KN-92 is an inactive derivative of KN-93. KN-92 is intended to be used as a control compound in studies designed to elucidate the antagonist activities of KN-93. KN-93 inhibits histamine-induced aminopyrine uptake in parietal cells (IC50 = 300 nM). KN-93 has been used to implicate roles for CaMKII in Ca2+-induced Ca2+ release in cardiac myocytes, constitutive phosphorylation of 5-lipoxygenase in 3T3 cells, and Ca2+-dependent activation of HIF-1α in colon cancer cell.
Physicochemical Properties
| Molecular Formula | C24H25CLN2O3S | |
| Molecular Weight | 456.98 | |
| Exact Mass | 456.127 | |
| Elemental Analysis | C, 63.08; H, 5.51; Cl, 7.76; N, 6.13; O, 10.50; S, 7.02 | |
| CAS # | 176708-42-2 | |
| Related CAS # | KN-92 phosphate;1135280-28-2;KN-92 hydrochloride;1431698-47-3 | |
| PubChem CID | 5353702 | |
| Appearance | Typically exists as solid at room temperature | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 602.1±65.0 °C at 760 mmHg | |
| Flash Point | 317.9±34.3 °C | |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C | |
| Index of Refraction | 1.630 | |
| LogP | 5.97 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 9 | |
| Heavy Atom Count | 31 | |
| Complexity | 650 | |
| Defined Atom Stereocenter Count | 0 | |
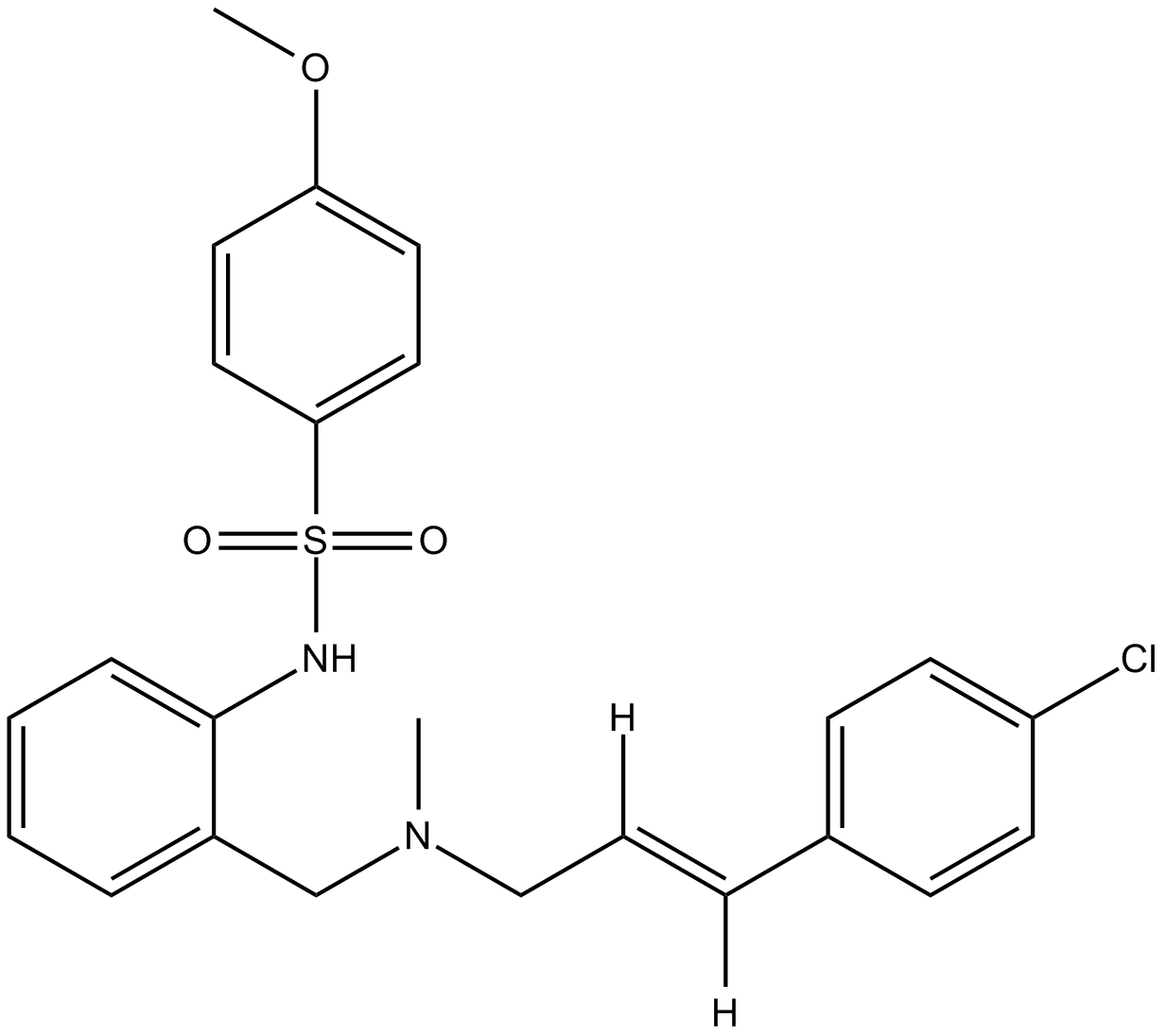
| SMILES | O=S(C1=CC=C(OC)C=C1)(NC2=CC=CC=C2CN(C/C=C/C3=CC=C(Cl)C=C3)C)=O |
|
| InChi Key | RUAOVVIUGUOYHA-AATRIKPKSA-N | |
| InChi Code | InChI=1S/C24H25ClN2O3S/c1-27(17-5-6-19-9-11-21(25)12-10-19)18-20-7-3-4-8-24(20)26-31(28,29)23-15-13-22(30-2)14-16-23/h3-16,26H,17-18H2,1-2H3/b6-5+ | |
| Chemical Name | N-[2-[[[(E)-3-(4-chlorophenyl)prop-2-enyl]-methylamino]methyl]phenyl]-4-methoxybenzenesulfonamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Inactive analog of KN-93; negative control | ||
| ln Vitro | LX-2 cell growth is inhibited by KN-93 (5-50 μM; 24 hours), but KN-92 (5-50 μM; 24 hours) is ineffective in preventing cell growth[2]. KN-93, not KN-92, is shown to decrease p53 and p21 expression when cell cycle regulator expression is analyzed[2]. | ||
| ln Vivo |
|
||
| Cell Assay |
Cell Viability Assay[2] Cell Types: Human hepatic stellate cells (LX-2) Tested Concentrations: 5-50 μM Incubation Duration: 24 hrs (hours) Experimental Results: Ineffective in blocking cell growth. |
||
| Animal Protocol |
|
||
| References |
[1]. Inhibition of the inositol trisphosphate receptor of mouse eggs and A7r5 cells by KN-93 via a mechanism unrelated to Ca2+/calmodulin-dependent protein kinase II antagonism. J Biol Chem. 2002;277(38):35061-35070. [2]. KN-93, a specific inhibitor of CaMKII inhibits human hepatic stellate cell proliferation in vitro. World J Gastroenterol. 2007;13(9):1445-1448. |
||
| Additional Infomation |
KN-93, a Ca(2+)/calmodulin-dependent protein kinase II (CaMKII) inhibitor, concentration-dependently and reversibly inhibited inositol 1,4,5-trisphosphate receptor (IP(3)R)-mediated [Ca(2+)](i) signaling in mouse eggs and permeabilized A7r5 smooth muscle cells, two cell types predominantly expressing type-1 IP(3)R (IP(3)R-1). KN-92, an inactive analog, was ineffective. The inhibitory action of KN-93 on Ca(2+) signaling depended neither on effects on IP(3) metabolism nor on the filling grade of Ca(2+) stores, suggesting a direct action on the IP(3)R. Inhibition was independent of CaMKII, since in identical conditions other CaMKII inhibitors (KN-62, peptide 281-309, and autocamtide-related inhibitory peptide) were ineffective and since CaMKII activation was precluded in permeabilized cells. Moreover, KN-93 was most effective in the absence of Ca(2+). Analysis of Ca(2+) release in A7r5 cells at varying [IP(3)], of IP(3)R-1 degradation in eggs, and of [(3)H]IP(3) binding in Sf9 microsomes all indicated that KN-93 did not affect IP(3) binding. Comparison of the inhibition of Ca(2+) release and of [(3)H]IP(3) binding by KN-93 and calmodulin (CaM), either separately or combined, was compatible with a specific interaction of KN-93 with a CaM-binding site on IP(3)R-1. This was also consistent with the much smaller effect of KN-93 in permeabilized 16HBE14o(-) cells that predominantly express type 3 IP(3)R, which lacks the high affinity CaM-binding site. These findings indicate that KN-93 inhibits IP(3)R-1 directly and may therefore be a useful tool in the study of IP(3)R functional regulation. [1] Aim: To investigate the effects of KN-93, a CaMKII selective inhibitor on cell proliferation and the expression of p53 or p21 protein in human hepatic stellate cells. Methods: Human hepatic stellate cells (LX-2) were incubated with various concentrations (0-50 micromol/L) of KN-93 or its inactive derivative, KN-92. Cell proliferation was measured by CCK-8 assay, and the expression of two cell cycle regulators, p53 and p21, was determined by SDS-PAGE and Western blotting. Results: KN-93 (5-50 micromol/L) decreased the proliferation of human hepatic stellate cells in a dose-dependent manner from 81.76% (81.76% +/- 2.58% vs 96.63% +/- 2.69%, P < 0.05) to 27.15% (27.15% +/- 2.86% vs 96.59% +/- 2.44%, P < 0.01) after 24 h treatment. Incubation of 10 micromol/L KN-93 induced the cell growth reduction in a time-dependent manner from 78.27% at 8 h to 11.48% at 48 h. However, KN-92, an inactive derivative of KN-93, did not inhibit cell proliferation effectively. Moreover, analysis of cell cycle regulator expression revealed that KN-93 rather than KN-92 reduced the expression of p53 and p21. Conclusion: KN-93 has potent inhibitory effect on proliferation of LX-2 cells by modulating the expression of two special cell cycle regulators, p53 and p21. [2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1883 mL | 10.9414 mL | 21.8828 mL | |
| 5 mM | 0.4377 mL | 2.1883 mL | 4.3766 mL | |
| 10 mM | 0.2188 mL | 1.0941 mL | 2.1883 mL |