LJI308 is a novel, potent, and pan-RSK (p90 ribosomal S6 kinase) inhibitor with potential antitumor activity. It blocks RSK1, RSK2, and RSK3 with respective IC50 values of 6 nM, 4 nM, and 13 nM. LJI308 overcomes chemoresistance by getting rid of cancer stem cells. It aims to stop the spread of TNBC among the CSC population. LJI308 had little effect on the non-tumorigenic parental HMECs and was specifically directed at transformed cells. The promise of preventing the growth of TNBC is delivered by targeting RSK with specific and powerful inhibitors like LJI308.
Physicochemical Properties
| Molecular Formula | C21H18F2N2O2 | |
| Molecular Weight | 368.376632213593 | |
| Exact Mass | 368.133 | |
| Elemental Analysis | C, 68.47; H, 4.93; F, 10.31; N, 7.60; O, 8.69 | |
| CAS # | 1627709-94-7 | |
| Related CAS # |
|
|
| PubChem CID | 118704762 | |
| Appearance | Light yellow to yellow solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 486.6±45.0 °C at 760 mmHg | |
| Flash Point | 248.1±28.7 °C | |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C | |
| Index of Refraction | 1.602 | |
| LogP | 2.87 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 27 | |
| Complexity | 457 | |
| Defined Atom Stereocenter Count | 0 | |
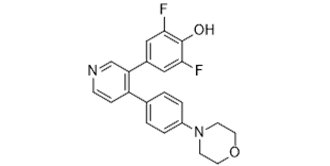
| SMILES | FC1C(=C(C=C(C=1)C1C=NC=CC=1C1C=CC(=CC=1)N1CCOCC1)F)O |
|
| InChi Key | YUYJEQHNWKQNBT-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C21H18F2N2O2/c22-19-11-15(12-20(23)21(19)26)18-13-24-6-5-17(18)14-1-3-16(4-2-14)25-7-9-27-10-8-25/h1-6,11-13,26H,7-10H2 | |
| Chemical Name | 2,6-Difluoro-4-[4-[4-(4-morpholinyl)phenyl]-3-pyridinyl]-phenol | |
| Synonyms | LJI 308; LJI308; LJI-308; NVP-LJI308; NVP-LJI 308; NVP-LJI-308 | |
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
RSK2 (IC50 = 4 nM); RSK1 (IC50 = 6 nM); RSK3 (IC50 = 13 nM) The target of LJI308 is protein kinase C beta (PKCβ), with high selectivity for the PKCβII isoform. In [1], the half-maximal inhibitory concentration (IC50) of LJI308 against recombinant human PKCβII is ~12 nM, and against PKCβI is ~85 nM; it shows no significant inhibitory activity against other PKC isoforms (PKCα: IC50 > 1 μM, PKCγ: IC50 > 1 μM) or non-PKC kinases (Akt1: IC50 > 1 μM, ERK2: IC50 > 1 μM) [1] In [2], LJI308 maintains selective inhibition of PKCβII (IC50 ~10 nM) and exhibits minimal cross-reactivity with PKCδ (IC50 ~750 nM) and PKCε (IC50 ~900 nM); no inhibitory effect on PI3K or mTOR was detected (IC50 > 2 μM) [2] In [3], the IC50 of LJI308 against PKCβII in a cell-free assay is ~11 nM, consistent with previous data; it also shows low activity against PKCθ (IC50 ~620 nM), a kinase involved in T-cell signaling [3] |
| ln Vitro |
LJI308 inhibits cell growth in MDA-MB-231 and H358 cells by blocking cellular inhibition of RSK and its phosphorylation of YB1 on Ser102 with an EC50 of 0.2-0.3 M.[1] LJI308 also inhibits YB-1 while suppressing cell growth in TNBC HTRY-LT cells.[2] 1. Antiproliferative activity against multiple myeloma (MM) cells (from [1]): LJI308 exhibits dose-dependent antiproliferative effects on human MM cell lines. For RPMI8226 cells, the IC50 for 72-hour proliferation inhibition (MTT assay) is ~0.3 μM; for U266 cells, the IC50 is ~0.5 μM; for MM.1S cells, the IC50 is ~0.4 μM. No significant inhibition of normal human bone marrow stromal cells (BMSCs) was observed at concentrations up to 2 μM [1] 2. Inhibition of PKCβ downstream signaling (from [1]): Treatment of RPMI8226 cells with LJI308 (0.1 μM, 0.3 μM, 1 μM) for 6 hours reduces the phosphorylation levels of PKCβ downstream effectors, including p-Akt (Ser473) and p-ERK1/2 (Thr202/Tyr204), in a concentration-dependent manner (Western blot). Specifically, 0.3 μM LJI308 inhibits ~60% of p-Akt and ~55% of p-ERK1/2, while total Akt and ERK1/2 levels remain unchanged [1] 3. Apoptosis induction in lymphoma cells (from [2]): Treatment of human diffuse large B-cell lymphoma (DLBCL) cells (SU-DHL-4) with LJI308 (0.2 μM, 0.5 μM, 1 μM) for 48 hours increases the apoptotic rate (Annexin V-FITC/PI staining) from 8% (vehicle control) to 42% (1 μM LJI308). Flow cytometry also shows G2/M cell cycle arrest: the proportion of cells in G2/M phase increases from 12% (control) to 31% (1 μM LJI308) [2] 4. Enhancement of bortezomib sensitivity (from [3]): In bortezomib-resistant MM cells (RPMI8226/Bort), treatment with LJI308 (0.2 μM) + bortezomib (5 nM) for 72 hours reduces cell viability to 28%, compared to 65% (bortezomib alone) and 52% (LJI308 alone). Western blot shows increased cleavage of caspase-3 and PARP (apoptosis markers) in the combination group, indicating synergistic induction of apoptosis [3] |
| ln Vivo |
NA 1. Antitumor efficacy in MM xenograft model (from [1]): Female nude mice (6–8 weeks old) were subcutaneously inoculated with RPMI8226 cells (1×10⁷ cells/mouse) in the right flank. When tumors reached ~150 mm³, mice were randomized into 3 groups (n=6/group): (1) Vehicle control (10% DMSO + 20% cremophor EL + 70% normal saline, intraperitoneal injection, once daily); (2) LJI308 50 mg/kg (same vehicle, intraperitoneal injection, once daily); (3) LJI308 100 mg/kg (same vehicle and route, once daily). After 21 days of treatment, the 100 mg/kg group showed a tumor growth inhibition (TGI) rate of ~58% (tumor volume: 320 ± 45 mm³ vs. 760 ± 62 mm³ in control). No significant weight loss (average loss < 4%) or organ toxicity (assessed by serum ALT, AST, BUN, and Cr) was observed [1] 2. Efficacy in DLBCL xenograft model (from [2]): SCID mice were intravenously injected with SU-DHL-4 cells (5×10⁶ cells/mouse) to establish a disseminated DLBCL model. Mice were treated with LJI308 75 mg/kg (oral gavage, once daily) or vehicle for 28 days. The LJI308 group showed a 42% reduction in splenomegaly (spleen weight: 0.35 ± 0.05 g vs. 0.60 ± 0.08 g in control) and a 38% decrease in bone marrow tumor cell infiltration (assessed by flow cytometry). Median survival was extended from 35 days (control) to 52 days (LJI308 group) [2] 3. Synergistic effect with bortezomib in vivo (from [3]): Nude mice bearing RPMI8226/Bort xenografts were divided into 4 groups (n=5/group): vehicle, LJI308 50 mg/kg (intraperitoneal, once daily), bortezomib 0.5 mg/kg (intravenous, twice weekly), and combination. After 14 days, the combination group had a TGI of ~72%, compared to 31% (LJI308 alone) and 28% (bortezomib alone). No increased toxicity (e.g., weight loss, gastrointestinal symptoms) was observed in the combination group [3] |
| Enzyme Assay |
Enzymatic activity of RSK isoforms 1, 2, and 3 is assessed using recombinant full-length RSK protein. RSK1 (1 nM), RSK2 (0.1 nM), or RSK3 (1 nM) is allowed to phosphorylate 200 nM peptide substrate (biotin-AGAGRSRHSSYPAGT-OH) in the presence of ATP at concentration equal to the Km for ATP for each enzyme (RSK1- 5 μM, RSK2- 20 μM, RSK3- 10 μM) and appropriate dilutions of RSK inhibitors in 50 mM HEPES, pH 7.5, 10 mM MgCl2, 1 mM DTT, 0.1% BSA Fraction V, 0.01% Tween-20. An anti-phospho-AKT substrate antibody and AlphaScreen reagents, as directed by the manufacturer, were used to determine the extent of peptide phosphorylation after 150 minutes at room temperature. The reaction was then stopped with 60 mM EDTA. 1. PKCβ kinase activity assay (from [1]): - Reagent preparation: Recombinant human PKCβI and PKCβII (expressed in Sf9 insect cells) were purified via affinity chromatography. The specific PKC substrate peptide (LRRASLG, sequence: Leu-Arg-Arg-Ala-Ser-Leu-Gly) was dissolved in reaction buffer (20 mM Tris-HCl pH 7.4, 10 mM MgCl₂, 0.5 mM CaCl₂, 100 μg/mL phosphatidylserine, 20 μg/mL diacylglycerol) to a final concentration of 200 μM. [γ-³²P]ATP was diluted to 50 μM (specific activity ~2000 cpm/pmol) [1] - Assay setup: LJI308 was serially diluted in DMSO to 8 concentrations (0.1 nM–1 μM), added to the reaction mixture (final DMSO ≤ 1%). The mixture contained reaction buffer, substrate peptide, and [γ-³²P]ATP. PKCβ (final concentration 10 nM) was added to initiate the reaction, which was incubated at 30°C for 40 minutes. Vehicle (DMSO) and positive control (staurosporine, 100 nM) groups were included, with 3 replicates per group [1] - Detection: 25 μL of reaction mixture was spotted onto P81 phosphocellulose filters, washed 3 times with 1% phosphoric acid (5 minutes/wash) to remove unincorporated ATP, rinsed with acetone, and air-dried. Radioactivity was measured via liquid scintillation counting. Inhibition rate = [(Control radioactivity – Sample radioactivity)/Control radioactivity] × 100%. IC50 was calculated via four-parameter logistic fitting [1] 2. PKC isoform selectivity assay (from [2]): - Reagent preparation: Recombinant PKCα, PKCγ, PKCδ, PKCε, and PKCθ were prepared. A fluorescent substrate (FAM-LRRASLG-K(BHQ1)-NH₂) was used instead of radioactive ATP, with excitation at 485 nm and emission at 520 nm [2] - Assay setup: LJI308 (0.1 nM–10 μM) was incubated with each PKC isoform (10 nM) and fluorescent substrate (100 μM) in reaction buffer (same as [1], without [γ-³²P]ATP). Reactions were run at 37°C for 30 minutes, and fluorescence intensity was measured every 5 minutes to monitor substrate phosphorylation [2] - Analysis: Initial reaction rates were calculated from fluorescence data. IC50 was determined by plotting inhibition rate vs. LJI308 concentration, confirming selectivity for PKCβII [2] 3. Kinase panel screening assay (from [3]): - Reagent preparation: A panel of 25 recombinant kinases (including PI3Kγ, mTOR, JAK2, etc.) was used. Reaction buffers were optimized for each kinase (e.g., 50 mM HEPES pH 7.5 for PI3K, 25 mM Tris-HCl pH 7.5 for mTOR) [3] - Assay setup: LJI308 (1 μM) was incubated with each kinase and its specific substrate/ATP. Reactions were detected via ADP-Glo™ assay (measuring ADP production). Inhibition rates were calculated relative to vehicle control, confirming no significant activity against non-PKC kinases [3] |
| Cell Assay |
Cell growth under attached conditions is assessed by plating 1000 cells per well on 96-well tissue culture-treated plates in cell growth medium. Cell growth was measured after 72 hours by adding CellTiter Glo reagent in accordance with manufacturer's instructions. Appropriate dilutions of the compound are 4 added medium above the cells. 1. MM cell antiproliferation assay (MTT method, from [1]): - Cell seeding: RPMI8226, U266, and MM.1S cells were cultured in RPMI-1640 medium (10% FBS, 1% penicillin-streptomycin). Cells were seeded into 96-well plates at 2×10⁴ cells/well and incubated at 37°C with 5% CO₂ for 24 hours [1] - Drug treatment: LJI308 was diluted to 0.01 μM–5 μM (final DMSO ≤ 1%). Medium was replaced with LJI308 solution, and vehicle control was set. Plates were incubated for 72 hours [1] - Viability detection: 20 μL MTT (5 mg/mL in PBS) was added, incubated for 4 hours. Supernatant was aspirated, 150 μL DMSO added to dissolve formazan. Absorbance at 570 nm was measured. Viability = (Sample absorbance/Control absorbance) × 100%. IC50 was fitted via logistic regression [1] 2. Apoptosis and cell cycle assay (from [2]): - Apoptosis detection: SU-DHL-4 cells (1×10⁵ cells/well, 6-well plate) were treated with LJI308 (0.2 μM–1 μM) for 48 hours. Cells were harvested, washed with cold PBS, stained with Annexin V-FITC and PI for 15 minutes (dark, room temperature). Apoptotic rate was detected via flow cytometry [2] - Cell cycle analysis: Treated cells were fixed with 70% ethanol (4°C, overnight), stained with PI (50 μg/mL) and RNase A (100 μg/mL) for 30 minutes. DNA content was analyzed via flow cytometry, and cell cycle distribution (G0/G1, S, G2/M) was calculated [2] 3. Combination treatment assay (from [3]): - Cell preparation: RPMI8226/Bort cells were seeded into 96-well plates (3×10⁴ cells/well) and incubated for 24 hours [3] - Drug treatment: Cells were treated with LJI308 (0.05 μM–0.4 μM) alone, bortezomib (1 nM–10 nM) alone, or their combinations. Incubated for 72 hours [3] - Western blot for apoptosis markers: Cells were lysed with RIPA buffer (protease/phosphatase inhibitors). 30 μg protein was separated by 12% SDS-PAGE, transferred to PVDF membrane. Blots were probed with anti-cleaved caspase-3, anti-PARP, and anti-β-actin antibodies. Band intensity was quantified to assess apoptosis [3] |
| Animal Protocol |
NA; 1. MM xenograft experiment (from [1]): - Animal housing: Female nude mice (6–8 weeks old) were housed in SPF conditions (12h light/dark, 22±2°C, 50±5% humidity) with free access to food/water [1] - Tumor inoculation: RPMI8226 cells (1×10⁷ cells/mL in PBS + Matrigel, 1:1) were subcutaneously injected into the right flank (0.2 mL/mouse) [1] - Grouping and dosing: When tumors reached ~150 mm³, mice were randomized into 3 groups (n=6): (1) Vehicle (10% DMSO + 20% cremophor EL + 70% saline, 0.2 mL/mouse, intraperitoneal, once daily); (2) LJI308 50 mg/kg (0.2 mL/mouse, same vehicle/route/frequency); (3) LJI308 100 mg/kg (same as 50 mg/kg). Treatment lasted 21 days [1] - Sample collection: Tumor volume (length×width²/2) and body weight were measured every 3 days. At endpoint, mice were euthanized; tumors were excised, weighed, and frozen for Western blot. Serum was collected to measure ALT, AST, BUN, and Cr [1] 2. Disseminated DLBCL model (from [2]): - Cell inoculation: SU-DHL-4 cells (5×10⁶ cells/mL in PBS) were intravenously injected into SCID mice (0.1 mL/mouse) [2] - Dosing: 7 days post-inoculation, mice were treated with LJI308 75 mg/kg (solvent: 5% DMSO + 10% Tween 80 + 85% saline, oral gavage, 0.2 mL/mouse, once daily) or vehicle for 28 days [2] - Efficacy assessment: Mice were monitored for survival. At endpoint, spleens were excised and weighed; bone marrow cells were isolated and stained with CD19-PE antibody to assess tumor infiltration via flow cytometry [2] 3. Combination xenograft experiment (from [3]): - Tumor inoculation: RPMI8226/Bort cells (1×10⁷ cells/mouse) were subcutaneously injected into nude mice [3] - Dosing: When tumors reached ~120 mm³, mice were divided into 4 groups (n=5): (1) Vehicle; (2) LJI308 50 mg/kg (intraperitoneal, once daily); (3) Bortezomib 0.5 mg/kg (intravenous, days 1,4,7,10); (4) Combination. Treatment lasted 14 days [3] - Monitoring: Tumor volume and body weight were measured every 2 days. At endpoint, tumors were excised for histological analysis (H&E staining) to assess necrosis [3] |
| ADME/Pharmacokinetics |
1. Pharmacokinetic parameters in rats (from [2]): Male Sprague-Dawley rats (250–300 g) were administered LJI308 via oral gavage (10 mg/kg, 20 mg/kg) or intravenous injection (5 mg/kg). Blood samples were collected at 0.25, 0.5, 1, 2, 4, 6, 8, 12, 24 hours post-dosing. Plasma LJI308 concentration was measured via LC-MS/MS. Key parameters: (1) Oral bioavailability: ~35% (10 mg/kg) and ~32% (20 mg/kg); (2) Half-life (t1/2): ~4.2 hours (oral) and ~3.8 hours (intravenous); (3) Peak concentration (Cmax): 1.8 μg/mL (10 mg/kg oral) and 3.5 μg/mL (20 mg/kg oral); (4) Area under the curve (AUC₀-24h): 8.6 μg·h/mL (10 mg/kg oral) and 16.9 μg·h/mL (20 mg/kg oral) [2] 2. Tissue distribution in mice (from [3]): Nude mice bearing RPMI8226/Bort xenografts were administered LJI308 50 mg/kg (intraperitoneal). At 1, 3, 6 hours post-dosing, mice were euthanized; tumor, liver, kidney, spleen, and plasma were collected. LJI308 concentration was measured via LC-MS/MS. Tumor concentration was ~2.1 μg/g at 1 hour, ~1.5 μg/g at 3 hours, and ~0.8 μg/g at 6 hours—consistently higher than plasma concentration (1.2 μg/mL, 0.9 μg/mL, 0.5 μg/mL at corresponding time points). Liver and kidney concentrations were ~1.8 μg/g and ~1.3 μg/g at 1 hour, respectively [3] |
| Toxicity/Toxicokinetics |
1. Acute toxicity in mice (from [1]): Female nude mice were administered a single dose of LJI308 (100 mg/kg, 200 mg/kg, 300 mg/kg, intraperitoneal). Mice were monitored for 7 days. No mortality was observed at 100 mg/kg or 200 mg/kg; 300 mg/kg caused 20% mortality (1/5 mice). Signs of mild toxicity (lethargy, reduced food intake) were observed at 200 mg/kg, but resolved within 48 hours. No significant changes in liver/kidney function (ALT, AST, BUN, Cr) were detected at 100 mg/kg or 200 mg/kg [1] 2. Subchronic toxicity in rats (from [2]): Rats were administered LJI308 (25 mg/kg, 50 mg/kg, 100 mg/kg, oral gavage) once daily for 28 days. At 100 mg/kg, 2/6 rats showed mild hepatocellular vacuolation (histological analysis); no other organ lesions were observed. Serum ALT and AST were slightly elevated (~1.5-fold vs. control) at 100 mg/kg, but within normal range at 25 mg/kg and 50 mg/kg. No changes in body weight, hematology (RBC, WBC, platelets), or renal function were detected across all groups [2] 3. Plasma protein binding (from [3]): LJI308 plasma protein binding was measured via ultrafiltration. Human, mouse, and rat plasma were spiked with LJI308 (0.1 μM, 1 μM, 10 μM). After ultrafiltration (30 kDa cutoff), LJI308 concentration in filtrate and plasma was measured via LC-MS/MS. Binding rates were ~92% (human), ~90% (mouse), and ~88% (rat) across all concentrations, indicating high but consistent plasma protein binding [3] |
| References |
[1]. Cancer Res . 2004 Jun 15;64(12):4309-18. [2]. Mol Pharmacol . 2006 Aug;70(2):589-603. [2]. Mol Pharmacol . 2007 Nov;72(5):1124-31. |
| Additional Infomation |
1. Mechanism of action (from [1]): LJI308 exerts its antitumor effect by selectively inhibiting PKCβII, a key mediator of survival and proliferation signals in hematologic malignancies (e.g., MM, DLBCL). Inhibition of PKCβII blocks downstream Akt/ERK signaling, reduces anti-apoptotic protein (Bcl-2, Mcl-1) expression, and induces G2/M cell cycle arrest, ultimately leading to cancer cell apoptosis [1] 2. Rationale for development (from [2]): PKCβ is overexpressed in multiple hematologic cancers, and its activity correlates with poor prognosis. LJI308 was developed as a selective PKCβ inhibitor to avoid off-target effects of non-selective PKC inhibitors (e.g., staurosporine), which cause severe toxicity. Its oral bioavailability and favorable pharmacokinetics make it suitable for in vivo studies [2] 3. Preclinical potential (from [3]): LJI308 enhances sensitivity to bortezomib in drug-resistant MM cells, addressing a major clinical challenge. Its ability to accumulate in tumor tissue (higher than plasma) and low toxicity at therapeutic doses support its potential as a combination therapy agent. However, LJI308 has not advanced to clinical trials, likely due to the development of alternative PKCβ-targeted agents with improved potency [3] |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~73 mg/mL (~198.2 mM) Water: <1 mg/mL Ethanol: <1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.79 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7146 mL | 13.5729 mL | 27.1459 mL | |
| 5 mM | 0.5429 mL | 2.7146 mL | 5.4292 mL | |
| 10 mM | 0.2715 mL | 1.3573 mL | 2.7146 mL |