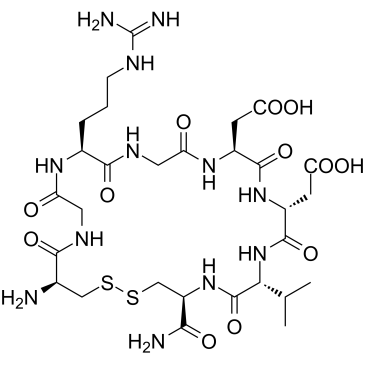
LXW7 is a novel and potent octamer disulfide cyclic peptide acting as an αvβ3 integrin inhibitor. Also a lignd for endothelial progenitor cells (EPCs) and endothelial cells (ECs).
Physicochemical Properties
| Molecular Formula | C29H48N12O12S2 |
| Molecular Weight | 820.894622802734 |
| Exact Mass | 820.295 |
| CAS # | 1313004-77-1 |
| Related CAS # | LXW7 TFA |
| PubChem CID | 89667563 |
| Appearance | Typically exists as solid at room temperature |
| LogP | -8.5 |
| Hydrogen Bond Donor Count | 13 |
| Hydrogen Bond Acceptor Count | 16 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 55 |
| Complexity | 1480 |
| Defined Atom Stereocenter Count | 6 |
| SMILES | CC(C)[C@@H]1C(=O)N[C@H](CSSC[C@H](C(=O)NCC(=O)N[C@H](C(=O)NCC(=O)N[C@H](C(=O)N[C@@H](C(=O)N1)CC(=O)O)CC(=O)O)CCCN=C(N)N)N)C(=O)N |
| InChi Key | UUJIUMRQOUEUEJ-NBJCVSNRSA-N |
| InChi Code | InChI=1S/C29H48N12O12S2/c1-12(2)22-28(53)40-17(23(31)48)11-55-54-10-13(30)24(49)35-8-18(42)37-14(4-3-5-34-29(32)33)25(50)36-9-19(43)38-15(6-20(44)45)26(51)39-16(7-21(46)47)27(52)41-22/h12-17,22H,3-11,30H2,1-2H3,(H2,31,48)(H,35,49)(H,36,50)(H,37,42)(H,38,43)(H,39,51)(H,40,53)(H,41,52)(H,44,45)(H,46,47)(H4,32,33,34)/t13-,14+,15+,16-,17-,22-/m1/s1 |
| Chemical Name | 2-[(4S,7R,10R,13S,19S,25S)-25-amino-4-carbamoyl-10-(carboxymethyl)-19-[3-(diaminomethylideneamino)propyl]-6,9,12,15,18,21,24-heptaoxo-7-propan-2-yl-1,2-dithia-5,8,11,14,17,20,23-heptazacyclohexacos-13-yl]acetic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
LXW7 (cGRGDdvc) is a cyclic RGD peptide that targets and binds to the αvβ3 integrin with high affinity. IC₅₀ for inhibiting Echistatin-FITC binding to αvβ3-K562 cells: 0.68 ± 0.08 μM. Also shows weak binding to αvβ5 and αIIbβ3 integrins, but no binding to α5β1 (K562 parent cells). [1] |
| ln Vitro |
LXW7 selectively binds αvβ3 integrin (Kd=76±10 nM). LXW7 significantly attaches to αvβ3-K562 cells, weakly binds to αvβ5-K562 cells and αIIbβ3-K562 cells, and does not bind to K562 cells. LXW7 has considerable potential as a highly efficient peptide ligand for targeted imaging and medication delivery [1]. LXW7 functions as a potent and specific endothelial progenitor cell (EPC) and endothelial cell (EC) targeting ligand [2]. LXW7 demonstrated the highest binding affinity among tested peptides (LXW1-8) for αvβ3 integrin, with an IC₅₀ of 0.68 ± 0.08 μM in a competitive binding assay using Echistatin-FITC. Biotinylated LXW7 (LXW7-Bio) showed near-identical binding affinity (IC₅₀ = 0.62 ± 0.07 μM) to αvβ3 integrin, unlike other biotinylated RGD cyclopentapeptides which showed 2- to 8-fold decreased affinity. LXW7 bound strongly to tumor cell lines expressing αvβ3 integrin, including U-87MG glioblastoma and A375M melanoma cells. The binding of biotinylated LXW7 to U-87MG and A375M cells was significantly blocked by an anti-αvβ3 antibody (LM609), confirming specificity. The binding affinities (Kd) of LXW7 for U-87MG, A375M, and αvβ3-K562 cells were similar, in the range of 72-89 nM. [1] |
| ln Vivo |
Rats treated with LXW7 (100 μg/kg; intravenous) had a significant decrease in brain water content (BWC) and infarct volume. Pro-inflammatory cytokine expression is decreased by LXW7 therapy [3]. In nude mice bearing U-87MG glioblastoma or A375M melanoma xenografts, intravenous injection of a tetravalent imaging complex (biotinylated LXW7/streptavidin-Cy5.5) resulted in specific accumulation of the fluorescent signal in the tumors at 6 hours post-injection, compared to a streptavidin-Cy5.5-only control. Ex vivo imaging confirmed high tumor uptake of the LXW7-based complex. Compared to biotinylated forms of cyclic RGD pentapeptides (cyclo(RGDfE), cyclo(RGDfK), cyclo(RGDyK)), biotinylated LXW7 showed higher tumor uptake and significantly lower liver uptake in the U-87MG xenograft model. Confocal microscopy of U-87MG xenograft cryosections showed that LXW7 also distributed to CD31-positive neovasculature. [1] |
| Cell Assay |
Flow Cytometry Competitive Binding Assay: To determine IC₅₀, different concentrations of LXW7 were premixed with 2 μM Echistatin-FITC and incubated with αvβ3-K562 cells in binding buffer (PBS containing 10% FBS and 1 mM MnCl₂) on ice for 30 minutes. Cells were analyzed by flow cytometry, and IC₅₀ was calculated using GraphPad Prism software. Cell Staining and Specificity Assay: To test binding, 1 μM biotinylated LXW7 was incubated with various integrin-transfected K562 cells (αvβ3, αvβ5, αIIbβ3) and parent K562 cells on ice for 30 min. Cells were washed, incubated with streptavidin-PE, and analyzed by flow cytometry. Blocking Experiment: To confirm specificity, 10 μg of anti-human αvβ3 antibody (LM609) was premixed with 1 μM biotinylated LXW7 before incubation with U-87MG or A375M cells, followed by streptavidin-PE incubation and flow cytometry analysis. [1] |
| Animal Protocol |
Animal/Disease Models: Male SD (SD (Sprague-Dawley)) rats (250-280 g) underwent middle cerebral artery occlusion (MCAO) [3] Doses: 100 μg/kg Route of Administration: intravenous (iv) (iv)injection Experimental Results: Infarct volume and BWC were Dramatically diminished compared with the control group MCAO+PBS (control group) group. Tumor Xenograft Establishment: Female athymic nude mice (5-6 weeks old) were injected subcutaneously in the right flank with 5×10⁶ U-87MG or A375M tumor cells suspended in 200 μL PBS. Imaging studies commenced when tumors reached 0.5-1.0 cm in diameter (21-28 days later). In Vivo Optical Imaging: Mice were anesthetized. A tetravalent imaging complex was prepared by mixing 7.2 nmol of biotinylated peptide (LXW7 or controls) with 1.8 nmol of streptavidin-Cy5.5 in PBS overnight at 4°C. A total of 1.8 nmol of this complex was injected via the tail vein. Optical images were acquired at 6 hours post-injection using a Kodak IS2000MM Image Station. Ex Vivo Imaging: Mice were sacrificed after in vivo imaging, and organs (tumors, liver, kidneys, etc.) were excised for ex vivo fluorescent imaging. [1] |
| ADME/Pharmacokinetics |
In vivo imaging data indicated that the LXW7-biotin/streptavidin-Cy5.5 complex accumulated in tumors and was also detected in the kidneys 6 hours post-injection. Liver uptake was notably low compared to other RGD cyclopentapeptides. [1] |
| References |
[1]. The use of one-bead one-compound combinatorial library technology to discover high-affinity αvβ3 integrin and cancer targeting arginine-glycine-aspartic acid ligands with a built-in handle. Mol Cancer Ther. 2010 Oct;9(10):2714-23. [2]. Discovery and Characterization of a Potent and Specific Peptide Ligand Targeting Endothelial Progenitor Cells and Endothelial Cells for Tissue Regeneration. ACS Chem Biol. 2017 Apr 21;12(4):1075-1086. [3]. LXW7 ameliorates focal cerebral ischemia injury and attenuates inflammatory responses in activated microglia in rats. Braz J Med Biol Res. 2016 Aug 1;49(9):e5287. |
| Additional Infomation |
LXW7 (cyclic cGRGDdvc) is a disulfide-cyclized RGD peptide discovered through screening one-bead-one-compound (OBOC) combinatorial libraries against αvβ3 integrin-transfected K562 cells. It contains a built-in handle (PEG linker) at its C-terminus, which allows for conjugation (e.g., biotinylation) without significant loss of binding affinity, a key advantage over traditional "head-to-tail" cyclic RGD peptides. Molecular docking simulations suggested that the D-Asp residue adjacent to the RGD motif in LXW7 forms a salt bridge with Arg214 in the β-subunit, and its C-terminal carbonyl may coordinate with a Ca²⁺ in the ADMIDAS site, potentially contributing to its specific binding and calculated favorable interaction energy. It is proposed as a promising targeting ligand for molecular imaging and drug delivery to tumors and tumor neovasculature expressing αvβ3 integrin. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.2182 mL | 6.0910 mL | 12.1819 mL | |
| 5 mM | 0.2436 mL | 1.2182 mL | 2.4364 mL | |
| 10 mM | 0.1218 mL | 0.6091 mL | 1.2182 mL |