. The major biochemical markers for this disease are increases of L-dopa, 3-methoxytyrosine, and 5-hydroxytryptophan in urine, plasma, and cerebrospinal fluid together with decreased cerebrospinal fluid concentrations of homovanillic acid and 5-hydroxyindoleacetic acid. In addition, concentrations of vanillactic acid are increased in the urine. Specific HPLC and gas chromatography-mass spectrometry methods are described that permit the identification and measurement of these metabolites in the above body fluids. Simplified assays for human plasma L-dopa decarboxylase and liver L-dopa and 5-hydroxytryptophan decarboxylase, used to demonstrate the enzyme deficiency, are also reported[1]. |
| ln Vivo | Oral L-DOPA sodium (20 mg/kg) lessens motor impairment brought on by rotenone [3]. In sprague-Dawley rats, oral administration of L-DOPA sodium (0–100 mg/kg) reverses tactile, heat, and cold allodynia without causing any adverse consequences [4]. |
| Animal Protocol |
Animal/Disease Models: C57BL/6J mice (7 weeks old) [3] Doses: 20 mg/kg Route of Administration: Oral Experimental Results: diminished rotenone-induced motor dysfunction. Animal/Disease Models: SD (SD (Sprague-Dawley)) rat [4] Doses: 10, 30, 50, 70 and 100 mg/kg Route of Administration: Orally Experimental Results:Reverse tactile, hot and cold allodynia in neuropathic rats without any side effects. |
| References |
[1]. Aromatic L-amino acid decarboxylase deficiency: diagnostic methodology. Clin Chem. 1992 Dec;38(12):2405-10. [2]. Dopamine dysregulation syndrome, addiction and behavioral changes in Parkinson's disease. Parkinsonism Relat Disord. 2008;14(4):273-80. Epub 2007 Nov 7. [3]. Additive Effects of Levodopa and a Neurorestorative Diet in a Mouse Model of Parkinson's Disease. Front Aging Neurosci. 2018 Aug 3;10:237. [4]. Anti-allodynic effects of levodopa in neuropathic rats. Yonsei Med J. 2013 Mar 1;54(2):330-5. [5]. Pharmacological validation of a mouse model of l-DOPA-induced dyskinesia. Exp Neurol. 2005 Jul;194(1):66-75. [6]. Pharmacokinetics of L-dopa in plasma and extracellular fluid of striatum in common marmosets. Brain Res. 2003 Dec 12;993(1-2):54-8. |
| Additional Infomation |
Degeneration of the dopaminergic system in Parkinson's disease and longstanding exposure to dopaminergic drugs may cause reward system malfunction. This may manifest as addiction to l-dopa and behavioral disturbances associated with the impulse control system. These disturbances include: gambling, excessive spending (shopping), hypersexuality and binge eating. We included one such patient's personal story to emphasize the devastating consequences of these potentially reversible phenomena: the patient describes in his own words how gambling induced by an exposure dopamine agonist therapy significantly worsened his disease-related difficulties.[2] Though Parkinson's disease (PD) clinical picture is generally dominated by motor impairment, non-motor symptoms, such as cognitive decline and gastrointestinal dysfunctions, may develop before motor symptoms and have major effects on quality of life. L-3,4-di-hydroxy-phenylalanine (Levodopa) is the most commonly used treatment of motor symptoms but has serious side-effects with prolonged use and does not stop the degenerative process. Moreover, gastrointestinal dysfunctions interfere with the absorption of levodopa and modify its effectiveness. Since most patients are on levodopa treatment, there is a need for combinational therapies that allow for an effective reduction of both motor and non-motor symptoms. We have recently shown that a diet containing precursors and cofactors required for membrane phospholipid synthesis, as well as prebiotic fibers, had therapeutic effects in a PD mouse model. We now investigate the effects of combined administration of the same diet together with levodopa in the rotenone model of PD. Mice were injected with rotenone or vehicle in the striatum. The dietary intervention started after full induction of motor symptoms. The effects of dietary intervention and oral treatment with different doses of levodopa were assessed weekly. Motor and cognitive functions were tested, intestinal transit was analyzed and histological examination of the brain and the colon was assessed. Our results confirm our previous findings that rotenone-induced motor and non-motor problems were alleviated by the Active diet (AD). Levodopa showed an additive beneficial effect on rotarod performance in rotenone-treated animals fed with the AD. No negative interaction effects were found between the AD and levodopa. Our findings suggest that the dietary intervention might confer additional clinical benefits on patients receiving levodopa treatment.[3] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |