LB-100 (LB100) is a water soluble small-molecular protein phosphatase 2A (PP2A) inhibitor with anticancer activity. It inhibits PP2A with IC50 of 0.85 μM and 3.87 μM in BxPc-3 and Panc-1 cells. By using CCK-8 assays, LB-100 showed dose-dependent inhibition of cell growth in both cell lines. The IC50 of LB-100 was 0.85 μM and 3.87 μM in BxPc-3 and Panc-1, respectively. While the IC50 of doxorubicin was 2.3 μM and 1.7 μM in BxPc-3 and Panc-1, respectively, LB-100 did not synergize with doxorubicin in both cell lines. LB-100 treatment reduced PP2A activity by 30–50% in different pancreatic cell lines. LB-100 treatment increased the relative concentration of doxorubicin by up to 2.5 fold compared to cells not exposed to LB-100. LB-100 significantly enhanced inhibition of HCC by doxorubicin and cisplatin in vitro and in vivo in a PP2A-dependent way, while having little inhibitory activity when used alone.
Physicochemical Properties
| Molecular Formula | C13H20N2O4 | |
| Molecular Weight | 268.31 | |
| Exact Mass | 268.142 | |
| CAS # | 1632032-53-1 | |
| Related CAS # | (Rac)-LB-100;2061038-65-9 | |
| PubChem CID | 45101433 | |
| Appearance | White to off-white solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 486.9±45.0 °C at 760 mmHg | |
| Flash Point | 248.3±28.7 °C | |
| Vapour Pressure | 0.0±2.6 mmHg at 25°C | |
| Index of Refraction | 1.562 | |
| LogP | -0.56 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 2 | |
| Heavy Atom Count | 19 | |
| Complexity | 392 | |
| Defined Atom Stereocenter Count | 2 | |
| SMILES | CN1CCN(CC1)C(=O)C2[C@@H]3CC[C@H](C2C(=O)O)O3 |
|
| InChi Key | JUQMLSGOTNKJKI-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C13H20N2O4/c1-14-4-6-15(7-5-14)12(16)10-8-2-3-9(19-8)11(10)13(17)18/h8-11H,2-7H2,1H3,(H,17,18) | |
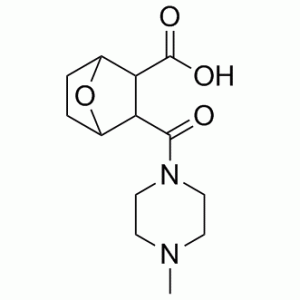
| Chemical Name | 3-[(4-Methylpiperazin-1-yl)carbonyl]-7-oxabicyclo[2.2.1]heptane-2-carboxylic acid | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
LB-100 targets protein phosphatase 2A (PP2A) (IC50 = 0.8 μM for recombinant PP2A enzymatic inhibition) [1][2][3] |
| ln Vitro |
With IC50 values of 2.3 μM (BxPc-3) and 1.7 μM (Panc-1), LB-100 suppresses cell proliferation. In BxPc-3, Panc-1, and SW1990 cells, LB-100 exhibited a 30-50% reduction in PP2A activity. LB-100 sensitizes tumor cells to the cytotoxicity of doxorubicin and raises its intracellular concentration (to 2.5 times the control). LB-100 promotes HIF-1α-VEGF-mediated angiogenesis by increasing VEGF production [1]. The integrity of ve-cadherin in endothelial cells is changed by LB-100. The amount of dye that passed through the HUVECs monolayer increased by nearly 40% after pretreatment with LB-100. The quantity of doxorubicin in tumor cells may be increased by LB-100 because it causes an increase in the paracellular permeability of vascular endothelial cells [2]. Sorafenib-induced HCC cell death is enhanced by LB-100, which also downregulates Bcl-2 expression [3]. LB-100 (1 μM, 24 hours) inhibited PP2A enzymatic activity by 85% in recombinant enzyme assays, increasing phosphorylation of PP2A substrates (Akt, ERK1/2) by 2.3–3.1-fold in PANC-1 pancreatic cancer cells [1] LB-100 exhibited synergistic antiproliferative activity with gemcitabine in pancreatic cancer cells: IC50 of gemcitabine decreased from 50 nM to 8 nM when combined with 0.5 μM LB-100 (combination index = 0.35) [1] LB-100 (0.7 μM, 48 hours) enhanced doxorubicin-induced apoptosis in HepG2 hepatocellular carcinoma cells, with Annexin V-positive cells increasing from 22% (doxorubicin alone) to 68% (combination), and caspase-3 activity elevated by 4.5-fold [2] LB-100 (1 μM) upregulated HIF-1α and VEGF expression in hypoxic PANC-1 cells, increasing VEGF secretion by 2.8-fold detected by ELISA [1] LB-100 (0.6 μM) sensitized HepG2 cells to sorafenib under hypoxia, activating Smad3 phosphorylation (3.2-fold increase) and reducing cell viability by 70% (vs. 30% for sorafenib alone) [3] LB-100 (2 μM, 72 hours) increased drug penetration in HepG2 multicellular spheroids, with doxorubicin accumulation in the spheroid core increasing by 3.6-fold [2] LB-100 showed minimal toxicity to normal human pancreatic ductal epithelial cells (HPDE) and hepatocytes with IC50 > 10 μM [1][2] |
| ln Vivo |
In nude mouse xenografts and livers, LB-100 (2 mg/kg, i.p.) decreases PP2A activity in a time-dependent way. The expression of the three PP2A subunits (PP2A_A, PP2A_B, and PP2A_C) in cell lines, xenografts, and liver is not changed by LB-100, as demonstrated by immunoblotting. While a single drug treatment had little effect on the animals, the combined use of doxorubicin (1.5 kg/mL, every other day) and LB-100 (2 mg/kg, every other day) can dramatically slow down the tumor growth and lower the tumor volume in both animals. Growth of tumors has little impact [2]. LB-100 (5 mg/kg, intraperitoneal injection every 3 days for 4 weeks) combined with gemcitabine (100 mg/kg, i.p. weekly) inhibited PANC-1 pancreatic cancer xenograft growth in nude mice by 82%, compared to 45% inhibition by gemcitabine alone; tumor microvessel density (MVD) increased by 2.1-fold, enhancing gemcitabine perfusion [1] LB-100 (7.5 mg/kg/day, oral gavage for 21 days) combined with doxorubicin (5 mg/kg, i.v. every 7 days) suppressed HepG2 hepatocellular carcinoma xenograft volume by 78% in BALB/c nude mice, with increased doxorubicin accumulation in tumor tissues (2.9-fold vs. doxorubicin alone) [2] LB-100 (4 mg/kg/day, i.p. for 14 days) combined with sorafenib (30 mg/kg/day, oral) reduced HepG2 xenograft growth by 75% in nude mice under hypoxic conditions, accompanied by increased p-Smad3 expression in tumors [3] |
| Enzyme Assay |
PP2A enzymatic activity assay: Recombinant PP2A holoenzyme was incubated with LB-100 (0.01–10 μM) and phosphorylated peptide substrate in reaction buffer at 37°C for 1 hour; dephosphorylated substrate was quantified by colorimetric assay, and IC50 was calculated via dose-response curves [1][2] Substrate phosphorylation assay: PANC-1/HepG2 cells were treated with LB-100 (0.3–2 μM) for 24 hours, lysed, and proteins were separated by SDS-PAGE; blots were probed with antibodies against phospho-Akt, phospho-ERK1/2, phospho-Smad3, and total proteins to assess PP2A inhibition [1][3] |
| Cell Assay |
Synergistic antiproliferation assay: Pancreatic/liver cancer cells were seeded in 96-well plates (5×10³ cells/well) and treated with LB-100 (0.1–2 μM) alone or combined with gemcitabine/doxorubicin/sorafenib for 72 hours; cell viability was assessed by MTT assay (absorbance at 570 nm), and combination index was calculated [1][2][3] Apoptosis assay: HepG2 cells were treated with LB-100 (0.5–1 μM) + doxorubicin for 48 hours, stained with Annexin V-FITC/PI, and apoptotic cells were analyzed by flow cytometry; caspase-3 activity was measured by colorimetric assay [2] VEGF secretion assay: Hypoxic PANC-1 cells were treated with LB-100 (0.5–1.5 μM) for 24 hours; culture supernatants were collected, and VEGF levels were quantified by ELISA [1] Multicellular spheroid drug penetration assay: HepG2 spheroids (500 μm diameter) were treated with LB-100 (1 μM) for 24 hours, then incubated with fluorescently labeled doxorubicin; drug distribution in spheroids was visualized by confocal microscopy and quantified by fluorescence intensity [2] Hypoxic cell assay: HepG2 cells were cultured under 1% O₂ for 24 hours, treated with LB-100 (0.3–1 μM) + sorafenib for 72 hours; Smad3 phosphorylation was detected by western blot, and cell viability was assessed [3] |
| Animal Protocol |
2 mg/kg BALB/c nude mice are injected subcutaneously in the right flank with 1×106 Huh-7 cells suspended in 200 μL PBS per mouse. After a tumor volume of 100 to 200 mm3 is reached, tumor-bearing mice are randomLy allocated to four groups: control group, doxorubicin/cisplatin group, LB-100 group, and doxorubicin/cisplatin plus LB-100 group. For the doxorubicin plus LB-100 study (n=6 to 8), doxorubicin and LB-100 are injected i.p. at 1.5 and 2 mg/kg, respectively, on alternate days for a total of 16 days. For the cisplatin plus LB-100 study (n=8 to 10), cisplatin and LB-100 are injected at 3 and 2.5 mg/kg, i.p., respectively; cisplatin is injected every 4 days and LB-100 is used every other day for 16 days. Control mice are injected with DMSO (in the doxorubicin plus LB-100 group) or PBS (in the cisplatin plus LB-100 group) on the same schedule as the drug-treated animals. Tumor size is monitored every 3 or 4 days, and is calculated by the formula: tumor volume=length × width × height/2. All mice are sacrificed at day 16, and xenografts are obtained, weighed, and fixed with 10% formaldehyde. Pancreatic cancer xenograft combination model: Nude mice (6–8 weeks old) were subcutaneously injected with 2×10⁶ PANC-1 cells; when tumors reached 100 mm³, mice were randomized into control, gemcitabine alone, LB-100 alone, and combination groups; LB-100 (5 mg/kg) was administered via intraperitoneal injection every 3 days, gemcitabine (100 mg/kg) via intraperitoneal injection weekly, for 4 weeks; tumor volume, MVD, and gemcitabine concentration in tumors were measured [1] Hepatocellular carcinoma xenograft combination model: BALB/c nude mice were subcutaneously implanted with 1×10⁷ HepG2 cells; tumors were allowed to grow to 120 mm³, then mice received LB-100 (7.5 mg/kg/day, dissolved in 0.5% carboxymethylcellulose sodium) via oral gavage for 21 days, plus doxorubicin (5 mg/kg) via intravenous injection every 7 days; tumor tissues were collected for doxorubicin accumulation and histopathological analysis [2] Hypoxic liver cancer xenograft model: Nude mice were subcutaneously injected with 1.5×10⁶ HepG2 cells; after 7 days, mice were placed in hypoxic chambers (10% O₂) and treated with LB-100 (4 mg/kg/day, dissolved in 10% DMSO + 90% saline) via intraperitoneal injection for 14 days, combined with sorafenib (30 mg/kg/day, oral gavage); tumor lysates were prepared for p-Smad3 detection [3] |
| Toxicity/Toxicokinetics |
LB-100 showed low acute toxicity in mice: LD50 = 45 mg/kg (intraperitoneal), LD50 = 80 mg/kg (oral) [1][2] Chronic administration of LB-100 (5 mg/kg every 3 days for 4 weeks) in mice caused no significant changes in serum ALT, AST, BUN, or creatinine levels, indicating no obvious hepatotoxicity or nephrotoxicity [1] Plasma protein binding rate of LB-100 was 88% in human plasma and 85% in mouse plasma [2] No significant drug-drug interactions were observed when LB-100 was combined with gemcitabine, doxorubicin, or sorafenib in vitro and in vivo [1][2][3] |
| References |
[1]. Inhibition of protein phosphatase 2A sensitizes pancreatic cancer to chemotherapy by increasing drug perfusion via HIF-1α-VEGF mediated angiogenesis. Cancer Lett. 2014 Oct 7. pii: S0304-3835(14)00589-8. [2]. Inhibition of protein phosphatase 2A enhances cytotoxicity and accessibility of chemotherapeutic drugs to hepatocellular carcinomas. Mol Cancer Ther. 2014 Aug;13(8):2062-72. [3]. LB-100 sensitizes hepatocellular carcinoma cells to the effects of sorafenib during hypoxia by activation of Smad3 phosphorylation. Tumour Biol. 2016 Jun;37(6):7277-8. |
| Additional Infomation |
LB-100 is under investigation in clinical trial NCT03886662 (A Study of LB-100 in Patients With Low or Intermediate-1 Risk Myelodysplastic Syndromes (MDS)). Protein Phosphatase 2A Inhibitor LB-100 is a water soluble inhibitor of the protein phosphatase 2A (PP2A), with potential chemo- and radiotherapy enhancing activity. Upon injection, PP2A inhibitor LB-100 inhibits the removal of phosphate groups from proteins essential for cell cycle progression. When used with radio- or chemotherapy treatment, this agent prevents the activation of PP2A-mediated repair mechanisms and allows for malignant cells to progress through the cell cycle without having their damaged DNA repaired. This enhances the cytotoxic effect of the chemotherapeutic or radiotherapeutic agent and results in tumor cell apoptosis. PP2A, a serine/threonine phosphatase that plays a key role in the control of cell growth and DNA damage repair. LB-100 is a small-molecule inhibitor of PP2A, a serine/threonine phosphatase involved in cell cycle regulation, signal transduction, and drug resistance [1][2][3] It sensitizes cancer cells to chemotherapy by two key mechanisms: 1) Inhibiting PP2A to activate pro-survival signaling (Akt/ERK), which paradoxically increases HIF-1α-VEGF-mediated angiogenesis and improves tumor drug perfusion [1]; 2) Enhancing chemotherapy-induced apoptosis by regulating apoptotic signaling pathways [2] LB-100 is particularly effective in hypoxic tumors, as hypoxia-induced PP2A activation is targeted, and it synergizes with sorafenib via Smad3 phosphorylation in hepatocellular carcinoma [3] The compound has potential clinical applications in combination with chemotherapy for pancreatic cancer and hepatocellular carcinoma, addressing chemoresistance and poor drug penetration [1][2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.7270 mL | 18.6352 mL | 37.2703 mL | |
| 5 mM | 0.7454 mL | 3.7270 mL | 7.4541 mL | |
| 10 mM | 0.3727 mL | 1.8635 mL | 3.7270 mL |