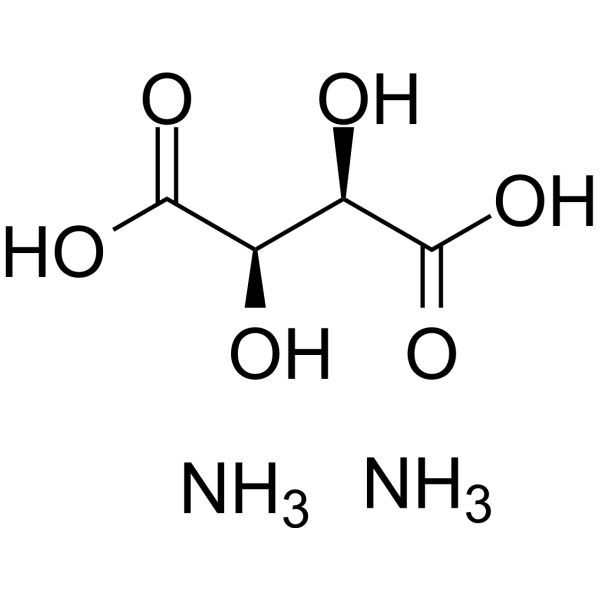
Physicochemical Properties
| Molecular Formula | C4H12N2O6 |
| Molecular Weight | 184.15 |
| Exact Mass | 184.069 |
| CAS # | 3164-29-2 |
| Related CAS # | L-Tartaric acid;87-69-4 |
| PubChem CID | 2724224 |
| Appearance |
Colorless, crystalline (sand-like) solid or white granule White crystals Crystals or white granules |
| Density | 1.601 g/mL at 25 °C(lit.) |
| Boiling Point | 399.3ºC at 760mmHg |
| Flash Point | 209.4ºC |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 12 |
| Complexity | 123 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | C(C(C(=O)O)O)(C(=O)O)O.N.N |
| InChi Key | NGPGDYLVALNKEG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C4H6O6.2H3N/c5-1(3(7)8)2(6)4(9)10;;/h1-2,5-6H,(H,7,8)(H,9,10);2*1H3 |
| Chemical Name | diazanium;2,3-dihydroxybutanedioate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Toxicity/Toxicokinetics |
Toxicity Summary IDENTIFICATION AND USE: Diammonium L-(+)-tartrate forms crystals or white granules. It is used in the textile industry. It is also suitable for clinical dosimetry using electron spin resonance (ESR). HUMAN STUDIES: Use of a 10% solution at pH 7 on patients' eyes following chemical burns seems to have caused no recognized additional injury, but the treatment is painful, especially if solution has decomposed and is no longer neutral. ANIMAL STUDIES: A 10% solution is not completely innocuous. Such a solution at pH 7 applied continuously for 30 minutes to a rabbit's eye after mechanical removal of epithelium caused no permanent damage, but did cause edema of the cornea lasting several days. Non-Human Toxicity Values LD50 Rabbit iv 113 mg/kg LD50 Rabbit sc 1130 mg/kg |
| References |
[1]. Growth, molecular structure, NBO analysis and vibrational spectral analysis of l-tartaric acid single crystal. Spectrochim Acta A Mol Biomol Spectrosc. 2014 Apr 5;123:127-41. [2]. L-tartaric acid synthesis from vitamin C in higher plants. Proc Natl Acad Sci U S A. 2006 Apr 4;103(14):5608-13. [3]. L-Tartaric Acid Exhibits Antihypertensive and Vasorelaxant Effects: The Possible Role of eNOS/NO/cGMP Pathways. Cardiovasc Hematol Agents Med Chem. 2023;21(3):202-212. [4]. Re-evaluation of l(+)-tartaric acid (E 334), sodium tartrates (E 335), potassium tartrates (E 336), potassium sodium tartrate (E 337) and calcium tartrate (E 354) as food additives. EFSA J. 2020 Mar 11;18(3):e06030. |
| Additional Infomation |
Ammonium tartrate is a white crystalline solid. It is soluble in water. The primary hazard is the threat to the environment. Immediate steps should be taken to limit its spread to the environment. It is used to manufacture fabrics and in medicine. Therapeutic Uses /EXPL THER/ This study is one step in the search for an ESR dosimeter material with a higher signal intensity than the commonly used l-a-alanine, to be useful in the clinical dose range (approximately 0.1-20 Gy). The substance ammonium tartrate was found and investigated regarding signal intensity, radical stability, dose response and dose resolution. The ESR signal intensity of ammonium tartrate was shown to be more than twice the intensity of the alanine signal. The data indicate that an unstable radiation induced radical contributes to the ESR signal initially; after a couple of hours it has converted to a secondary radical which has a decay slow enough to be considered stable during the first two weeks after irradiation. Ammonium tartrate has a linear dose response in the investigated range of 0.5-4000 Gy and a dose resolution of 0.1 Gy at the 0.5 Gy level where, as a comparison, the corresponding value for alanine is 0.3 Gy. We thus find the substance suitable for clinical dosimetry. /EXPL THER/ The crystalline substance ammonium tartrate is investigated with respect to its suitability as a clinical dosimetry material. The properties investigated are: the radical stability for absorbed doses relevant for clinical use, the improvement in sensitivity when the crystals are deuterated and the linear electron transfer (LET)-dependence. After photon irradiation to an absorbed dose of 20 Gy, the signal increases rapidly during the first 6 hr. After this period, the changes are more moderate and can be corrected for. The signal-to-noise ratio of irradiated ammonium tartrate is twice the corresponding value of alanine. By deuterating the crystals, the sensitivity can be further improved by a factor of 1.4. As expected, the signal decreases with increasing LET of the radiation, but no changes in the spectrum shape was observed. /EXPL THER/ This paper continues analyses on organic compounds for application in neutron dosimetry performed through electron spin resonance (ESR). Here, the authors present the results obtained by ESR measurements of a blend of ammonium tartrate dosemeters and gadolinium oxide (5% by weight). The choice of low amount of Gd is due to the need of improving neutron sensitivity while not significantly influencing tissue equivalence. A study of the effect of gadolinium presence on tissue equivalence was carried out. The experiments show that the neutron sensitivity is enhanced by more than an order of magnitude even with this small additive content. Monte Carlo simulations on the increment of energy release due to gadolinium presence were carried, and the results were in good agreement with the experimental data. /EXPL THER/ Use of /a 10% solution of ammonium tartrate at pH 7/ on patients' eyes following chemical burns seems to have caused no recognized additional injury, but the treatment is painful, especially if solution has decomposed and is no longer neutral. Solutions of ammonium tartrate, as well as solutions of other ammonium salts, dissolve certain metal salts which are poorly soluble in water by forming complexes with them. The solubilizing action of ammonium tartrate on calcium carbonate, lead carbonate, and copper precipitates has been exploited for dissolving incrustations of these compounds from the cornea of enucleated pig eyes, living rabbit eyes, and eyes of patients. Many observations have been published on the use of this treatment, in many instances applied indiscriminately and illogically where no metal incrustations existed. The rational basis for use of complexing ammonium salts such as ammonium tartrate to solubilize deposits of lime and other metals in the cornea apparently was forgotten during the half century after the introduction of this treatment, and an unfortunate custom developed of treating all sorts of alkali burns of the eye with neutral ammonium tartrate solution, with no evidence that this was of any value, except possibly in removing deposits of lime (calcific deposits). A careful evaluation of neutral ammonium tartrate solution in treatment of sodium hydroxide burns has been carried out ... on the eyes of rabbits, and has established clearly that this old treatment did not accomplish any better removal of alkali from the tissues than irrigation with water, and did not have any better influence on the clinical course or final results of the injury. |
Solubility Data
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.4304 mL | 27.1518 mL | 54.3036 mL | |
| 5 mM | 1.0861 mL | 5.4304 mL | 10.8607 mL | |
| 10 mM | 0.5430 mL | 2.7152 mL | 5.4304 mL |