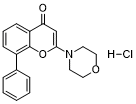
LY-294002 hydrochloride is a morpholine-containing compound based on the flavonoid quercetin, acting as a potent and cell-permeable PI3K inhibitor, inhibiting PI3Kα/δ/β with IC50 of 0.5 μM/0.57 μM/0.97 μM in cell-free assays, respectively. It is also an inhibitor of BET (e.g. of BRD2, BRD3, and BRD4). In solution, it is more stable than Wortmannin (PI3K inhibitor). LY294002 is selective against p110α, p110β, p110γ and p110δ, by acting on the ATP binding site of the catalytic subunit of PI3K.
Physicochemical Properties
| Molecular Formula | C19H17NO3.HCL |
| Molecular Weight | 343.80412 |
| Exact Mass | 343.098 |
| Elemental Analysis | C, 66.38; H, 5.28; Cl, 10.31; N, 4.07; O, 13.96 |
| CAS # | 934389-88-5 |
| Related CAS # | LY294002;154447-36-6 |
| PubChem CID | 11957589 |
| Appearance | Solid powder |
| LogP | 4.163 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 24 |
| Complexity | 463 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | Cl.O=C1C2C=CC=C(C=2OC(N2CCOCC2)=C1)C1C=CC=CC=1 |
| InChi Key | OQZQSRICUOWBLW-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C19H17NO3.ClH/c21-17-13-18(20-9-11-22-12-10-20)23-19-15(7-4-8-16(17)19)14-5-2-1-3-6-14;/h1-8,13H,9-12H2;1H |
| Chemical Name | 2-(4-Morpholinyl)-8-phenyl-4H-1-benzopyran-4-one hydrochloride |
| Synonyms | LY-294002 HCl; LY294002 HCl;LY294002 hydrochloride; LY 294002 hydrochloride; LY 294002 HCl |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | p110α (IC50 = 0.5 μM); p110δ (IC50 = 0.57 μM); p110β (IC50 = 0.97 μM); human CK2 (IC50 = 98 nM); human CK2α2 (IC50 = 3.869 μM); DNA-PK (IC50 = 1.4 μM) |
| ln Vitro | In a dose-dependent manner, LY294002 hydrochloride (0-75 μM; 24 and 48 hours) significantly reduces human nasopharyngeal cancer CNE-2Z cells[4]. LY294002 hydrochloride (0-75 μM; 24 and 48 hours) dose-dependently increases the rate of apoptosis in CNE-2Z cells[4]. In CNE-2Z cells, LY294002 hydrochloride (10–75 μM) dramatically reduces p-Akt (S473) expression levels and increases caspase-9 activity. There is no variation in the total Akt protein level across varied concentrations [4]. Treatment with LY294002 hydrochloride (5, 10, 100 µM; for 2 hours) partially inhibits the nuclear translocation of YAP produced by Lysophosphatidic acid (LPA) (20 µM; for 4 hours), which is followed by a decrease in p-AKT levels[6]. |
| ln Vivo | In a dose-dependent manner, LY294002 hydrochloride (10, 25, 50, 75 mg/kg; ip; twice weekly; for 4 weeks) considerably lowers the mean NPC tumor burden. The effectiveness of LY294002 (10, 25 mg/kg) in reducing tumor burden is lower[4]. In Sprague-Dawley rats, LY294002 hydrochloride (1.2 mg/kg ip; i.p.) for 14 days inhibits the deleterious effects of leptin (60 ug/kg) on spermatozoa[5]. |
| Enzyme Assay | PI3K inhibition by LY294002 is determined in a radiometric assay using purified, recombinant enzymes with 1μM ATP. At room temperature (24oC), the kinase reaction lasts for an hour before being stopped by the addition of PBS. Then, IC50 values are calculated by fitting a variable slope sigmoidal dose-response curve. Kinase selectivity screening is used to determine the inhibition of CK2 and GSK3β (glycogen synthase kinase 3β). In 10μM ATP, LY294002 is evaluated against the Upstate panel of kinases. |
| Cell Assay |
Cell Proliferation Assay Cell Types: CNE-2Z cells[4] Tested Concentrations: 0 μM, 10 μM, 25 μM, 50 μM, and 75 μM Incubation Duration: 24 hrs (hours) and 48 hrs (hours) Experimental Results: diminished CNE-2Z cells in a dose-dependent fashion. Apoptosis Analysis Cell Types: CNE-2Z cells[4] Tested Concentrations: 0 μM, 10 μM, 25 μM, 50 μM, and 75 μM Incubation Duration: 24 hrs (hours) and 48 hrs (hours) Experimental Results: Induced apoptosis rate in a dose-dependent manner. Western Blot Analysis Cell Types: CNE-2Z cells[4] Tested Concentrations: 0 μM, 10 μM, 25 μM, 50 μM, and 75 μM Incubation Duration: 24 hrs (hours) and 48 hrs (hours) Experimental Results: diminished phosphorylated Akt (S473) expression levels were Dramatically, up-regulated caspase-9 activity in CNE-2Z cells in treated group. |
| Animal Protocol |
Animal/Disease Models: Athymic nude mice (6-8 weeks) with CNE-2Z xenograft[4] Doses: 10 mg/kg, 25 mg/kg, 50 mg/kg, and 75 mg/kg Route of Administration: IP; twice weekly, for 4 weeks Experimental Results: Mean Nasopharyngeal carcinoma (NPC) tumor burden was remarkably decreased in a dose-dependent manner. |
| References |
[1]. The effect of PI3K inhibitor LY294002 and gemcitabine hydrochloride combined with ionizing radiation on the formation of vasculogenic mimicry of Panc-1 cells in vitro and in vivo. Neoplasma. 2016;63(1):80-92. [2]. Evidence for functional redundancy of class IA PI3K isoforms in insulin signalling. Biochem J. 2007 Jun 15;404(3):449-58. [3]. Exploring the specificity of the PI3K family inhibitor LY294002. Biochem J. 2007 May 15;404(1):15-21. [4]. Phosphatidylinositol 3-kinase inhibitor(LY294002) induces apoptosis of human nasopharyngeal carcinoma invitro and in vivo. J Exp Clin Cancer Res. 2010 Apr 22;29:34. [5]. LY294002, a PI3K pathway inhibitor, prevents leptin-induced adverse effects on spermatozoa in Sprague-Dawley rats. Andrologia. 2019 Apr;51(3):e13196. [6]. Lysophosphatidic acid induces YAP-promoted proliferation of human corneal endothelial cells via PI3K and ROCK pathways. Mol Ther Methods Clin Dev. 2015 Apr 29;2:15014. |
| Additional Infomation |
This research's purpose was to explore the existence of vasculogenic mimicry (VM) in both 3-D matrices of Panc-1 cells in vitro and orthotopic Panc-1 xenografts in vivo and to test the hypothesis that PI3K inhibitor LY294002 and gemcitabine hydrochloride would offer clear treatment benefit when integrated into ionizing radiation (IR) therapeutic regimens for treatment of pancreatic cancer. We explored the existence of VM in both 3-D matrices of Panc-1 cells and orthotopic Panc-1 xenografts. We subsequently investigated the activation of the PI3K/MMPs/Ln-5γ2 signaling pathway in response to IR. LY294002 and gemcitabine hydrochloride were then evaluated for their radiosensitizing effect solely and in combination. We found that VM existed in both 3-D matrices of Panc-1 cells in vitro and orthotopic Panc-1 xenografts in vivo. The expressions of p-Akt and MMP- 2 were found to increase in response to IR. LY294002 and gemcitabine hydrochloride combined with IR better inhibited cell migration, VM formation and MMP-2 mRNA expression of Panc-1 cells in vitro, and we also proved that the novel therapeutic regimen better inhibited tumor growth, tumor metastasis and VM formation of orthotopic Panc-1 xenografts by suppressing the PI3K/MMPs/Ln-5γ2 signaling pathway in vivo. Our present study is among the first to prove the VM formation in orthotopic Panc-1 xenografts. Furthermore, our current study is also among the first to provide preliminary evidence for the use of the novel therapeutic regimen LY294002 and gemcitabine hydrochloride combined with IR for treatment of pancreatic cancer.[1] The PI3Ks (phosphatidylinositol 3-kinases) regulate cellular signalling networks that are involved in processes linked to the survival, growth, proliferation, metabolism and specialized differentiated functions of cells. The subversion of this network is common in cancer and has also been linked to disorders of inflammation. The elucidation of the physiological function of PI3K has come from pharmacological studies, which use the enzyme inhibitors Wortmannin and LY294002, and from PI3K genetic knockout models of the effects of loss of PI3K function. Several reports have shown that LY294002 is not exclusively selective for the PI3Ks, and could in fact act on other lipid kinases and additional apparently unrelated proteins. Since this inhibitor still remains a drug of choice in numerous PI3K studies (over 500 in the last year), it is important to establish the precise specificity of this compound. We report here the use of a chemical proteomic strategy in which an analogue of LY294002, PI828, was immobilized onto epoxy-activated Sepharose beads. This affinity material was then used as a bait to fish-out potential protein targets from cellular extracts. Proteins with high affinity for immobilized PI828 were separated by one-dimensional gel electrophoresis and identified by liquid chromatography-tandem MS. The present study reveals that LY294002 not only binds to class I PI3Ks and other PI3K-related kinases, but also to novel targets seemingly unrelated to the PI3K family.[3] Background: To evaluate whether PI3K/Akt pathway could effect on apoptosis and its mechanism in nasopharyngeal carcinoma cells. Methods: The activation of the PI3K/Akt and its effect on CNE-2Z cells in vivo and in vitro was investigated by MTT assay, flow cytometry, western blot, ELISA, terminal deoxyribonucleotide transferase-mediated nick-end labeling assays (TUNEL), and immunohistochemical analyses, using PI3K inhibitor, LY294002. Results: The results showed that LY294002 inhibited the phosphorylating of Akt (S473), cell proliferation, and induced apoptosis in CNE-2Z cells. However, our experiment results also demonstrated that apoptosis-induced LY294002 was directly regulated by caspase-9 activation pathway. Conclusion: These data suggested that PI3K inhibitor, LY294002, induced apoptosis by caspase-9 activation pathway and might be as a potentially useful target for therapeutic intervention in nasopharyngeal carcinoma patients.[4] |
Solubility Data
| Solubility (In Vitro) | Typically soluble in DMSO (e.g. > 10 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9087 mL | 14.5433 mL | 29.0867 mL | |
| 5 mM | 0.5817 mL | 2.9087 mL | 5.8173 mL | |
| 10 mM | 0.2909 mL | 1.4543 mL | 2.9087 mL |