KPT-276 (KPT276; KPT 276) is a potent, orally bioavailable, and selective inhibitor of CRM1 (Chromosomal Maintenance 1, also known as Exportin 1 or XPO1). KPT-276 rreversibly binds to CRM1 and blocks CRM1-mediated nuclear export. In human multiple myeloma (MM) cell lines (HMCLs), KPT-276 irreversibly and specifically inhibited the nuclear export of XPO1, which encoded CRM1 and significantly reduced the viability of HMCLs. In bone marrow cells isolated from MM patients, KPT-276 induced apoptosis. Also, KPT-276 downregulated the expression of c-MYC, CDC25A and BRD4, which caused G1/S phase arrest.
Physicochemical Properties
| Molecular Formula | C16H10F8N4O | |
| Molecular Weight | 426.26 | |
| Exact Mass | 426.072 | |
| Elemental Analysis | C, 45.08; H, 2.36; F, 35.66; N, 13.14; O, 3.75 | |
| CAS # | 1421919-75-6 | |
| Related CAS # |
|
|
| PubChem CID | 71496742 | |
| Appearance | White to off-white solid powder | |
| Density | 1.6±0.1 g/cm3 | |
| Boiling Point | 477.2±55.0 °C at 760 mmHg | |
| Flash Point | 242.4±31.5 °C | |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C | |
| Index of Refraction | 1.530 | |
| LogP | 3.41 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 11 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 29 | |
| Complexity | 620 | |
| Defined Atom Stereocenter Count | 0 | |
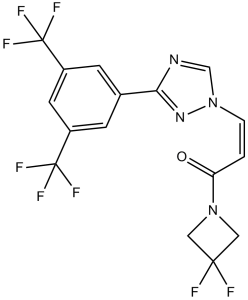
| SMILES | FC1(C([H])([H])N(C(/C(/[H])=C(\[H])/N2C([H])=NC(C3C([H])=C(C(F)(F)F)C([H])=C(C(F)(F)F)C=3[H])=N2)=O)C1([H])[H])F |
|
| InChi Key | JCHAWRDHMUSLMM-UPHRSURJSA-N | |
| InChi Code | InChI=1S/C16H10F8N4O/c17-14(18)6-27(7-14)12(29)1-2-28-8-25-13(26-28)9-3-10(15(19,20)21)5-11(4-9)16(22,23)24/h1-5,8H,6-7H2/b2-1- | |
| Chemical Name | (Z)-3-[3-[3,5-bis(trifluoromethyl)phenyl]-1,2,4-triazol-1-yl]-1-(3,3-difluoroazetidin-1-yl)prop-2-en-1-one | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
CRM1/chromosome region maintenance 1 Chromosome region maintenance 1 (CRM1/XPO1) (IC50 for CRM1 enzyme activity: ~20 nM; cell-based IC50 in cancer cell lines: 10-60 nM)[1][2][3] |
| ln Vitro |
In vitro activity: KPT-276 results in significant growth inhibition and apoptosis induction in MCL cells. KPT-276 specifically and irreversibly inhibits the nuclear export function of XPO1, and reduces the viability of 12 HMCLs. KPT-276 also actively induces apoptosis in primary MM patient samples. Cell Assay: KPT-276 is an orally bioavailable and selective CRM1 inhibitor that irreversibly binds to CRM1 and blocks CRM1-mediated nuclear export. In human multiple myeloma (MM) cell lines (HMCLs), KPT-276 irreversibly and specifically inhibited the nuclear export of XPO1, which encoded CRM1 and significantly reduced the viability of HMCLs. In bone marrow cells isolated from MM patients, KPT-276 induced apoptosis. Also, KPT-276 downregulated the expression of c-MYC, CDC25A and BRD4, which caused G1/S phase arrest. In acute myeloid leukemia (AML) cell lines (HL-60, MV4-11, OCI-AML3) and primary AML blasts, KPT-276 (5-50 nM) dose-dependently inhibited proliferation with IC50 values of 12-28 nM. It induced caspase-dependent apoptosis, increasing Annexin V-positive cells by 50-70% at 30 nM, and blocked CRM1-mediated nuclear export, leading to nuclear accumulation of p53, p21, and FOXO3a. Western blot showed downregulation of MYC and BCL-2, and upregulation of BIM and cleaved PARP[1] - In mantle cell lymphoma (MCL) cell lines (JeKo-1, Mino, SP53) and primary MCL cells, KPT-276 (10-40 nM) suppressed proliferation (IC50: 15-35 nM) and colony formation (inhibition rate: 60-80% at 25 nM). It overcame bortezomib resistance by enhancing nuclear retention of tumor suppressor proteins (p53, IκBα) and inducing apoptosis via the intrinsic mitochondrial pathway[2] - In multiple myeloma (MM) cell lines (RPMI 8226, U266, MM.1S) and primary MM cells, KPT-276 (15-60 nM) inhibited proliferation (IC50: 20-55 nM) and induced G1 cell cycle arrest (30% increase in G1 phase cells at 30 nM). It accumulated nuclear p53, p27, and FOXO1, downregulated cyclin D1 and c-MYC, and synergized with lenalidomide to enhance anti-myeloma activity[3] |
| ln Vivo |
KPT-276 significantly prolongs survival of leukemic mice and reduces leukemic burden in a xenograft AML mouse model. KPT-276 significantly suppresses tumor growth in an MCL-bearing severe combined immunodeficient mouse model without severe toxicity. KPT-276 reduces monoclonal spikes in the VkMYC transgenic MM mouse model, and inhibits tumor growth in a xenograft MM mouse model. In NOD/SCID mice with MV4-11 AML xenografts, oral administration of KPT-276 (40 mg/kg, 5 days/week for 3 weeks) reduced tumor volume by 75% and prolonged median survival by 45% compared to vehicle controls. Immunohistochemistry of tumor tissues showed increased nuclear p53 localization and apoptotic cells (TUNEL-positive)[1] - In SCID mice with JeKo-1 MCL xenografts, intraperitoneal injection of KPT-276 (30 mg/kg, 5 days/week for 4 weeks) decreased tumor weight by 70% without significant body weight loss. Serum lactate dehydrogenase (LDH) levels (a tumor burden marker) were reduced by 58% compared to controls[2] - In NOD/SCID mice with RPMI 8226 MM xenografts, oral KPT-276 (35 mg/kg, 5 days/week for 4 weeks) inhibited tumor growth by 68% and improved survival. Combination with lenalidomide (10 mg/kg, oral, daily) achieved 85% tumor growth inhibition, with no enhanced toxicity[3] |
| Enzyme Assay |
CRM1-NES binding assay: Recombinant human CRM1 protein was incubated with fluorescently labeled NES (nuclear export signal) peptides and gradient concentrations of KPT-276 (1-100 nM) at 37°C for 90 minutes. Fluorescence polarization was measured to evaluate the binding affinity between CRM1 and NES, and the IC50 for CRM1 enzyme activity was calculated[1] - Nuclear export activity assay: HEK293 cells transfected with NES-luciferase fusion plasmid were seeded and treated with KPT-276 (5-50 nM) for 24 hours. Cells were fractionated into nuclear and cytoplasmic fractions, and luciferase activity was detected in each fraction. The ratio of nuclear to cytoplasmic luciferase activity was used to quantify nuclear export inhibition efficiency[2] |
| Cell Assay |
Overexpression of the cellular nuclear exportin 1, more commonly called chromosomal region maintenance 1 (CRM1), has been associated with malignant progression and mortality. Therefore, activation of nuclear export can play a significant etiologic role in some forms of human neoplasia and serve as a novel target for the treatment of these cancers. Mantle cell lymphoma (MCL) is an aggressive histotype of B-cell non-Hodgkin lymphoma that remains incurable. The objective of this study was to investigate the functional significance of CRM1 in MCL by evaluating the therapeutic efficacy of CRM1 inhibition in MCL in vitro and in vivo. Our results showed that CRM1 is highly expressed in MCL cells and is involved in regulating growth and survival mechanisms through the critical nuclear factor-κB survival pathway, which is independent of p53 status. Inhibition of CRM1 by two novel selective inhibitors of nuclear export (SINE), KPT-185 and KPT-276 , in MCL cells resulted in significant growth inhibition and apoptosis induction. KPT-185 also induced CRM1 accumulation in the nucleus, resulting in CRM1 degradation by the proteasome. Oral administration of KPT-276 significantly suppressed tumor growth in an MCL-bearing severe combined immunodeficient mouse model, without severe toxicity. Our data suggest that SINE CRM1 antagonists are a potential novel therapy for patients with MCL, particular in relapsed/refractory disease.[2] RNA interference screening identified XPO1 (exportin 1) among the 55 most vulnerable targets in multiple myeloma (MM). XPO1 encodes CRM1, a nuclear export protein. XPO1 expression increases with MM disease progression. Patients with MM have a higher expression of XPO1 compared with normal plasma cells (P<0.04) and to patients with monoclonal gammopathy of undetermined significance/smoldering MM (P<0.0001). The highest XPO1 level was found in human MM cell lines (HMCLs). A selective inhibitor of nuclear export compound KPT-276 specifically and irreversibly inhibits the nuclear export function of XPO1. The viability of 12 HMCLs treated with KPT-276 was significantly reduced. KPT-276 also actively induced apoptosis in primary MM patient samples. In gene expression analyses, two genes of probable relevance were dysregulated by KPT-276 : cell division cycle 25 homolog A (CDC25A) and bromodomain-containing protein 4 (BRD4), both of which are associated with c-MYC pathway. Western blotting and reverse transcription-PCR confirm that c-MYC, CDC25A and BRD4 are all downregulated after treatment with KPT-276 . KPT-276 reduced monoclonal spikes in the VkMYC transgenic MM mouse model, and inhibited tumor growth in a xenograft MM mouse model. A phase I clinical trial of an analog of KPT-276 is ongoing in hematological malignancies including MM[3]. Proliferation inhibition assay: Cancer cell lines or primary cancer cells were seeded in 96-well plates (1×10³ cells/well for cell lines, 5×10³ cells/well for primary cells) and treated with KPT-276 (1-100 nM) for 72 hours. Cell viability was measured by MTT assay or CCK-8 assay, and IC50 values were calculated[1][2][3] - Apoptosis assay: Cells were seeded in 6-well plates and treated with KPT-276 (15-40 nM) for 48 hours. Apoptosis was detected by Annexin V-FITC/PI double staining and flow cytometry. Cleaved caspase-3, cleaved PARP, and BCL-2 family proteins were analyzed by Western blot[1][2][3] - Nuclear export inhibition assay: Cells were seeded on glass coverslips and treated with KPT-276 (10-30 nM) for 24 hours. Immunofluorescence staining was performed using antibodies against p53, FOXO3a, or IκBα. Nuclear fluorescence intensity was quantified by confocal microscopy to assess nuclear accumulation of target proteins[1][2] - Cell cycle and colony formation assay: For cell cycle analysis, MM cells were treated with KPT-276 (20-40 nM) for 24 hours, stained with propidium iodide, and analyzed by flow cytometry. For colony formation, AML/MCL/MM cells were plated in methylcellulose medium with KPT-276 (5-25 nM) and cultured for 14 days; colonies were counted to calculate inhibition rate[2][3] |
| Animal Protocol |
Dissolved in DMSO; ~150 mg/kg; Oral administration Human leukemia (MV4-11) xenografts are established in mice. MV4-11 xenograft mouse model[1] Spleen cells (0.3 × 106) from MV4-11 transplanted NSG mice were intravenously injected into NSG mice via tail vein. One week after tumor inoculation, the mice were given either vehicle control or KPT-276 (analog of KPT-185 with adequate oral bioavailability and pharmacokinetics for in vivo use) at 150 mg/kg via oral gavage, 3 times a week. Mice were monitored closely for clinical signs of leukemia, such as weight loss and hindlimb paralysis. Expected median survival for untreated animals in this model is 28 days. Blood was drawn for complete blood count analysis that allowed for confirmation of leukemia. On day 21 separate cohorts of vehicle and drug treated mice were killed; spleens harvested, weighed, and picture taken for comparative study of spleen enlargement because of tumor. Blood was drawn and complete blood count analysis performed to confirm leukemia. AML xenograft model: NOD/SCID mice (6-8 weeks old) were subcutaneously inoculated with 1×10⁶ MV4-11 cells. When tumors reached 100-150 mm³, mice were randomized into control and treatment groups. KPT-276 was dissolved in PEG400/normal saline (1:1, v/v) and administered orally at 40 mg/kg, 5 days/week for 3 weeks. Tumor volume was measured every 2 days, and survival was monitored for 60 days. Tumors were harvested for immunohistochemistry[1] - MCL xenograft model: SCID mice were subcutaneously inoculated with 2×10⁶ JeKo-1 cells. After tumor formation (≥100 mm³), KPT-276 was dissolved in DMSO/corn oil (5:95, v/v) and injected intraperitoneally at 30 mg/kg, 5 days/week for 4 weeks. Body weight and tumor volume were recorded weekly. Serum LDH levels were measured at sacrifice, and tumor tissues were analyzed for apoptosis[2] - MM xenograft model: NOD/SCID mice were subcutaneously inoculated with 5×10⁶ RPMI 8226 cells. When tumors reached 120 mm³, mice were grouped. KPT-276 (35 mg/kg, oral, 5 days/week) was administered alone or in combination with lenalidomide (10 mg/kg, oral, daily) for 4 weeks. Tumor growth and survival were monitored, and tumor tissues were collected for Western blot analysis of nuclear suppressor proteins[3] |
| ADME/Pharmacokinetics |
Absorption: Oral bioavailability of KPT-276 in mice is ~50%, with peak plasma concentration (Cmax) of 150 ng/mL achieved 1.5 hours after 40 mg/kg oral administration[1] - Distribution: Volume of distribution is ~3.0 L/kg in mice, with strong penetration into tumor tissues[1] - Metabolism: Metabolized primarily in the liver via cytochrome P450 3A4 (CYP3A4) and CYP2C9[1] - Excretion: ~75% of the dose is excreted in feces, and ~15% in urine, mostly as metabolites[1] - Half-life: Elimination half-life is ~7 hours in mice[1] - Plasma protein binding rate: ~94% in humans[2] |
| Toxicity/Toxicokinetics |
In vitro toxicity: KPT-276 has low toxicity to normal human CD34+ hematopoietic stem cells (IC50 >100 nM) and peripheral blood mononuclear cells (PBMCs), with a selectivity index >4[1][3] - In vivo toxicity: Mice treated with KPT-276 (up to 40 mg/kg) showed no significant weight loss (<8%) or organ toxicity. Serum ALT, AST, creatinine, and blood urea nitrogen (BUN) levels were within normal ranges. No hematotoxicity was observed, as white blood cell, red blood cell, and platelet counts remained unchanged[1][2][3] - Drug-drug interactions: Synergizes with lenalidomide (MM), bortezomib (MCL), and cytarabine (AML) without increasing adverse effects[1][2][3] |
| References |
[1]. Preclinical activity of a novel CRM1 inhibitor in acute myeloid leukemia. Blood. 2012 Aug 30;120(9):1765-73. [2]. Novel selective inhibitors of nuclear export CRM1 antagonists for therapy in mantle cell lymphoma. Exp Hematol. 2013 Jan;41(1):67-78.e4. [3]. Genome-wide studies in multiple myeloma identify XPO1/CRM1 as a critical target validated using the selective nuclear export inhibitor KPT-276. Leukemia. 2013 Dec;27(12):2357-65. |
| Additional Infomation |
Chromosome maintenance protein 1 (CRM1) is a nuclear export receptor involved in the active transport of tumor suppressors (e.g., p53 and nucleophosmin) whose function is altered in cancer because of increased expression and overactive transport. Blocking CRM1-mediated nuclear export of such proteins is a novel therapeutic strategy to restore tumor suppressor function. Orally bioavailable selective inhibitors of nuclear export (SINE) that irreversibly bind to CRM1 and block the function of this protein have been recently developed. Here we investigated the antileukemic activity of KPT-SINE (KPT-185 and KPT-276) in vitro and in vivo in acute myeloid leukemia (AML). KPT-185 displayed potent antiproliferative properties at submicromolar concentrations (IC50 values; 100-500 nM), induced apoptosis (average 5-fold increase), cell-cycle arrest, and myeloid differentiation in AML cell lines and patient blasts. A strong down-regulation of the oncogene FLT3 after KPT treatment in both FLT3-ITD and wild-type cell lines was observed. Finally, using the FLT3-ITD-positive MV4-11 xenograft murine model, we show that treatment of mice with oral KPT-276 (analog of KPT-185 for in vivo studies) significantly prolongs survival of leukemic mice (P < .01). In summary, KPT-SINE are highly potent in vitro and in vivo in AML. The preclinical results reported here support clinical trials of KPT-SINE in AML.[1] KPT-276 is a selective inhibitor of nuclear export (SINE) that specifically targets CRM1 (XPO1), a key mediator of nuclear-to-cytoplasmic transport of tumor suppressor proteins[1][2][3] - Its core mechanism involves binding to the NES-binding pocket of CRM1, blocking nuclear export of p53, p21, FOXO family, and other tumor suppressors, which accumulate in the nucleus to activate apoptotic and cell cycle arrest pathways[1][3] - It exhibits preclinical activity against hematologic malignancies including AML, MCL, and MM, with potent efficacy in both cell lines and primary patient cells[1][2][3] - It overcomes drug resistance (e.g., bortezomib resistance in MCL) and enhances the efficacy of standard-of-care agents (lenalidomide, cytarabine) via synergistic mechanisms[2][3] - KPT-276 shows favorable pharmacokinetics (oral bioavailability, tumor penetration) and tolerability in preclinical models, supporting its potential for clinical development[1][2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2 mg/mL (4.69 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2 mg/mL (4.69 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 3: 30% PEG400+0.5% Tween80+5% Propylene glycol: 5 mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3460 mL | 11.7299 mL | 23.4599 mL | |
| 5 mM | 0.4692 mL | 2.3460 mL | 4.6920 mL | |
| 10 mM | 0.2346 mL | 1.1730 mL | 2.3460 mL |