Physicochemical Properties
| Molecular Formula | C3H7NO3 |
| Molecular Weight | 105.0926 |
| Exact Mass | 105.042 |
| CAS # | 56-45-1 |
| Related CAS # | L-Serine-13C3;201595-68-8;L-Serine-13C;89232-77-9;L-Serine-15N;59935-32-9;L-Serine-d7;935275-35-7;L-Serine-1-13C;81201-84-5;L-Serine-13C3,15N;202407-34-9;L-Serine-d3;105591-10-4;L-Serine-15N,d3;L-Serine-13C3,15N,d3;1994299-33-0;L-Serine-2-13C;89232-76-8;L-Serine1-13C,15N;2483830-04-0;L-Serine-d2;95034-57-4;L-Serine-15N,d3 |
| PubChem CID | 5951 |
| Appearance | White to off-white solid powder |
| Density | 1.6 |
| Boiling Point | 394.8±32.0 °C at 760 mmHg |
| Melting Point | 222ºC |
| Flash Point | 192.6±25.1 °C |
| Vapour Pressure | 0.0±2.1 mmHg at 25°C |
| Index of Refraction | 1.519 |
| LogP | -1.58 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 7 |
| Complexity | 72.6 |
| Defined Atom Stereocenter Count | 1 |
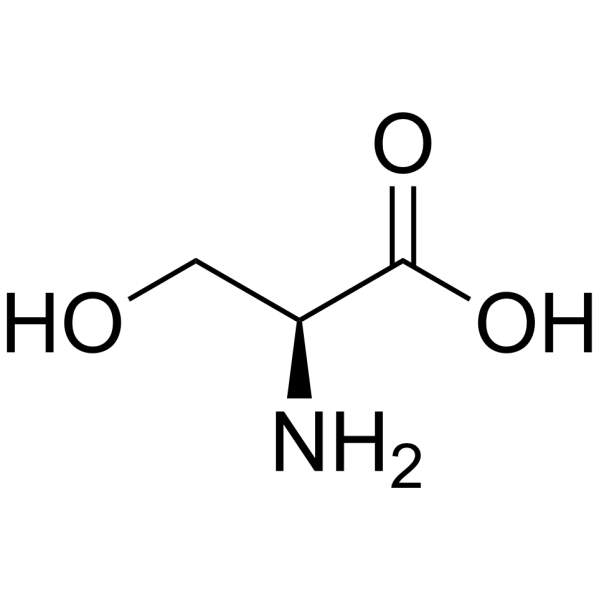
| SMILES | C([C@@H](C(=O)O)N)O |
| InChi Key | MTCFGRXMJLQNBG-REOHCLBHSA-N |
| InChi Code | InChI=1S/C3H7NO3/c4-2(1-5)3(6)7/h2,5H,1,4H2,(H,6,7)/t2-/m0/s1 |
| Chemical Name | (2S)-2-amino-3-hydroxypropanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Biological pathways involving amino acid metabolism, neurotransmitter synthesis, and phospholipid biosynthesis (e.g., conversion to glycine/cysteine, production of phosphatidylserine, synthesis of D-serine) (No IC50/Ki/EC50 data available; acts as an essential substrate for these physiological processes) [1] |
| ln Vitro |
The primary source of one-carbon groups needed for the de novo synthesis of deoxythymidine monophosphate and purine nucleotides is L-serine. Because insufficient amounts of L-serine and its metabolites cannot be produced, they are regarded conditionally essential amino acids in cell culture. L-serine and its metabolites have been shown to be important for both cell proliferation and particular activities of the central nervous system. [1]. - In primary cultures of rat hippocampal neurons, L-Serine (0.1-1 mM) promoted neuronal survival and reduced apoptosis induced by serum deprivation. At 0.5 mM, it increased neuron viability from 45% (serum-deprived group) to 82% (MTT assay) and decreased Annexin V-positive cells from 38% to 12% (flow cytometry). It also upregulated the expression of synaptic protein synaptophysin by 1.8-fold (Western blot) [1] - In rat astrocyte cultures, L-Serine (0.2-2 mM) enhanced phosphatidylserine (PS) biosynthesis. At 1 mM, it increased [³H]-serine incorporation into PS by 2.3-fold (radioisotope labeling assay) and upregulated the activity of serine palmitoyltransferase (a key enzyme in sphingolipid synthesis) by 40% (enzymatic assay) [1] - In human hepatocyte cell lines (HepG2), L-Serine (0.5-5 mM) protected against ethanol-induced oxidative stress. At 2 mM, it reduced intracellular reactive oxygen species (ROS) by 55% (DCFH-DA staining) and increased glutathione (GSH) levels by 1.6-fold (colorimetric assay), thereby inhibiting ethanol-induced hepatocyte necrosis [1] |
| ln Vivo |
- In L-serine-deficient mouse pups (induced by maternal diet lacking L-serine during gestation/lactation), oral supplementation of L-Serine (200 mg/kg/day) from postnatal day 1 to 21 reversed neurodevelopmental abnormalities. It restored brain weight (from 85% of normal to 98%), normalized hippocampal neuron density (from 70% to 96%), and improved motor coordination (rotarod test latency increased from 15 s to 48 s) [1] - In a rat model of hepatic steatosis (induced by high-fat diet), L-Serine (500 mg/kg/day, oral gavage for 8 weeks) reduced liver triglyceride (TG) accumulation by 42% (from 180 mg/g liver to 104 mg/g liver) and decreased serum alanine transaminase (ALT) levels from 125 U/L to 68 U/L. Histological analysis showed reduced hepatic lipid droplets and inflammation [1] - In APP/PS1 transgenic mice (Alzheimer’s disease model), L-Serine (300 mg/kg/day, drinking water for 12 weeks) improved spatial memory (Morris water maze escape latency decreased from 62 s to 28 s) and reduced brain Aβ₄₂ deposition by 35% (immunohistochemistry). It also increased brain D-serine levels by 1.7-fold (HPLC) [1] |
| Enzyme Assay |
- For serine palmitoyltransferase (SPT) activity assay: Rat astrocyte lysates were incubated with L-Serine (0.1-2 mM), palmitoyl-CoA (50 μM), and pyridoxal 5'-phosphate (10 μM) in Tris-HCl buffer (pH 7.4) at 37°C for 60 minutes. The reaction was stopped by adding trichloroacetic acid (TCA), and the product 3-ketodihydrosphingosine was measured by fluorometric assay (excitation 340 nm, emission 450 nm). SPT activity was calculated as nmol product formed per mg protein per hour [1] - For glutathione (GSH) synthesis assay: HepG2 cell lysates were treated with L-Serine (0.5-5 mM) and cysteine (0.2 mM) in phosphate-buffered saline (PBS) at 37°C for 30 minutes. GSH levels were detected by reacting with 5,5'-dithiobis(2-nitrobenzoic acid) (DTNB), and absorbance was measured at 412 nm. GSH concentration was calculated using a standard curve [1] |
| Cell Assay |
- Primary hippocampal neuron survival assay: Hippocampi were dissected from E18 rat embryos, and neurons were isolated by trypsin digestion and plated on poly-L-lysine-coated 96-well plates. After 3 days in culture, medium was replaced with serum-free medium containing L-Serine (0.1-1 mM) for 48 hours. Neuron viability was measured by MTT assay (490 nm absorbance). For apoptosis detection, neurons were stained with Annexin V-FITC/PI and analyzed by flow cytometry [1] - Astrocyte phosphatidylserine (PS) biosynthesis assay: Rat astrocytes were seeded in 6-well plates and labeled with [³H]-serine (1 μCi/mL) in medium containing L-Serine (0.2-2 mM) for 24 hours. Cells were lysed, and lipids were extracted with chloroform/methanol (2:1, v/v). PS was separated by thin-layer chromatography (TLC), and radioactivity was counted by liquid scintillation spectrometry to calculate [³H]-serine incorporation into PS [1] |
| Animal Protocol |
- L-serine-deficient mouse pup supplementation experiment: Female C57BL/6 mice were fed a L-serine-free diet from gestation day 0 to lactation day 21. Pups (n=10 per group) were orally gavaged with L-Serine (200 mg/kg/day, dissolved in 0.9% saline) or saline from postnatal day 1 to 21. On postnatal day 21, pups were euthanized; brain weight was measured, and hippocampal neuron density was analyzed by hematoxylin-eosin staining. Motor coordination was evaluated by rotarod test (5 rpm, 60 s cut-off) on postnatal day 18 [1] - Rat hepatic steatosis treatment experiment: Male Sprague-Dawley rats (250-300 g, n=8 per group) were fed a high-fat diet (45% fat) for 4 weeks to induce steatosis. They were then treated with L-Serine (500 mg/kg/day, dissolved in 0.9% saline) or saline via oral gavage for 8 weeks. Blood samples were collected every 2 weeks to measure serum ALT. At the end of treatment, rats were euthanized; liver TG content was measured by enzymatic assay, and liver histology was examined by Oil Red O staining [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion IN PT AGE 2-9 YR, SERINE PRESENT IN ACID MUCOPOLYSACCHARIDES. EXCESSIVE ACCUMULATION & EXCRETION IN URINE OF MUCOPOLYSACCHARIDES MAY BE RELATED TO ABNORMAL BONDING BETWEEN MUCOPOLYSACCHARIDES & PROTEIN. IN PT AGE 2-9 YR, URINARY SERINE EXCRETION INCR FROM 0.059-0.162 UMOL/24 HR & PLASMA SERINE LEVELS INCR FROM 0.102-0.158 UMOL/ML. IN PT AGE 2-9 YR, SERINE IS PROBABLY NOT ESTERIFIED THROUGH ITS BETA-HYDROXYL GROUP TO ACID MUCOPOLYSACCHARIDES BUT IS LINKED BY CARBOXYL GROUP. DETERMINATION OF SERINE LEVELS IN 13 REGIONS OF THE RAT CEREBRAL CORTEX FAILED TO SHOW ANY MARKED DIFFERENCES IN THE AMINO ACID CONTENTS OF CORTEX AREAS OF DIVERSE FUNCTIONS. - In Sprague-Dawley rats, oral administration of L-Serine (500 mg/kg) showed rapid absorption: Tmax = 1.2 hours, Cmax = 125 μg/mL, and AUC₀₋₈h = 480 μg·h/mL. Oral bioavailability was ~90% (compared to intravenous administration). It distributed widely to tissues, with brain concentration reaching ~35% of plasma concentration at 2 hours post-administration [1] - L-Serine is metabolized primarily in the liver and kidneys: ~60% is converted to glycine via serine hydroxymethyltransferase, and ~25% is used for cysteine synthesis. Elimination half-life (t1/2) is ~1.8 hours, with ~85% of metabolites excreted in urine within 24 hours (mainly as urea and amino acid derivatives) [1] |
| Toxicity/Toxicokinetics |
Toxicity Summary L-Serine plays a role in cell growth and development (cellular proliferation). The conversion of L-serine to glycine by serine hydroxymethyltransferase results in the formation of the one-carbon units necessary for the synthesis of the purine bases, adenine and guanine. These bases when linked to the phosphate ester of pentose sugars are essential components of DNA and RNA and the end products of energy producing metabolic pathways, ATP and GTP. In addition, L-serine conversion to glycine via this same enzyme provides the one-carbon units necessary for production of the pyrimidine nucleotide, deoxythymidine monophosphate, also an essential component of DNA. Interactions INTRACEREBROVENTRICULAR INJECTIONS OF ALANINE (0.5-2.0 UG) INTO RABBITS CAUSED DECR IN BODY TEMP IN A 10-DEG ENVIRONMENT. THE EFFECT WAS SLIGHTLY SUBADDITIVE WITH THAT OF SERINE. ERYTHROBLASTIC LEUKEMIC CELLS INCUBATED IN MEDIA CONTAINING ESSENTIAL AMINO ACID L-SERINE BOUND APPROX 30% MORE INSULIN LABELED WITH (125)IODINE THAN THOSE INCUBATED WITHOUT SERINE. - Acute toxicity: In ICR mice, oral LD50 of L-Serine was >5000 mg/kg; no mortality or toxic symptoms (lethargy, ataxia) were observed at doses up to 5000 mg/kg [1] - Subacute toxicity: Sprague-Dawley rats were orally administered L-Serine (500, 1000, 2000 mg/kg/day) for 4 weeks. No significant changes in body weight, food intake, or organ weights (liver, kidney, brain) were observed. Serum biochemistry (ALT, AST, BUN, creatinine) and hematology parameters (RBC, WBC, platelets) remained within normal ranges. No plasma protein binding data were reported (as L-Serine exists primarily in free form in plasma) [1] |
| References |
[1]. L-Serine in disease and development. Biochem J. 2003 May 1;371(Pt 3):653-61. |
| Additional Infomation |
Pharmacodynamics Serine is classified as a nutritionally non-essential amino acid. Serine is critical for the production of the body's proteins, enzymes and muscle tissue. Serine is needed for the proper metabolism of fats and fatty acids. It also helps in the production of antibodies. Serine is used as a natural moisturizing agent in some cosmetics and skin care products. The main source of essential amino acids is from the diet, non-essential amino acids are normally synthesize by humans and other mammals from common intermediates. - L-Serine is a non-essential amino acid in humans (endogenously synthesized via glycolysis intermediate 3-phosphoglycerate), but becomes essential in conditions of impaired synthesis (e.g., liver disease, genetic disorders) [1] - Its therapeutic potential includes: 1) Neuroprotection (supporting neuronal survival, improving cognitive function in neurodegenerative diseases); 2) Hepatic protection (reducing steatosis and oxidative stress); 3) Supporting fetal/neonatal neurodevelopment (preventing abnormalities in L-serine-deficient states) [1] - L-Serine is a precursor for D-serine (a co-agonist of NMDA receptors), phosphatidylserine (a key membrane phospholipid), and glutathione (an antioxidant), which underlies its multiple biological activities [1] |
Solubility Data
| Solubility (In Vitro) |
H2O : ~50 mg/mL (~475.78 mM) Methanol :< 1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: 100 mg/mL (951.57 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 9.5157 mL | 47.5783 mL | 95.1565 mL | |
| 5 mM | 1.9031 mL | 9.5157 mL | 19.0313 mL | |
| 10 mM | 0.9516 mL | 4.7578 mL | 9.5157 mL |