LY-334370 HCl, the hydrochloride of LY-334370, is a novel, potent and selective 5-HT1F receptor agonist with Ki values of 1.6 nM. It displays antimigraine effects.
Physicochemical Properties
| Molecular Formula | C21H23CLFN3O |
| Molecular Weight | 387.878227472305 |
| Exact Mass | 387.151 |
| Elemental Analysis | C, 65.03; H, 5.98; Cl, 9.14; F, 4.90; N, 10.83; O, 4.12 |
| CAS # | 199673-74-0 |
| Related CAS # | 199673-74-0(LY 334370 hydrochloride);182563-08-2 (LY 334370 Free Base); |
| PubChem CID | 19710026 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 5.181 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 27 |
| Complexity | 486 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | C(NC1C=CC2=C(C=1)C(C1CCN(C)CC1)=CN2)(=O)C1=CC=C(F)C=C1.[H]Cl |
| InChi Key | DGPDGAPZTPSHBL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H22FN3O.ClH/c1-25-10-8-14(9-11-25)19-13-23-20-7-6-17(12-18(19)20)24-21(26)15-2-4-16(22)5-3-15;/h2-7,12-14,23H,8-11H2,1H3,(H,24,26);1H |
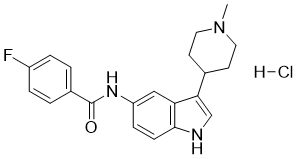
| Chemical Name | 4-Fluoro-N-[3-(1-methyl-4-piperidinyl)-1H-indol-5-yl]benzamide hydrochloride |
| Synonyms | LY 334370; LY-334370; LY334370; LY 334370 hydrochloride; 199673-74-0; LY334370 (hydrochloride); 4-Fluoro-N-[3-(1-methyl-4-piperidinyl)-1H-indol-5-yl]benzamide hydrochloride; 4-fluoro-N-[3-(1-methyl-4-piperidinyl)-1H-indol-5-yl]benzamidehydrochloride; BenzaMide, 4-fluoro-N-[3-(1-Methyl-4-piperidinyl)-1H-indol-5-yl]-, Monohydrochloride; 4-fluoro-N-[3-(1-methylpiperidin-4-yl)-1H-indol-5-yl]benzamide;hydrochloride; SCHEMBL8604274; LY 334370 hydrochloride; LY 334370 HCl. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
5-HT1F Receptor ( Ki = 1.6 nM ) Serotonin 5-HT1F receptor agonist (Ki = 1.6 nM) [1] |
| ln Vitro |
LY334370 does not exhibit vasoconstrictor effects on human cerebral arteries in vitro, unless it is administered at a dose of 10-5 M, at which point it causes an 8.5±5.7% contraction, which is not statistically significant[1]. In isolated human cerebral arteries from the temporal-parietal region, LY334370 (up to 10^-5 M) did not produce a significant vasoconstrictor effect. At 10^-5 M, it caused only 8.5 ± 5.7% contraction relative to the maximum KCl-induced contraction, which was not statistically significant. In contrast, sumatriptan potently constricted the same vessels with an Emax of 50 ± 10% and a pD2 of 6.8 ± 0.2. [1] |
| ln Vivo |
Intravenous LY334370 administration at 3 mg/kg (n = 3) or 10 mg/kg (n = 6) causes an increase in dural blood vessel diameter of 135±6% and 106±11%, respectively, after electrical stimulation; these values do not differ significantly from the corresponding control values. Since dural blood vessel diameter is actually 43±4 arbitrary units before drug injection and 43±4 arbitrary units 15 minutes after injection of LY334370 (10 mg/kg), LY334370 has no effect on dural blood vessel diameter per se[1]. Neurogenic Dural Vasodilation in Rats: Intravenous administration of LY334370 at 3 mg/kg or 10 mg/kg had no significant effect on the neurogenic vasodilation of dural blood vessels evoked by electrical stimulation of the dura mater in anesthetized rats. The compound also had no effect on baseline dural vessel diameter. [1] Trigeminal Neuronal Responses in Rats: Intravenous LY334370 (0.3, 1, and 3 mg/kg) produced a significant and dose-dependent inhibition of the evoked responses of second-order neurons in the trigeminal nucleus caudalis to electrical stimulation of the dura mater in anesthetized rats. The inhibition reached a maximum of -64 ± 14% from control at 19 ± 3 minutes after the 3 mg/kg dose. [1] Carrageenan-induced Hyperalgesia in Rats: Intravenous administration of LY334370 at 3 mg/kg had no effect on the mechanical hyperalgesia induced by carrageenan injection into the rat paw. In the same assay, morphine (1-3 mg/kg, sc) completely reversed the hyperalgesia in a dose-dependent manner. [1] Nociceptive Reflexes in Rabbits: Intravenous LY334370 (0.3 - 3 mg/kg) had no significant effect on baseline, facilitated, or wind-up nociceptive reflex responses in spinalized, decerebrate rabbits. [1] |
| Cell Assay | n this study, human cerebral arteries are used. Segments are prepared as previously described, but briefly they are placed in a buffer solution containing (mM) NaCl 119, NaHCo3 15, KCl 4.6, CaCl2 1.5, NaH2PO4 1.2, MgCl2 1.2, and glucose 5.5. Sections of the vessel, measuring 0.5 mm in diameter and 1 to 2 mm in length, are placed in a tissue bath that is heated to 37°C and contains a buffer solution that has been agitated with 5% CO2 and 95% O2. Four mN of tension is applied to the vessel segments, and they are left to stabilize there for one to one and a half hours. Exposure to 60 mM KCl is used to test the reactivity of vessels. Only if the response is similar to the segment used for LY334370 testing is this repeated twice for each segment. As a percentage of the maximum KC1 response, responses to LY334370 are computed[1]. |
| Animal Protocol |
There are 300–400 g male Sprague-Dawley rats used. Intravital microscopy and a video dimension analyzer are used to measure the diameter of a branch of the middle meningeal artery in the intact skull of rats housed in a stereotaxic frame after their skulls have been thinned and exposed through drilling. A bipolar stimulating electrode, positioned on the cranial window surface about 200 μM away from the vessel of interest, is used to induce neurogenic vasodilation. After LY334370 (3 or 10 mg/kg, iv.) is administered five minutes after an electrical stimulation response that produces a control vasodilation is produced, and after another fifteen minutes, the electrical stimulation is repeated. For each response, the mean maximum percentage increase in dural vessel diameter relative to the pre-stimulus baseline is calculated. Analysis of variance is then used to compare the vasodilation responses evoked in the presence or absence of LY334370, and paired t-tests are employed to make these comparisons[1]. Human Cerebral Artery Study: Human cerebral artery segments (0.5 mm diameter, 1-2 mm length) from the temporal-parietal region were mounted in a temperature-controlled tissue bath (37°C) with oxygenated buffer. They were placed under 4 mN tension and stabilized. Reactivity was tested with 60 mM KCl. Cumulative concentration-response curves to LY334370 or sumatriptan were constructed, with responses calculated as a percentage of the maximum KCl contraction. [1] Neurogenic Dural Vasodilation Study in Rats: Male Sprague-Dawley rats (300-400 g) were anesthetized with pentobarbitone sodium. A branch of the middle meningeal artery was visualized through a thinned skull, and its diameter was measured using intravital microscopy. Neurogenic vasodilation was evoked by electrical stimulation (5 Hz, 1 ms, 50-300 μA for 10 sec) via an electrode placed on the cranial window. After a control response, LY334370 (3 or 10 mg/kg) or vehicle was administered intravenously, and the stimulation was repeated 15 minutes later. [1] Trigeminal Neuronal Response Study in Rats: Male Sprague-Dawley rats (320-420 g) were anesthetized with halothane and maintained on sodium pentobarbitone infusion. Single-unit extracellular recordings were made from neurons in the caudal trigeminal nucleus. Neuronal responses were evoked by electrical stimulation (100-200 μs, 1-3 mA, 1 Hz for 20 sec) of the middle meningeal artery, repeated every 200 sec. LY334370 was administered intravenously in cumulative doses (0.3, 1, 3 mg/kg) at 10-minute intervals. [1] Rat Carrageenan-induced Hyperalgesia Study: Male Sprague-Dawley rats (100-120 g) had their baseline paw pressure vocalization threshold determined. Carrageenan (4.5 mg in 0.15 ml) was injected into one hind paw. Two hours later, rats received either morphine (0.1-3.0 mg/kg, sc), morphine vehicle (saline), LY334370 (3 mg/kg, iv), or LY334370 vehicle (distilled water). Vocalization thresholds were re-determined 1 hour after drug/vehicle administration. [1] Rabbit Nociceptive Reflex Study: New Zealand White rabbits (2.5-3.5 kg) were anesthetized, spinalized at C2, and decerebrated. Motor unit activity was recorded from the hindlimb. Baseline responses to single electrical shocks (1 ms, 2x threshold voltage) and facilitated/wind-up responses to a 20-shock conditioning stimulus (1 Hz) were recorded. LY334370 (0.3-3 mg/kg, iv) or vehicle was administered, and its effects on these reflex responses were evaluated. [1] |
| ADME/Pharmacokinetics |
Phase I clinical studies indicated that LY334370 had good oral bioavailability, with a time to maximum concentration (tmax) between 1 and 2 hours and an elimination half-life of approximately 15 hours. [1] |
| Toxicity/Toxicokinetics |
Phase I clinical studies showed that LY334370 was well-tolerated up to an oral dose of 400 mg. Reported side effects included weakness, somnolence, and dizziness. No cardiovascular effects (assessed by blood pressure, pulse, or ECG) were observed. [1] |
| References | [1]. Cephalalgia. 1999 Dec;19(10):851-8. |
| Additional Infomation |
This study investigated whether the selective 5HT1F receptor agonist LY334370 has other possible antimigraine mechanisms in addition to the proposed inhibition of dural plasma extravasation. LY334370 (up to 10(-5) M) had no vasoconstrictor effects on human cerebral arteries in vitro. It had no effect (up to 10 mg kg-1, i.v.) on neurogenic vasodilation of dural blood vessels produced by electrical stimulation of the dura mater in anesthetized rats. Nor had it any effect (at 3 mg kg-1, i.v.) on the hyperalgesia produced by injection of carrageenan into the paw of conscious rats or on nociceptive reflex responses in the spinalized, decerebrate rabbit (up to 3 mg kg-1, i.v.), indicating that it has no general analgesic properties. However, it significantly inhibited activation of second-order neurons in the trigeminal nucleus caudalis produced by electrical stimulation of the dura mater in anesthetised rats at 3 mg kg-1, i.v. These results provide evidence to suggest that LY334370 has a central mechanism of action in blocking the transmission of nociceptive impulses within the trigeminal nucleus caudalis and that this may represent a mechanism through which it has its antimigraine effect.[1] LY334370 (4-fluoro-N-[3-(1-methyl-4-piperidinyl)-1H-indol-5-yl]-benzamide) is a potent and selective 5-HT1F receptor agonist developed as an antimigraine agent. It has approximately 100-fold selectivity over 5-HT1B and 5-HT1D receptors. [1] The proposed antimigraine mechanism of LY334370 involves a central action to block the transmission of nociceptive impulses within the trigeminal nucleus caudalis, rather than through cranial vasoconstriction or inhibition of neurogenic dural vasodilation. It does not appear to possess general analgesic properties. [1] Preliminary Phase II clinical data showed that oral doses of 80 and 200 mg of LY334370 provided significant pain relief at 2 hours in patients with moderate to severe migraine headache compared to placebo. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5781 mL | 12.8906 mL | 25.7812 mL | |
| 5 mM | 0.5156 mL | 2.5781 mL | 5.1562 mL | |
| 10 mM | 0.2578 mL | 1.2891 mL | 2.5781 mL |