Physicochemical Properties
| Molecular Formula | C31H24O12 |
| Molecular Weight | 588.515069961548 |
| Exact Mass | 588.127 |
| Elemental Analysis | C, 63.27; H, 4.11; O, 32.62 |
| CAS # | 68705-66-8 |
| PubChem CID | 155169 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 3.706 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 43 |
| Complexity | 1020 |
| Defined Atom Stereocenter Count | 4 |
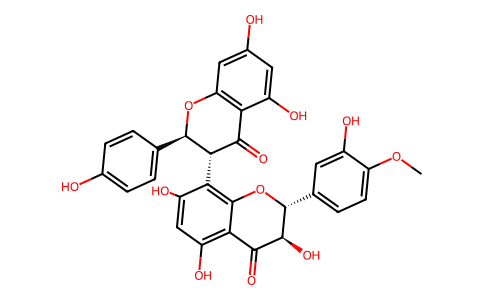
| SMILES | OC1=CC(O)=CC2=C1C(=O)[C@H](C1=C3C(C(=O)[C@H](O)[C@@H](C4=CC=C(OC)C(O)=C4)O3)=C(O)C=C1O)[C@@H](C1=CC=C(O)C=C1)O2 |
| InChi Key | GJWXCPDVDRIBKP-CNTBMXMRSA-N |
| InChi Code | InChI=1S/C31H24O12/c1-41-20-7-4-13(8-16(20)34)30-28(40)27(39)24-19(37)11-18(36)23(31(24)43-30)25-26(38)22-17(35)9-15(33)10-21(22)42-29(25)12-2-5-14(32)6-3-12/h2-11,25,28-30,32-37,40H,1H3/t25-,28-,29+,30+/m0/s1 |
| Chemical Name | (2R,3R)-8-[(2S,3R)-5,7-dihydroxy-2-(4-hydroxyphenyl)-4-oxo-2,3-dihydrochromen-3-yl]-3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-2,3-dihydrochromen-4-one |
| Synonyms | Kolaflavanone; 68705-66-8; (3,8'-Bi-4H-1-benzopyran)-4,4'-dione, 2,2',3,3'-tetrahydro-3',5,5',7,7'-pentahydroxy-2'-(3-hydroxy-4-methoxyphenyl)-2-(4-hydroxyphenyl)-, (2S,2'R,3R,3'R)-; CHEBI:28521; VY4E467596; (2R,3R)-8-[(2S,3R)-5,7-dihydroxy-2-(4-hydroxyphenyl)-4-oxo-2,3-dihydrochromen-3-yl]-3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-2,3-dihydrochromen-4-one; (2R,3R)-8-((2S,3R)-5,7-dihydroxy-2-(4-hydroxyphenyl)-4-oxo-2,3-dihydrochromen-3-yl)-3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-2,3-dihydrochromen-4-one; GlyTouCan:G83156FQ; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Mitotic kinesin Eg5 (IC₅₀ = 2.4 μM, ATPase activity assay) [1] |
| ln Vitro |
Kolaflavanone inhibited proliferation of HeLa, MCF-7, and A549 cancer cell lines with IC₅₀ values of 12.3 μM, 15.7 μM, and 18.2 μM, respectively (MTT assay, 48-hour exposure) [1] Induced G2/M phase arrest in HeLa cells at 15 μM after 24 hours, demonstrated by increased proportion of cells in G2/M phase (25.8% vs. 12.1% in control) via flow cytometry with propidium iodide staining [1] Triggered apoptosis in HeLa cells, evidenced by Annexin V-FITC/PI dual staining showing 34.7% apoptotic cells at 20 μM vs. 5.2% in control after 48 hours [1] Disrupted mitotic spindle formation in HeLa cells, leading to monopolar spindle phenotype (observed in 68% of mitotic cells at 10 μM) via immunofluorescence microscopy using α-tubulin antibodies [1] |
| Enzyme Assay |
Kolaflavanone inhibited proliferation of HeLa, MCF-7, and A549 cancer cell lines with IC₅₀ values of 12.3 μM, 15.7 μM, and 18.2 μM, respectively (MTT assay, 48-hour exposure) [1] Induced G2/M phase arrest in HeLa cells at 15 μM after 24 hours, demonstrated by increased proportion of cells in G2/M phase (25.8% vs. 12.1% in control) via flow cytometry with propidium iodide staining [1] Triggered apoptosis in HeLa cells, evidenced by Annexin V-FITC/PI dual staining showing 34.7% apoptotic cells at 20 μM vs. 5.2% in control after 48 hours [1] Disrupted mitotic spindle formation in HeLa cells, leading to monopolar spindle phenotype (observed in 68% of mitotic cells at 10 μM) via immunofluorescence microscopy using α-tubulin antibodies [1] |
| Cell Assay |
Antiproliferative assay: Cells seeded in 96-well plates (5×10³ cells/well) were treated with gradient concentrations of kolaflavanone for 48 hours. MTT reagent added (0.5 mg/mL), incubated 4 hours, followed by DMSO dissolution. Absorbance measured at 570 nm with reference at 630 nm [1] Cell cycle analysis: HeLa cells treated with kolaflavanone were fixed in 70% ethanol, stained with propidium iodide (50 μg/mL) containing RNase A (100 μg/mL), and analyzed by flow cytometry (488 nm excitation) [1] Apoptosis assay: Cells stained with Annexin V-FITC and propidium iodide in binding buffer for 15 minutes in dark. Fluorescence quantified by flow cytometry (FITC: 488 nm excitation/530 nm emission; PI: 575 nm emission) [1] Immunofluorescence microscopy: HeLa cells grown on coverslips were fixed with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100, blocked with 1% BSA, incubated with α-tubulin primary antibody (1:200) followed by Alexa Fluor 488-conjugated secondary antibody (1:500). DNA counterstained with DAPI [1] |
| References | [1]. Kolaflavanone, a biflavonoid derived from medicinal plant Garcinia, is an inhibitor of mitotic kinesin Eg5. J Biochem. 2021 Dec 28;170(5):611-622. |
| Additional Infomation |
Mechanism: Kolaflavanone binds to the allosteric loop L5 in Eg5's motor domain, inhibiting ATP hydrolysis and microtubule sliding, thereby inducing mitotic arrest [1] Source: Isolated from seeds of Garcinia kola (Clusiaceae family) [1] Structural class: Biflavonoid with molecular formula C₃₁H₂₄O₁₂ [1] Kolaflavanone is a biflavonoid isolated from the seeds of Garcinia kola that has been shown to exhibit hepatoprotective activity. It has a role as a hepatoprotective agent and a plant metabolite. It is a biflavonoid, a ring assembly, a member of dihydroflavonols, a secondary alpha-hydroxy ketone and a member of 4'-methoxyflavanones. Kolaflavanone has been reported in Garcinia eugeniifolia, Cunila, and Garcinia kola with data available. Mitotic kinesin Eg5 remains a validated target in antimitotic therapy because of its essential role in the formation and maintenance of bipolar mitotic spindles. Although numerous Eg5 inhibitors of synthetic origin are known, only a few inhibitors derived from natural products have been reported. In our study, we focused on identifying novel Eg5 inhibitors from medicinal plants, particularly Garcinia species. Herein, we report the inhibitory effect of kolaflavanone (KLF), a Garcinia biflavonoid, on the ATPase and microtubule-gliding activities of mitotic kinesin Eg5. Additionally, we showed the interaction mechanism between Eg5 and KLF via in vitro and in silico analyses. The results revealed that KLF inhibited both the basal and microtubule-activated ATPase activities of Eg5. The inhibitory mechanism is allosteric, without a direct competition with adenosine-5'-diphosphate for the nucleotide-binding site. KLF also suppressed the microtubule gliding of Eg5 in vitro. The Eg5-KLF model obtained from molecular docking showed that the biflavonoid exists within the α2/α3/L5 (α2: Lys111-Glu116 and Ile135-Asp149, α3: Asn206-Thr226; L5: Gly117-Gly134) pocket, with a binding pose comparable to known Eg5 inhibitors. Overall, our data suggest that KLF is a novel allosteric inhibitor of mitotic kinesin Eg5.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6992 mL | 8.4959 mL | 16.9918 mL | |
| 5 mM | 0.3398 mL | 1.6992 mL | 3.3984 mL | |
| 10 mM | 0.1699 mL | 0.8496 mL | 1.6992 mL |