Physicochemical Properties
| Molecular Formula | C21H22N2O4S |
| Molecular Weight | 398.477 |
| Exact Mass | 398.13 |
| Elemental Analysis | C, 63.30; H, 5.57; N, 7.03; O, 16.06; S, 8.05 |
| CAS # | 309928-48-1 |
| PubChem CID | 2888648 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 578.1±60.0 °C at 760 mmHg |
| Flash Point | 303.4±32.9 °C |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.642 |
| LogP | 4.7 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 28 |
| Complexity | 600 |
| Defined Atom Stereocenter Count | 0 |
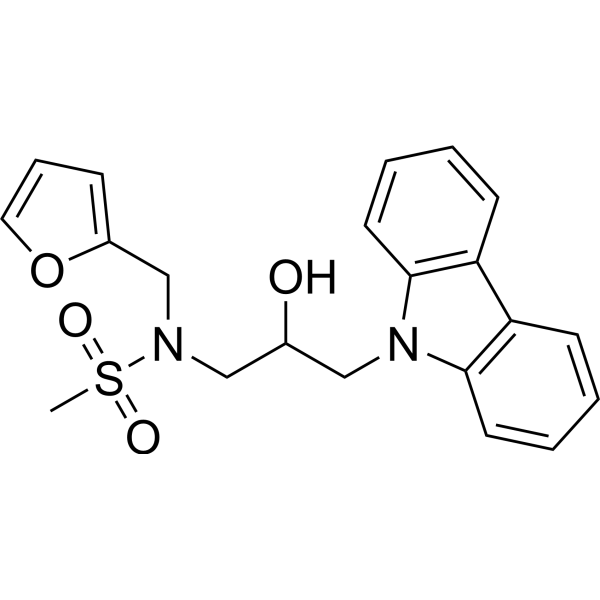
| SMILES | S(C([H])([H])[H])(N(C([H])([H])C1=C([H])C([H])=C([H])O1)C([H])([H])C([H])(C([H])([H])N1C2=C([H])C([H])=C([H])C([H])=C2C2=C([H])C([H])=C([H])C([H])=C12)O[H])(=O)=O |
| InChi Key | OQAFDLPAPSSOHY-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H22N2O4S/c1-28(25,26)22(15-17-7-6-12-27-17)13-16(24)14-23-20-10-4-2-8-18(20)19-9-3-5-11-21(19)23/h2-12,16,24H,13-15H2,1H3 |
| Chemical Name | N-(3-carbazol-9-yl-2-hydroxypropyl)-N-(furan-2-ylmethyl)methanesulfonamide |
| Synonyms | KL 001; KL-001; KL001 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | KL001 (0.03-71 μM) induces a controlled circadian manner, resulting in a longer period and smaller amplitude of circadian rhythms in stable U2OS reporter cell lines expressing Bmal1-dLuc or Per2-dLuc [1). Without altering their basal expression in primary mouse hepatocytes, KL001 (2-8 μM; 18 hours) inhibits the glucagon-dependent induction of Pck1 and G6pc genes [1]. |
| References |
[1]. Identification of small molecule activators of cryptochrome. Science. 2012 Aug 31;337(6098):1094-7. [2]. Circadian molecular clocks and cancer. Cancer Lett. 2014 Jan 1;342(1):9-18. [3]. Crystal structure of mammalian cryptochrome in complex with a small molecule competitor of its ubiquitin ligase. Cell Res. 2013 Dec;23(12):1417-9. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~5 mg/mL (~12.55 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (3.14 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.25 mg/mL (3.14 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 1.25 mg/mL (3.14 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5095 mL | 12.5477 mL | 25.0954 mL | |
| 5 mM | 0.5019 mL | 2.5095 mL | 5.0191 mL | |
| 10 mM | 0.2510 mL | 1.2548 mL | 2.5095 mL |