KIN001-051 (Lck inhibitor; C-8863; KIN-001-051) is a novel, potent, cell-permeable and ATP-competitive inhibitor of Lymphocyte-specific protein tyrosine kinase (LCK, IC50s).
Physicochemical Properties
| Molecular Formula | C23H22N4O |
| Molecular Weight | 370.456 |
| Exact Mass | 370.179 |
| Elemental Analysis | C, 74.57; H, 5.99; N, 15.12; O, 4.32 |
| CAS # | 213743-31-8 |
| Related CAS # | 213743-31-8 |
| PubChem CID | 6603792 |
| Appearance | Off-white to light yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 605.1±55.0 °C at 760 mmHg |
| Flash Point | 319.7±31.5 °C |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.696 |
| LogP | 5.82 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 28 |
| Complexity | 497 |
| Defined Atom Stereocenter Count | 0 |
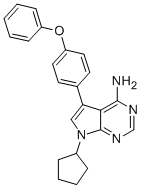
| SMILES | NC1=C2C(N(C3CCCC3)C=C2C4=CC=C(OC5=CC=CC=C5)C=C4)=NC=N1 |
| InChi Key | FMETVQKSDIOGPX-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C23H22N4O/c24-22-21-20(14-27(17-6-4-5-7-17)23(21)26-15-25-22)16-10-12-19(13-11-16)28-18-8-2-1-3-9-18/h1-3,8-15,17H,4-7H2,(H2,24,25,26) |
| Chemical Name | 7-cyclopentyl-5-(4-phenoxyphenyl)pyrrolo[2,3-d]pyrimidin-4-amine |
| Synonyms | KIN001 051; Lck inhibitor; C-8863; KIN001051; C8863; KIN001-051 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Lck (64-509) (IC50 <1 nM); LckCD (IC50 = 2 nM) |
| ln Vitro | Among a variety of tyrosine, non-receptor tyrosine inhibitors, and tyrosine/threonine inhibitors, RK-24466 is selective for Lck. RK-24466 is at least 100 times more effective than PP1 as a powerful drug against CD3-stimulated IL2 generation in Jurkat cells. Good cell selectivity is demonstrated by RK-24466 [1]. RK-24466 dramatically reduces the proliferation of VSMC. RK-24466 significantly reduces the expression of cell nuclear factor (PCNA) and cyclin D1, promotes phosphorylation of blast tumor protein (pRb), and modifies extracellular signal-regulated mitochondria (ERK) inhibitors to inhibit the proliferation and migration of VSMC[2]. |
| ln Vivo | In mice models, RK-24466 was administered intraperitoneally at a low dose, with the goal of post-forceps suppression of T cell injection-stimulated (a-CD3 mAb) IL-2 production (ED50=4 mg/kg). (ED50=25 mg/kg), most likely due to the latter regimen's detrimental malabsorption. Additionally, RK-24466 has the ability to suppress probing T cell immunological responses. A 70% suppression of in vitro IFNγ production was noted after the previous agonistic (KLH) challenge of the drained response following three days of treatment of RK-24466 twice daily (100 mg/kg route) during the in vivo priming phase [3]. Following balloon damage to the carotid artery, RK-24466 inhibits VSMC migration from endothelium-depleted aortic rings and neointima [2]. |
| Cell Assay | CCK-8 is used to measure cellular proliferation after VSMCs are cultured in 10% FBS-supplemented DMEM containing either vehicle (DMSO 2%, v/v) or increasing concentrations of the RK-24466 (1 to 10 μM) for 24 hours in order to investigate the concentration-dependent effect of the RK-24466[2]. |
| Animal Protocol | Rats: In the group receiving treatment with RK-24466, an intravenous injection of 5 μM of RK-24466 is administered via the femoral vein. The rats are put under anesthesia and have their carotid arteries removed at 14 days following BI. The right carotid artery is injured by balloon along its whole length. An intact intra-animal control is provided by the left carotid artery. The intima to media thickness ratio is measured and an H&E stained section is imaged in order to evaluate the formation of neointima[2]. |
| References |
[1]. Pyrrolo[2,3-d]pyrimidines containing an extended 5-substituent as potent and selective inhibitors of lck I. Bioorg Med Chem Lett. 2000 Oct 2;10(19):2167-70. [2]. 7-cyclopentyl-5-(4-phenoxyphenyl)-7H-pyrrolo[2,3-d] pyrimidin-4-ylamine inhibits the proliferation and migration of vascular smooth muscle cells by suppressing ERK and Akt pathways. Eur J Pharmacol. 2017 Mar 5;798:35-42. [3]. Pyrrolo[2,3-d]pyrimidines containing an extended 5-substituent as potent and selective inhibitorsof lck II. Bioorg Med Chem Lett. 2000 Oct 2;10(19):2171-4. |
| Additional Infomation | RK-24466 is a member of the class of pyrrolopyrimidines that is 7H-pyrrolo[2,3-d]pyrimidine substituted by amino, 4-phenoxyphenyl, and cyclopentyl groups at positions 4, 5 and 7, respectively. It is a potent inhibitor of Lck that inhibits Lck (64-509) and LckCD isoforms (IC50 of less than 1 and 2 nM, respectively). It has a role as a geroprotector and an EC 2.7.10.2 (non-specific protein-tyrosine kinase) inhibitor. It is a primary amino compound, a pyrrolopyrimidine, an aromatic amine, an aromatic ether and a member of cyclopentanes. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 37~45 mg/mL (99.9~121.5 mM) Ethanol: ~5 mg/mL (~13.5 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.25 mg/mL (6.07 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 22.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.25 mg/mL (6.07 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 22.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.25 mg/mL (6.07 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 22.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6993 mL | 13.4967 mL | 26.9935 mL | |
| 5 mM | 0.5399 mL | 2.6993 mL | 5.3987 mL | |
| 10 mM | 0.2699 mL | 1.3497 mL | 2.6993 mL |