Physicochemical Properties
| Molecular Formula | C16H14O3 |
| Molecular Weight | 258.291704654694 |
| Exact Mass | 258.116 |
| CAS # | 1189508-77-7 |
| Related CAS # | Ketoprofen;22071-15-4 |
| PubChem CID | 3825 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 3.105 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 19 |
| Complexity | 331 |
| Defined Atom Stereocenter Count | 0 |
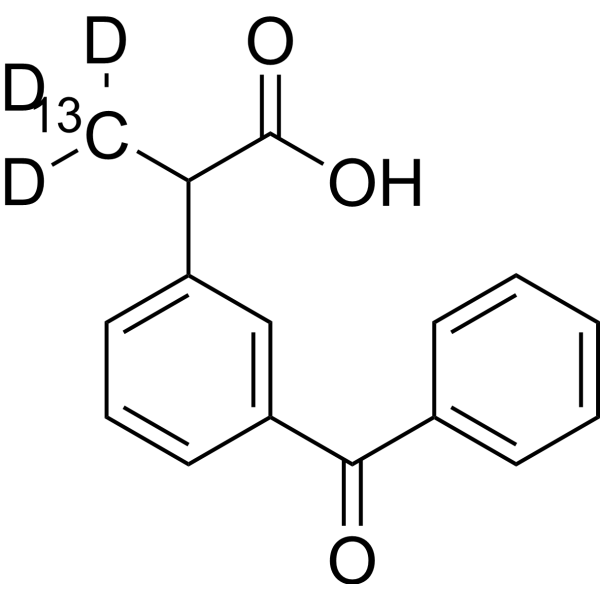
| SMILES | CC(C1=CC(=CC=C1)C(=O)C2=CC=CC=C2)C(=O)O |
| InChi Key | DKYWVDODHFEZIM-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H14O3/c1-11(16(18)19)13-8-5-9-14(10-13)15(17)12-6-3-2-4-7-12/h2-11H,1H3,(H,18,19) |
| Chemical Name | 2-(3-benzoylphenyl)propanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Drug compounds have included stable heavy isotopes of carbon, hydrogen, and other elements, mostly as quantitative tracers while the drugs were being developed. Due to its potential effects on medication pharmacokinetics and metabolic properties, deuteration has drawn interest [1][2]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Ketoprofen is rapidly and well-absorbed orally, with peak plasma levels occurring within 0.5 to 2 hours. In a 24 hour period, approximately 80% of an administered dose of ketoprofen is excreted in the urine, primarily as the glucuronide metabolite. Oral-dose cl=6.9 +/- 0.8 L/h [Ketoprofen Immediate-release capsules (4 × 50 mg)] Oral-dose cl=6.8 +/- 1.8 L/h [Ketoprofen Extended-release capsules (1 × 200 mg)] 0.08 L/kg/h 0.7 L/kg/h [alcoholic cirrhosis patients] Metabolism / Metabolites Rapidly and extensively metabolized in the liver, primarily via conjugation to glucuronic acid. No active metabolites have been identified. Ketoprofen has known human metabolites that include Ketoprofen glucuronide. Biological Half-Life Conventional capsules: 1.1-4 hours Extended release capsules: 5.4 hours due to delayed absorption (intrinsic clearance is same as conventional capsules) |
| Toxicity/Toxicokinetics |
Hepatotoxicity Prospective studies show that 1% to 2% of patients taking ketoprofen experience at least transient serum aminotransferase elevations. These may resolve even with drug continuation. Marked aminotransferase elevations (>3 fold elevated) occur in Likelihood score: C (probable rare cause of clinically apparent liver injury). Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Although ketoprofen has low levels in breastmilk, one center reported that they had received reports of adverse renal and gastrointestinal side effects in breastfed infants whose mothers were taking ketoprofen. Other agents are preferred, especially while nursing a newborn or preterm infant. ◉ Effects in Breastfed Infants All adverse reactions in breastfed infants reported in France between January 1985 and June 2011 were compiled by a French pharmacovigilance center. Of 174 reports, ketoprofen was reported to cause adverse reactions in 8 infants and to be one of the drugs most often suspected in serious adverse reactions, such as esophageal ulceration, erosive gastritis, meningeal hemorrhage, and renal insufficiency. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding 99% bound, primarily to albumin |
| References |
[1]. Impact of Deuterium Substitution on the Pharmacokinetics of Pharmaceuticals. Ann Pharmacother. 2019;53(2):211-223. [2]. Structure-based design of cyclooxygenase-2 selectivity into ketoprofen. Bioorg Med Chem Lett. 2002 Feb 25;12(4):533-7. |
| Additional Infomation |
Ketoprofen is an oxo monocarboxylic acid that consists of propionic acid substituted by a 3-benzoylphenyl group at position 2. It has a role as a non-steroidal anti-inflammatory drug, an antipyretic, an EC 1.14.99.1 (prostaglandin-endoperoxide synthase) inhibitor, an environmental contaminant, a xenobiotic and a drug allergen. It is a member of benzophenones and an oxo monocarboxylic acid. It is functionally related to a propionic acid. Ketoprofen, a propionic acid derivative, is a nonsteroidal anti-inflammatory agent (NSAIA) with analgesic and antipyretic properties. Ketoprofen is a Nonsteroidal Anti-inflammatory Drug. The mechanism of action of ketoprofen is as a Cyclooxygenase Inhibitor. Ketoprofen is a nonsteroidal antiinflammatory drug (NSAID) used in treatment of acute pain and chronic arthritis. Ketoprofen has been linked to a low rate of serum enzyme elevations during therapy and to rare instances of clinically apparent acute liver injury. Ketoprofen has been reported in Homo sapiens with data available. Ketoprofen is a propionic acid derivate and nonsteroidal anti-inflammatory drug (NSAID) with anti-inflammatory, analgesic and antipyretic effects. Ketoprofen inhibits the activity of the enzymes cyclo-oxygenase I and II, resulting in a decreased formation of precursors of prostaglandins and thromboxanes. The resulting decrease in prostaglandin synthesis, by prostaglandin synthase, is responsible for the therapeutic effects of ibuprofen. Ketoprofen also causes a decrease in the formation of thromboxane A2 synthesis, by thromboxane synthase, thereby inhibiting platelet aggregation. An IBUPROFEN-type anti-inflammatory analgesic and antipyretic. It is used in the treatment of rheumatoid arthritis and osteoarthritis. See also: Ketoprofen lysine (is active moiety of); Ketoprofen sodium (is active moiety of); Ketoprofen; Tulathromycin (component of) ... View More ... Drug Indication For symptomatic treatment of acute and chronic rheumatoid arthritis, osteoarthritis, ankylosing spondylitis, primary dysmenorrhea and mild to moderate pain associated with musculotendinous trauma (sprains and strains), postoperative (including dental surgery) or postpartum pain. FDA Label Treatment of musculoskeletal and connective tissue pain Mechanism of Action The anti-inflammatory effects of ketoprofen are believed to be due to inhibition cylooxygenase-2 (COX-2), an enzyme involved in prostaglandin synthesis via the arachidonic acid pathway. This results in decreased levels of prostaglandins that mediate pain, fever and inflammation. Ketoprofen is a non-specific cyclooxygenase inhibitor and inhibition of COX-1 is thought to confer some of its side effects, such as GI upset and ulceration. Ketoprofen is thought to have anti-bradykinin activity, as well as lysosomal membrane-stabilizing action. Antipyretic effects may be due to action on the hypothalamus, resulting in an increased peripheral blood flow, vasodilation, and subsequent heat dissipation. Pharmacodynamics Ketoprofen is a nonsteroidal anti-inflammatory agent (NSAIA) with analgesic and antipyretic properties. Ketoprofen has pharmacologic actions similar to those of other prototypical NSAIDs, which inhibit prostaglandin synthesis. Ketoprofen is used to treat rheumatoid arthritis, osteoarthritis, dysmenorrhea, and alleviate moderate pain. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.8716 mL | 19.3581 mL | 38.7162 mL | |
| 5 mM | 0.7743 mL | 3.8716 mL | 7.7432 mL | |
| 10 mM | 0.3872 mL | 1.9358 mL | 3.8716 mL |