Physicochemical Properties
| Molecular Formula | C14H7F6N5O |
| Molecular Weight | 375.228702783585 |
| Exact Mass | 375.055 |
| Elemental Analysis | C, 44.81; H, 1.88; F, 30.38; N, 18.66; O, 4.26 |
| CAS # | 1388841-50-6 |
| PubChem CID | 57519758 |
| Appearance | Off-white to yellow solid powder |
| LogP | 3.993 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 26 |
| Complexity | 491 |
| Defined Atom Stereocenter Count | 0 |
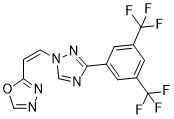
| SMILES | C1=C(C=C(C=C1C(F)(F)F)C(F)(F)F)C2=NN(C=N2)/C=C\C3=NN=CO3 |
| InChi Key | LDFXTRYMMZGKIC-UPHRSURJSA-N |
| InChi Code | InChI=1S/C14H7F6N5O/c15-13(16,17)9-3-8(4-10(5-9)14(18,19)20)12-21-6-25(24-12)2-1-11-23-22-7-26-11/h1-7H/b2-1- |
| Chemical Name | (Z)-2-{2-[3-(3,5-Bis-trifluoromethyl-phenyl)-[1,2,4]triazol-1-yl]-vinyl}-[1,3,4]oxadiazole |
| Synonyms | KPT-251; KPT251; (Z)-2-(2-(3-(3,5-bis(trifluoromethyl)phenyl)-1H-1,2,4-triazol-1-yl)vinyl)-1,3,4-oxadiazole; KPT251; SCHEMBL11318201; KPT251?; LDFXTRYMMZGKIC-UPHRSURJSA-N; KPT 251 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | CRM1/chromosome region maintenance 1 |
| ln Vitro | On the central convex surface of the CRM1 ring, KPT-251 binds in the NES binding groove [1]. KPT-251 (for 72 hours) prevents the growth of melanoma cells [2]. The phosphorylation levels of p53, pRb, survivin, and ERK are regulated by KPT-251 (1 μM; 0-48 hours) [2]. Apoptosis and cell cycle arrest are induced by KPT-251 (0.1 and 1 μM; 0-72 hours) [2]. |
| ln Vivo | Significant survival improvements are obtained by KPT-251 (75 mg/kg/day; i.e., three times per week for five weeks) which successfully suppresses the development of MV4-11 cells transplanted into NSG mice [1]. In a mouse melanoma xenograft model, KPT-251 (50 mg/kg; oral; every other day for 21 days) reduces the formation of tumors [2]. |
| Cell Assay |
Western Blot Analysis[2] Cell Types: Melanoma BRAF WT (Mewo) and mutant cells (A375) Tested Concentrations: 1 μM Incubation Duration: 4, 8, 24 and 48 hrs (hours) Experimental Results: Prevents cytoplasmic p53 degradation, decreases survivin levels, increases ERK phosphorylation pRb and p-pRb levels were diminished in BRAF WT and mutants. Cell cycle analysis[2] Cell Types: Mewo and A375 Cell Tested Concentrations: 1 μM Incubation Duration: 24, 48 and 72 hrs (hours) Experimental Results: Shortening of S phase and G1 and/or G2 cell cycle arrest can be observed. Apoptosis analysis[2] Cell Types: Mel-Juso, SK-MEL-28, SK-MEL-5 and A375 Cell Tested Concentrations: 0.1 and 1 μM Incubation Duration: 24, 48 and 72 hrs (hours) Experimental Results: caspase-3 and - 7 increased activity in the melanoma cell lines tested in a dose- and time-dependent manner. |
| Animal Protocol |
Animal/Disease Models: 7weeks old female NOD-SCID-IL2Rcγnull (NSG) mice, 2 × 106 MV4-11 cells expressing luciferase were introduced through tail vein injection [1] Doses: 75 mg/kg/day Route of Administration: gavage, 3 times weekly for 5 weeks Experimental Results: demonstrated Dramatically increased survival, leukemia progression only after discontinuation of treatment, prevented leukemic cell infiltration into mouse bone marrow and spleen, and protected from normal hematopoiesis Cellular effects. Animal/Disease Models: Athymic nude mouse Nu/Nu, melanoma xenograft model [2] Doses: 50 mg/kg Route of Administration: Orally, once every other day for 21 days Experimental Results: Inhibited tumor growth, increased cleaved caspase-3 and lower Ki67. |
| References |
[1]. Antileukemic activity of nuclear export inhibitors that spare normal hematopoietic cells. Leukemia. 2013 Jan;27(1):66-74. [2]. CRM1 and BRAF inhibition synergize and induce tumor regression in BRAF-mutant melanoma. Mol Cancer Ther. 2013 Jul;12(7):1171-9. |
| Additional Infomation |
Drugs that target the chief mediator of nuclear export, chromosome region maintenance 1 protein (CRM1) have potential as therapeutics for leukemia, but existing CRM1 inhibitors show variable potencies and a broad range of cytotoxic effects. Here, we report the structural analysis and antileukemic activity of a new generation of small-molecule inhibitors of CRM1. Designated selective inhibitors of nuclear export (SINE), these compounds were developed using molecular modeling to screen a small virtual library of compounds against the nuclear export signal (NES) groove of CRM1. The 2.2-Å crystal structure of the CRM1-Ran-RanBP1 complex bound to KPT-251, a representative molecule of this class of inhibitors, shows that the drug occupies part of the groove in CRM1 that is usually occupied by the NES, but penetrates much deeper into the groove and blocks CRM1-directed protein export. SINE inhibitors exhibit potent antileukemic activity, inducing apoptosis at nanomolar concentrations in a panel of 14 human acute myeloid leukemia (AML) cell lines representing different molecular subtypes of the disease. When administered orally to immunodeficient mice engrafted with human AML cells, KPT-251 had potent antileukemic activity with negligible toxicity to normal hematopoietic cells. Thus, KPT-SINE CRM1 antagonists represent a novel class of drugs that warrant further testing in AML patients.[1] Resistance to BRAF inhibitor therapy places priority on developing BRAF inhibitor-based combinations that will overcome de novo resistance and prevent the emergence of acquired mechanisms of resistance. The CRM1 receptor mediates the nuclear export of critical proteins required for melanoma proliferation, survival, and drug resistance. We hypothesize that by inhibiting CRM1-mediated nuclear export, we will alter the function of these proteins resulting in decreased melanoma viability and enhanced BRAF inhibitor antitumoral effects. To test our hypothesis, selective inhibitors of nuclear export (SINE) analogs KPT-185, KPT-251, KPT-276, and KPT-330 were used to induce CRM1 inhibition. Analogs PLX-4720 and PLX-4032 were used as BRAF inhibitors. Compounds were tested in xenograft and in vitro melanoma models. In vitro, we found CRM1 inhibition decreases melanoma cell proliferation independent of BRAF mutation status and synergistically enhances the effects of BRAF inhibition on BRAF-mutant melanoma by promoting cell-cycle arrest and apoptosis. In melanoma xenograft models, CRM1 inhibition reduces tumor growth independent of BRAF or NRAS status and induces complete regression of BRAF V600E tumors when combined with BRAF inhibition. Mechanistic studies show that CRM1 inhibition was associated with p53 stabilization and retinoblastoma protein (pRb) and survivin modulation. Furthermore, we found that BRAF inhibition abrogates extracellular signal-regulated kinase phosphorylation associated with CRM1 inhibition, which may contribute to the synergy of the combination. In conclusion, CRM1 inhibition impairs melanoma survival in both BRAF-mutant and wild-type melanoma. The combination of CRM1 and BRAF inhibition synergizes and induces melanoma regression in BRAF-mutant melanoma.[2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6650 mL | 13.3252 mL | 26.6503 mL | |
| 5 mM | 0.5330 mL | 2.6650 mL | 5.3301 mL | |
| 10 mM | 0.2665 mL | 1.3325 mL | 2.6650 mL |