Physicochemical Properties
| Molecular Formula | C6H11NOS2 |
| Molecular Weight | 177.28 |
| Exact Mass | 177.028 |
| Elemental Analysis | C, 40.65; H, 6.25; N, 7.90; O, 9.02; S, 36.17 |
| CAS # | 142825-10-3 |
| Related CAS # | 4478-93-7 (racemic); 142825-10-3 (R-isomer); 155320-20-0 (S-isomer); |
| PubChem CID | 9577379 |
| Appearance | Colorless to light yellow liquid |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 368.2±25.0 °C at 760 mmHg |
| Flash Point | 176.5±23.2 °C |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.567 |
| LogP | 0.23 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 10 |
| Complexity | 152 |
| Defined Atom Stereocenter Count | 1 |
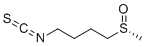
| SMILES | [S@@](C([H])([H])[H])(C([H])([H])C([H])([H])C([H])([H])C([H])([H])N=C=S)=O |
| InChi Key | SUVMJBTUFCVSAD-SNVBAGLBSA-N |
| InChi Code | InChI=1S/C6H11NOS2/c1-10(8)5-3-2-4-7-6-9/h2-5H2,1H3/t10-/m1/s1 |
| Chemical Name | 1-isothiocyanato-4-[(R)-methylsulfinyl]butane |
| Synonyms | (R)-Sulforaphane; L-Sulforaphane; L-Sulforaphane; (R)-sulforaphane; 142825-10-3; (-)-Sulforaphane; (R)-1-Isothiocyanato-4-(methylsulfinyl)butane; 1-isothiocyanato-4-[(R)-methylsulfinyl]butane; 4-Methylsulfinylbutyl isothiocyanate; CHEBI:47808; (-)-Sulforaphane |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Kelch-like ECH-associated protein 1 (Keap1), activating nuclear factor erythroid 2-related factor 2 (Nrf2) [7] - Aryl hydrocarbon receptor (AhR) [5] - Estrogen receptor α (ERα) [5] - AMP-activated protein kinase (AMPK) [7] |
| ln Vitro |
- In HT29 human colon cancer cells, R-sulforaphane induced cell cycle arrest at G2/M phase and apoptosis in a dose-dependent manner. It reduced cyclin B1 and cdc2 expression, increased p21 levels, triggered cytochrome c release from mitochondria, activated caspases-3 and -9, and cleaved poly(ADP-ribose) polymerase (PARP) [3] - In rat H9c2 cells, R-sulforaphane prevented doxorubicin-induced oxidative stress and cell death. It elevated activities of superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), reduced reactive oxygen species (ROS) and malondialdehyde (MDA) levels, upregulated Bcl-2, and downregulated Bax and cleaved caspase-3 [4] - In human breast cell lines (MCF-7, MDA-MB-231, and MCF-10A), R-sulforaphane modulated the expression profile of AhR, ERα, Nrf2, NQO1, and GSTP. It increased Nrf2, NQO1, and GSTP levels, and altered AhR and ERα expression in a cell type-specific manner [5] - In pancreatic cancer cells under high glucose conditions, R-sulforaphane activated Nrf2 via AMPK-dependent signaling, inhibiting cell proliferation, migration, invasion, and promoting apoptosis. This effect was reversed by AMPK inhibition [7] - R-sulforaphane inhibited phase I enzymes (e.g., cytochrome P450 1A1) and induced phase II detoxifying enzymes (e.g., quinone reductase) in various cancer cell lines, contributing to anticarcinogenic activity [6] |
| ln Vivo |
- In rats, R-sulforaphane was a more potent inducer of carcinogen-detoxifying enzyme systems (e.g., quinone reductase, glutathione S-transferase) in liver and lung compared to the S-isomer. It increased enzyme activities and mRNA levels in these tissues [2] - In mice, R-sulforaphane exhibited anticarcinogenic activity by reducing carcinogen-induced preneoplastic lesions in the colon and inducing phase II detoxifying enzymes in liver, colon, and small intestine [6] |
| Enzyme Assay |
- For phase II enzyme activity: Tissue or cell homogenates were prepared and incubated with specific substrates. The activity of quinone reductase was measured by monitoring the reduction of a substrate at a specific wavelength. Glutathione S-transferase activity was assessed by measuring the conjugation of glutathione with a substrate, with absorbance changes recorded over time [2,6] - For antioxidant enzyme activity: Cell lysates were reacted with substrates for SOD, CAT, and GPx. SOD activity was determined by inhibiting the reduction of a tetrazolium salt, CAT by measuring H2O2 decomposition, and GPx by monitoring glutathione oxidation [4] |
| Cell Assay |
- For HT29 colon cancer cells: Cells were treated with R-sulforaphane (0-40 μM) for varying durations. Cell cycle was analyzed by propidium iodide staining and flow cytometry. Apoptosis was assessed via annexin V-FITC/PI staining, TUNEL assay, and caspase activity kits. Western blot measured cyclin B1, cdc2, p21, cytochrome c, caspases, and PARP [3] - For H9c2 cells: Cells were pretreated with R-sulforaphane (0-5 μM) before doxorubicin exposure. ROS and MDA levels were measured using fluorescent probes and colorimetric kits, respectively. Cell viability was determined by MTT assay, and protein expression (Bcl-2, Bax, cleaved caspase-3) was analyzed by western blot [4] - For breast cell lines: Cells were treated with R-sulforaphane, and mRNA and protein levels of AhR, ERα, Nrf2, NQO1, and GSTP were measured by RT-PCR and western blot, respectively [5] - For pancreatic cancer cells: High glucose-cultured cells were treated with R-sulforaphane. Proliferation was assessed by CCK-8 assay, migration/invasion by transwell assays, and apoptosis by flow cytometry. Western blot detected Nrf2, HO-1, NQO1, and p-AMPK, while immunofluorescence analyzed Nrf2 nuclear translocation [7] |
| Animal Protocol |
- In rat studies: Rats were administered R-sulforaphane via oral gavage at specific doses. After treatment, liver and lung tissues were collected. Enzyme activities (quinone reductase, glutathione S-transferase) were measured in tissue homogenates, and mRNA levels were analyzed by RT-PCR [2] - In mouse studies: Mice received R-sulforaphane (200 μmol/kg) via oral gavage. Tissues (liver, colon, small intestine) were harvested to assess phase II enzyme activities. For carcinogenesis models, mice were co-treated with carcinogens and R-sulforaphane, and preneoplastic lesions were counted histologically [6] |
| References |
[1]. Novel gram-scale production of enantiopure R-sulforaphane from Tuscan black kale seeds. Molecules. 2014 May 27;19(6):6975-86. [2]. The natural chemopreventive phytochemical R-sulforaphane is a far more potent inducer of the carcinogen-detoxifying enzyme systems in rat liver and lung than the S-isomer. Int J Cancer. 2011 Jun 15;128(12):2775-82. [3]. Sulforaphane, a naturally occurring isothiocyanate, induces cell cycle arrest and apoptosis in HT29 human colon cancer cells. Cancer Res. 2000;60(5):1426-1433. [4]. Sulforaphane prevents doxorubicin-induced oxidative stress and cell death in rat H9c2 cells. Int J Mol Med. 2015 Jul;36(1):53-64. [5]. R-sulforaphane modulates the expression profile of AhR, ERα, Nrf2, NQO1, and GSTP in human breast cell lines. Mol Cell Biochem. 2021;476(2):525-533. [6]. Anticarcinogenic activities of sulforaphane and structurally related synthetic norbornylisothiocyanates. Proc Natl Acad Sci U S A. 1994 Apr 12;91(8):3147-50. [7]. Activation of Nrf2 by Sulforaphane Inhibits High Glucose-Induced Progression of PancreaticCancer via AMPK Dependent Signaling. ell Physiol Biochem. 2018;50(3):1201-1215. |
| Additional Infomation |
- R-sulforaphane is an enantiomer of sulforaphane, a naturally occurring isothiocyanate in cruciferous vegetables. It is produced from glucoraphanin in Tuscan black kale seeds via enzymatic hydrolysis [1] - Its anticarcinogenic effects involve inducing phase II detoxifying enzymes, inhibiting phase I enzymes, and regulating cell cycle and apoptosis pathways [3,6] - The higher potency of R-sulforaphane compared to the S-isomer in inducing detoxifying enzymes suggests stereoselective interactions with molecular targets [2] (R)-sulforaphane is a sulforaphane in which the sulfinyl group has R configuration. Naturally occurring compound found in brocolli that acts as a potent inducer of phase II detoxification enzymes. It is an enantiomer of a (S)-sulforaphane. Sulforaphane is a naturally-occurring phytochemical belonging to the class of isothiocyanates. As the aglycone metabolite of glucosinolate glucoraphanin (sulforaphane glucosinolate), sulforaphane acts as an antioxidant and potent stimulator of endogenous detoxifying enzymes. This agent displays anticarcinogenic properties due to its ability to induce phase II detoxification enzymes, such as glutathione S-transferase and quinone reductase, thereby providing protection against certain carcinogens and toxic, reactive oxygen species. Broccoli sprouts contain large amounts of sulforaphane, which is also found in other cruciferous vegetables including cabbage and kale. (NCI04) |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~564.05 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (14.10 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (14.10 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (14.10 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.6408 mL | 28.2040 mL | 56.4079 mL | |
| 5 mM | 1.1282 mL | 5.6408 mL | 11.2816 mL | |
| 10 mM | 0.5641 mL | 2.8204 mL | 5.6408 mL |