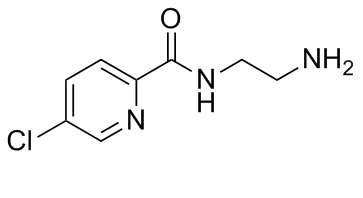
Lazabemide (also known as Ro 19-6327; proposed trade names Pakio, Tempium) is novel, potent, selective, reversible monoamine oxidase B (MAO-B) inhibitor with IC50 values are 0.03 and > 100 μM for MAO-B and MAO-A respectively. Lazabemide was under development as an antiparkinsonian agent but was never marketed.
Physicochemical Properties
| Molecular Formula | C8H10N3OCL |
| Molecular Weight | 199.6375 |
| Exact Mass | 199.051 |
| CAS # | 103878-84-8 |
| Related CAS # | Lazabemide hydrochloride;103878-83-7 |
| PubChem CID | 71307 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 397.4±37.0 °C at 760 mmHg |
| Flash Point | 194.2±26.5 °C |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.572 |
| LogP | -0.01 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 13 |
| Complexity | 177 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | JZXRLKWWVNUZRB-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C8H10ClN3O/c9-6-1-2-7(12-5-6)8(13)11-4-3-10/h1-2,5H,3-4,10H2,(H,11,13) |
| Chemical Name | N-(2-aminoethyl)-5-chloropyridine-2-carboxamide |
| Synonyms | Ro 19-6327; Ro-19-6327; Ro 19 6327 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Both radiolabeled inhibitors have high affinity for their respective enzymes and are selective ligands based on their in vitro binding characteristics. In rat cerebral cortex, 3H-Ro 19-6327 had KD and Bmax values of 18.4 nM and 3.45 pmol/mg protein, respectively [1]. Lazabemide's IC50 values are as follows: 86 μM for NA uptake, 123 μM for 5HT uptake, and >500 μM for DA uptake, respectively[1]. Human MAO-B and MAO-A are inhibited by lazabemide (5 μM) with IC50 values of 6.9 nM and >10 nM, respectively. It inhibits rat MAO-B and MAO-A in enzymatic experiments with IC50 values of 37 nM and >10 μM, respectively [2]. In terms of their capacity to trigger the release of endogenous monoamines from synaptosomes, lazabemide and L-deprenyl are not the same. Thus, compared to L-deprenyl, lazabemide (500 μM) caused a greater release of 5 HT, but it was less successful in releasing DA. On the other hand, lazabemide is essentially inert upon 5-HT and DA release [2]. In renal tubular epithelial cells treated with 50 microM L-DOPA, lazabemide (250 nM) effectively suppresses DOPAC production without increasing the concentration of freshly generated DA [3]. |
| ln Vitro |
Both radiolabeled inhibitors have high affinity for their respective enzymes and are selective ligands based on their in vitro binding characteristics. In rat cerebral cortex, 3H-Ro 19-6327 had KD and Bmax values of 18.4 nM and 3.45 pmol/mg protein, respectively [1]. Lazabemide's IC50 values are as follows: 86 μM for NA uptake, 123 μM for 5HT uptake, and >500 μM for DA uptake, respectively[1]. Human MAO-B and MAO-A are inhibited by lazabemide (5 μM) with IC50 values of 6.9 nM and >10 nM, respectively. It inhibits rat MAO-B and MAO-A in enzymatic experiments with IC50 values of 37 nM and >10 μM, respectively [2]. In terms of their capacity to trigger the release of endogenous monoamines from synaptosomes, lazabemide and L-deprenyl are not the same. Thus, compared to L-deprenyl, lazabemide (500 μM) caused a greater release of 5 HT, but it was less successful in releasing DA. On the other hand, lazabemide is essentially inert upon 5-HT and DA release [2]. In renal tubular epithelial cells treated with 50 microM L-DOPA, lazabemide (250 nM) effectively suppresses DOPAC production without increasing the concentration of freshly generated DA [3]. Lazabemide (³H-Ro 19-6327) demonstrated high-affinity, saturable, and pharmacologically specific binding to MAO-B in tissue sections from rat CNS, peripheral organs, and human postmortem brain. The equilibrium dissociation constant (K_D) for ³H-Ro 19-6327 binding to rat brain frontal cortex was 18.4 nM, with a maximum binding capacity (B_max) of 3.45 pmol/mg protein. The binding was competitively inhibited by known MAO-B inhibitors (e.g., L-deprenyl) but not by MAO-A inhibitors, confirming its selectivity for MAO-B. [1] Quantitative enzyme radioautography using ³H-Ro 19-6327 revealed the detailed distribution and abundance of MAO-B in microscopic regions. In rat brain, MAO-B was most abundant in circumventricular organs, ependyma, olfactory nerve layer (inner part), paraventricular thalamus, mammillary nuclei, raphe nuclei, and cerebellar Bergmann glia cells. In peripheral rat tissues, high abundance was found in the endocrine pancreas, liver (periportal > central regions), epididymis, and duodenum. [1] In human postmortem brain sections, MAO-B binding sites (visualized with ³H-Ro 19-6327) were generally more abundant than MAO-A sites. High levels were found in ependymal cells, the polymorphic layer of the dentate gyrus, dorsal raphe nucleus, substantia nigra (pars reticulata), and interpeduncular nucleus. [1] |
| ln Vivo | Lazabemide pretreatment demonstrated lower DOPAC levels compared with comparable vehicle-treated controls, and Lazabemide (3 mg/kg) inhibited ischemia-reperfusion-induced hydroxyl radical generation [4]. |
| Enzyme Assay |
The in vitro binding characteristics of tritiated Lazabemide (³H-Ro 19-6327) to MAO-B were determined using quantitative enzyme radioautography on tissue sections. [1] Cryostat sections (12 μm) of fresh-frozen rat and human postmortem tissues were mounted on slides. Sections were incubated with ³H-Ro 19-6327 in a buffer (pH 7.4) containing 50 mM Tris, 120 mM NaCl, 1 mM MgCl₂, 5 mM KCl, and 0.5 mM EGTA. Incubation was performed at 20°C for 90 minutes to reach steady state. [1] Non-specific binding was determined by co-incubating adjacent sections with 1 μM L-deprenyl (for ³H-Ro 19-6327) or 50 μM L-deprenyl (for saturation experiments). After incubation, sections were rinsed (30 sec + 30 sec + 60 sec in ice-cold buffer followed by a quick rinse in ice-cold distilled water) to remove unbound ligand. [1] Binding kinetics (association/dissociation) and saturation experiments were performed. For saturation, sections were incubated with increasing concentrations of ³H-Ro 19-6327 (1-250 nM). Radioactivity was quantified either by wiping tissues onto glass fiber filters for liquid scintillation counting or by apposing the labeled sections to tritium-sensitive film for 3 weeks followed by densitometric analysis using a computer-assisted image analysis system. Optical densities were converted to pmol/mg protein using calibrated standards. [1] Competition binding experiments were performed by incubating sections with 20 nM ³H-Ro 19-6327 in the presence of various concentrations (1 pM to 1 mM) of competing MAO inhibitors (e.g., L-deprenyl, Ro 41-1049) to determine IC₅₀ values. [1] |
| Cell Assay |
Uptake assay in rat forebrain synaptosomes: Crude synaptosome preparations were obtained from rat forebrain homogenates. Synaptosomes were preincubated for 5 minutes at 37°C in Krebs buffer containing the MAO inhibitor pargyline (10 µM) in the presence or absence of test drugs (Lazabemide, Ro 16-6491, or L-deprenyl). Subsequently, a tritiated amine ([³H]NA, [³H]DA, or [³H]5-HT) at a concentration of 3 nM was added, and incubation continued for an additional 4 minutes. The reaction was stopped by centrifugation at 4°C. The pellets were washed twice and solubilized, and radioactivity was measured by liquid scintillation spectrometry. Uptake in samples incubated at 4°C served as the blank. [2] Endogenous amine release from synaptosomes: To study drug-induced release, synaptosomes were preincubated for 5 minutes at 37°C in Krebs buffer containing pargyline (10 µM) and the general uptake inhibitor diclofensine (1 µM) to minimize re-uptake. Samples were then incubated for an additional 15 minutes in the presence or absence of test drugs. After centrifugation, the pellet was homogenized in perchloric acid containing an internal standard. The supernatant was analyzed for endogenous NA, DA, and 5-HT content using reversed-phase HPLC with coulometric detection. [2] [¹⁴C]5-HT uptake in rat platelets: Platelet-rich plasma (PRP) was preincubated for 5 minutes at 37°C with or without test drugs. [¹⁴C]5-HT (1 µM) was then added, and incubation continued for 5 minutes. Platelets were sedimented by centrifugation, washed, and lysed. Radioactivity and protein content in the lysate were determined. [2] [¹⁴C]5-HT liberation from rat platelets: PRP was preincubated with [¹⁴C]5-HT (1 µM) for 10 minutes at 37°C. Platelets were then washed and resuspended in buffer. Aliquots of this suspension were reincubated for 60 minutes at 37°C with or without test drugs. After sedimentation and washing, platelet pellets were lysed, and remaining radioactivity was measured. [2] |
| References |
[1]. Quantitative enzyme radioautography with 3H-Ro 41-1049 and 3H-Ro 19-6327 in vitro: localization and abundance of MAO-A and MAO-B in rat CNS, peripheral organs, and human brain. J Neurosci. 1992 May;12(5):1977-99. [2]. In vitro effects on monoamine uptake and release by the reversible monoamine oxidase-B inhibitors lazabemide and N-(2-aminoethyl)-p-chlorobenzamide: a comparison with L-deprenyl. Biochem Pharmacol. 1995 Jun 29;50(1):97-102. [3]. The activity of MAO A and B in rat renal cells and tubules. Life Sci. 1998;62(8):727-37. [4]. MAO inhibitors, clorgyline and lazabemide, prevent hydroxyl radical generation caused by brain ischemia/reperfusion in mice. Pharmacology. 1995 Jun;50(6):357-62. |
| Additional Infomation |
Lazabemide is described as a novel, highly selective, reversible inhibitor of MAO-B. At the time of the study, it was noted to be in phase II clinical trials for Parkinsonism. [1] The study utilized Lazabemide primarily as a research tool (tritiated radioligand) for mapping the distribution and abundance of MAO-B via quantitative enzyme radioautography, providing a sensitive and quantitative alternative to histochemical and immunohistochemical methods. [1] The distribution of MAO-B, as revealed by Lazabemide binding, suggests its physiological roles may include acting as a scavenger for trace amines (e.g., phenethylamine) in astrocytes and ependymal cells, and potentially preventing the accumulation of false neurotransmitters in serotoninergic neurons. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~5 mg/mL (~25.05 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 0.5 mg/mL (2.50 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.5 mg/mL (2.50 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 0.5 mg/mL (2.50 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.0090 mL | 25.0451 mL | 50.0902 mL | |
| 5 mM | 1.0018 mL | 5.0090 mL | 10.0180 mL | |
| 10 mM | 0.5009 mL | 2.5045 mL | 5.0090 mL |