Physicochemical Properties
| Molecular Formula | C24H30N6O2S |
| Molecular Weight | 466.6 |
| Exact Mass | 466.215 |
| CAS # | 294623-49-7 |
| PubChem CID | 9912381 |
| Appearance | White to off-white solid powder |
| LogP | 3.868 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 33 |
| Complexity | 733 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | FIVYCSWOCXEWSE-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H30N6O2S/c1-29-13-15-30(16-14-29)23-27-20(17-33-23)18-5-7-19(8-6-18)21(31)28-24(9-3-2-4-10-24)22(32)26-12-11-25/h5-8,17H,2-4,9-10,12-16H2,1H3,(H,26,32)(H,28,31) |
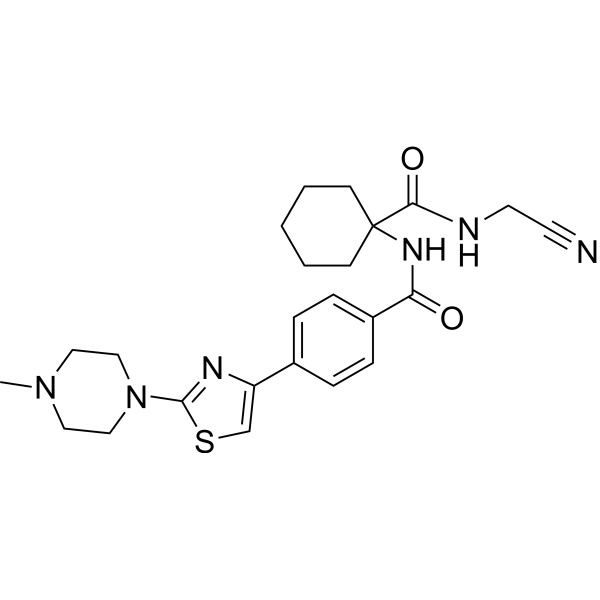
| Chemical Name | N-[1-(cyanomethylcarbamoyl)cyclohexyl]-4-[2-(4-methylpiperazin-1-yl)-1,3-thiazol-4-yl]benzamide |
| Synonyms | L-006235; L 006235; L006235 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In the rabbit bone resorption experiment, L-006235 inhibits bone resorption with an IC50 of 5 nM[1]. In HepG2 cells, L-006235 (10 μM; 1 h) exhibits punctate fluorescence throughout the cytoplasm [2]. |
| ln Vivo | L-006235, a suggested share nutrient, decreases N-telopeptide (NTx) and creatine nutrient (Cre) in rhesus monkeys by up to 76% when administered poqd for 8-11 days [1]. L-006235 (20 mg/kg; po; deposition) has a lengthy terminal half-life (204 minutes), a high bioavailability (68%), and a Cmax (1.4 μM) [1]. |
| Animal Protocol |
Animal/Disease Models: Rhesus monkey (15 years old) underwent ovariectomy (OVX) [1] Doses: 0.6, 3, 15 mg/kg Route of Administration: Orally one time/day for 8-11 days Experimental Results: Average reduction in uNTx/Cre The 76%, 68% and 31% doses were 15, 3 and 0.6mg/kg respectively. |
| References |
[1]. Design and synthesis of tri-ring P3 benzamide-containing aminonitriles as potent, selective, orally effective inhibitors of cathepsin K. J Med Chem. 2005 Dec 1;48(24):7520-34. [2]. Lysosomotropism of basic cathepsin K inhibitors contributes to increased cellular potencies against off-target cathepsins and reduced functional selectivity. J Med Chem. 2005 Dec 1;48(24):7535-43. [3]. Cathepsin K inhibitors prevent bone loss in estrogen-deficient rabbits. J Bone Miner Res. 2011 Feb;26(2):252-62. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~31.25 mg/mL (~66.97 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.46 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.46 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.46 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1432 mL | 10.7158 mL | 21.4316 mL | |
| 5 mM | 0.4286 mL | 2.1432 mL | 4.2863 mL | |
| 10 mM | 0.2143 mL | 1.0716 mL | 2.1432 mL |