Physicochemical Properties
| Molecular Formula | C30H27N3O6 |
| Molecular Weight | 525.561 |
| Exact Mass | 525.19 |
| Elemental Analysis | C, 68.56; H, 5.18; N, 8.00; O, 18.27 |
| CAS # | 126643-38-7 |
| PubChem CID | 14392217 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.56g/cm3 |
| Index of Refraction | 1.761 |
| LogP | 5.271 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 39 |
| Complexity | 1060 |
| Defined Atom Stereocenter Count | 3 |
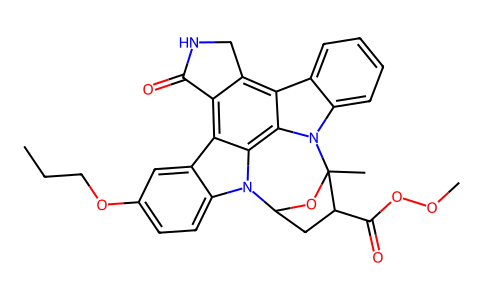
| SMILES | CCCOC1C=CC2=C(C3=C4C(NCC4=C4C5N(C6(OC(N2C3=5)CC6C(OOC)=O)C)C2=CC=CC=C24)=O)C=1 |
| InChi Key | ZZWAFVPZTZSAEM-RIGQTMPJSA-N |
| InChi Code | InChI=1S/C30H27N3O6/c1-4-11-38-15-9-10-19-17(12-15)23-24-18(14-31-27(24)34)22-16-7-5-6-8-20(16)33-26(22)25(23)32(19)21-13-30(36,28(35)37-3)29(33,2)39-21/h5-10,12,21,36H,4,11,13-14H2,1-3H3,(H,31,34)/t21-,29+,30+/m1/s1 |
| Chemical Name | methyl (15S,16R,18R)-16-hydroxy-15-methyl-3-oxo-23-propoxy-28-oxa-4,14,19-triazaoctacyclo[12.11.2.115,18.02,6.07,27.08,13.019,26.020,25]octacosa-1,6,8,10,12,20(25),21,23,26-nonaene-16-carboxylate |
| Synonyms | KT 5926; KT-5926; 9-Hydroxy-9-methoxycarbonyl-8-methyl-14-n-propoxy-2,3,9,10-tetrahydro-8,11-epoxy-1H,8H,11H-2,7b,11a-triazadibenzo(a,g)cycloocta(cde)trinden-1-one; Methyl (15S,16R,18R)-16-hydroxy-15-methyl-3-oxo-23-propoxy-28-oxa-4,14,19-triazaoctacyclo[12.11.2.115,18.02,6.07,27.08,13.019,26.020,25]octacosa-1,6,8,10,12,20(25),21,23,26-nonaene-16-carboxylate; 9-hydroxy-9-methoxycarbonyl-8-methyl-14-n-propoxy-2,3,9,10-tetrahydro-8,11-epoxy-1H,8H,11H-2,7b,11a-triazadibenzo(a,g)cycloocta[cde]trinden-1-one; UNII-1YP81K304O; KT5926 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Myosin light chain kinase |
| ln Vitro | In this study, researchers investigated the antiviral mechanisms of K-252a, a broad non-specific protein kinase inhibitor which was isolated from Nocardiopsis sp. and its derivative (KT5926), against vesicular stomatitis virus (VSV) replication in BHK-21 cells. Although K-252a (5 microM) and KT5926 (15 microM) similarly suppressed the viral primary and secondary transcriptions and genomic RNA synthesis in vivo, the inhibitory mechanisms did not seem to be the same; phosphorylation of the viral NS protein was suppressed by K-252a, which might account for the decreased viral RNA synthesis caused by K-252a. On the other hand, KT5926, being known to preferentially inhibit myosin light chain kinase (MLCK), had little effect on NS protein phosphorylation. Cellular casein kinase II, which is believed to be involved in the phosphorylation of the N-terminal side (domain I) of NS protein, was not inhibited at all by KT5926 even at 15 microM under in vitro assay conditions, and was only weakly inhibited by K-252a at 1 to 10 microM. Neither inhibitor seemed to directly affect viral protein synthesis, but affected it indirectly as a secondary effect of reduced viral RNA synthesis. These results suggest that both the KT5926-sensitive and the KT5926-resistant but K-252a-sensitive functions are involved in the essential processes of viral RNA synthesis. The KT5926-sensitive function(s) might not be involved in the NS protein phosphorylation, but may participate in some other way in the process of virus replication. On the other hand, the KT5926-resistant, K-252a-sensitive function(s) are probably involved in NS protein phosphorylation. The possible nature of those functions is discussed[1]. |
| References | [1]. Studies on the antiviral mechanisms of protein kinase inhibitors K-252a and KT5926 against the replication of vesicular stomatitis virus. Biol Pharm Bull. 1998 May;21(5):498-505. |
| Additional Infomation | KT 5926 is an organic heterooctacyclic compound that is 5-propoxy-1H,1'H-2,2'-biindole in which the nitrogens have undergone formal oxidative coupling to positions 2 and 5 of methyl (3R)-3-hydroxy-2-methyltetrahydrofuran-3-carboxylate (the 2S,3R,5R product), and in which the 3 and 3' positions of the biindole moiety have also undergone formal oxidative coupling to positions 3 and 4 of 1,5-dihydro-2H-pyrrol-2-one. It has a role as an EC 2.7.11.18 (myosin-light-chain kinase) inhibitor. It is a gamma-lactam, an organic heterooctacyclic compound, a methyl ester, a hemiaminal, a tertiary alcohol and an indolocarbazole. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9027 mL | 9.5137 mL | 19.0273 mL | |
| 5 mM | 0.3805 mL | 1.9027 mL | 3.8055 mL | |
| 10 mM | 0.1903 mL | 0.9514 mL | 1.9027 mL |