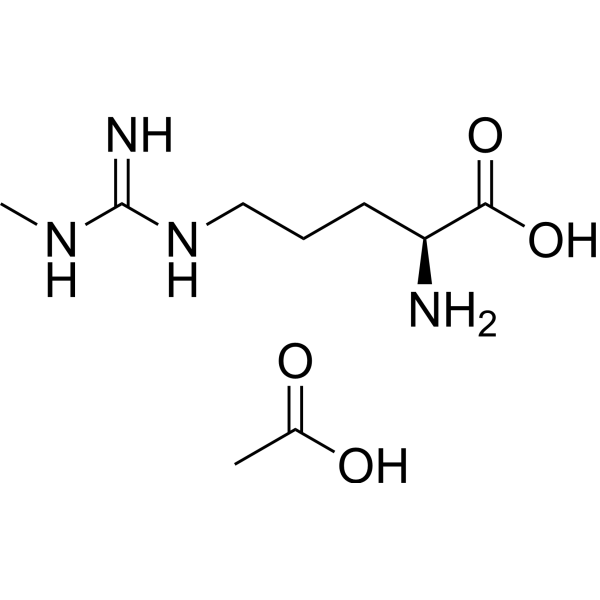
Physicochemical Properties
| Molecular Formula | C9H20N4O4 |
| Molecular Weight | 248.28 |
| Exact Mass | 248.148 |
| CAS # | 53308-83-1 |
| PubChem CID | 135242 |
| Appearance | White to off-white solid powder |
| Boiling Point | 392.7ºC at 760 mmHg |
| Melting Point | 180-190ºC |
| Flash Point | 191.3ºC |
| LogP | 0.595 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 17 |
| Complexity | 224 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | CC(=O)O.CN=C(N)NCCC[C@@H](C(=O)O)N |
| InChi Key | IKPNWIGTWUZCKM-JEDNCBNOSA-N |
| InChi Code | InChI=1S/C7H16N4O2.C2H4O2/c1-10-7(9)11-4-2-3-5(8)6(12)13;1-2(3)4/h5H,2-4,8H2,1H3,(H,12,13)(H3,9,10,11);1H3,(H,3,4)/t5-;/m0./s1 |
| Chemical Name | acetic acid;(2S)-2-amino-5-[(N'-methylcarbamimidoyl)amino]pentanoic acid |
| Synonyms | LNMMA acetate; L NMMA acetate; L-NMMA acetate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | From 100 μM onward, L-NMMA generated concentration-dependent suppression (2Hz) of induced relaxation; the average maximal inhibition at 1 mM was about 35%. When L-arginine (L-Arg) was previously incubated with D-arginine, the inhibitory action of L-NMMA remained unaltered. The relaxation brought on by isoproterenol is unaffected by L-NMMA [2]. L-NMMA infusion decreased arteriolar diameter and increased arteriolar tone in a dose-dependent manner. L-NMMA functions quite instantly. When it comes to the endothelium-dependent vasodilator ACh, but not the endothelium-independent NP, L-NMMA reduces the vasodilatory response. L-NMMA dramatically increases the dose-related vasoconstriction that NE produces [3]. |
| References |
[1]. L-thiocitrulline. A stereospecific, heme-binding inhibitor of nitric-oxide synthases. J Biol Chem. 1994 Oct 21;269(42):26083-91. [2]. Effect of NG-monomethyl L-arginine (L-NMMA) and NG-nitro L-arginine (L-NOARG) on non-adrenergic non-cholinergic relaxation in the circular muscle of the human ileum. Br J Pharmacol. 1991 Aug;103(4):1970-2. [3]. Effect of NG-monomethyl-L-arginine on arcade arterioles of rat spinotrapezius muscles. Am J Physiol. 1991 Jul;261(1 Pt 2):H46-52. |
Solubility Data
| Solubility (In Vitro) | H2O : ≥ 50 mg/mL (~201.39 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 100 mg/mL (402.77 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.0277 mL | 20.1386 mL | 40.2771 mL | |
| 5 mM | 0.8055 mL | 4.0277 mL | 8.0554 mL | |
| 10 mM | 0.4028 mL | 2.0139 mL | 4.0277 mL |