KI-20227 is a potent and orally bioavailable inhibitor of c-Fms tyrosine kinase (M-CSFR, CSF1R) (IC50 are 2, 12, 217 and 451 nM for c-Fms, VEGFR-2, PDGFRβ and c-Kit respectively). In a model of bone metastasis, Ki20227 inhibits the differentiation of osteoclasts and the destruction of osteolytic bone. Ki20227 slows the advancement of the disease in a mouse model of collagen-induced arthritis. Experimental autoimmune encephalomyelitis is suppressed by Ki20227.
Physicochemical Properties
| Molecular Formula | C24H24N4O5S |
| Molecular Weight | 480.539 |
| Exact Mass | 480.146 |
| Elemental Analysis | C, 59.99; H, 5.03; N, 11.66; O, 16.65; S, 6.67 |
| CAS # | 623142-96-1 |
| Related CAS # | 623142-96-1 |
| PubChem CID | 9869779 |
| Appearance | Off-white to pink solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 621.8±55.0 °C at 760 mmHg |
| Flash Point | 329.8±31.5 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.651 |
| LogP | 4.18 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 34 |
| Complexity | 661 |
| Defined Atom Stereocenter Count | 0 |
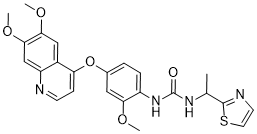
| SMILES | S1C([H])=C([H])N=C1C([H])(C([H])([H])[H])N([H])C(N([H])C1C([H])=C([H])C(=C([H])C=1OC([H])([H])[H])OC1C([H])=C([H])N=C2C([H])=C(C(=C([H])C=12)OC([H])([H])[H])OC([H])([H])[H])=O |
| InChi Key | SHPFDGWALWEPGS-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H24N4O5S/c1-14(23-26-9-10-34-23)27-24(29)28-17-6-5-15(11-20(17)30-2)33-19-7-8-25-18-13-22(32-4)21(31-3)12-16(18)19/h5-14H,1-4H3,(H2,27,28,29) |
| Chemical Name | 1-[4-(6,7-dimethoxyquinolin-4-yl)oxy-2-methoxyphenyl]-3-[1-(1,3-thiazol-2-yl)ethyl]urea |
| Synonyms | KI20227; KI-20227; KI 20227 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | CSF1R (IC50 = 2 nM); VEGFR2 (IC50 = 12 nM); c-Kit (IC50 = 451 nM); PDGFRβ (IC50 = 217 nM) |
| ln Vitro | Ki20227 is a novel quinoline-urea derivative that inhibits the tyrosine kinase of c-Fms. It is discovered that Ki20227 has inhibitory concentrations of 2, 12, 451, and 217 nmol/L for c-Fms, vascular endothelial growth factor receptor-2 (KDR), stem cell factor receptor (c-Kit), and platelet-derived growth factor receptor B, respectively. Moreover, Ki20227 has been shown to suppress M-CSF-dependent M-NFS-60 cell growth in vitro, but not M-CSF-independent A375 human melanoma cell growth. Moreover, Ki20227 dose-dependently suppresses the growth of tartrate-resistant acid phosphatase-positive osteoclast-like cells in an osteoclast-like cell formation assay employing mouse bone marrow cells.[1] |
| ln Vivo | In vivo experiments using nude rats administering oral Ki20227 after intracardiac injection of A375 cells show suppression of osteoclast-like cell accumulation and bone resorption caused by metastatic tumor cells. Furthermore, in rats that have had their ovaries removed, Ki20227 reduces the quantity of osteoclast-like cells that are acid phosphatase-positive and resistant to tartrate on the surfaces of their bones. The results of this study indicate that Ki20227 suppresses osteoclast accumulation in vivo caused by M-CSF, thereby inhibiting osteolytic bone destruction.[1] |
| Cell Assay |
The 96-well culture plate is seeded with M-NFS-60, HUVEC, and A375 cells, which are then cultured for a full day. Following this, culture media are swapped out and left to incubate for an additional 72 hours, either with or without Ki20227 (0.1–3,000 nmol/L). For 12 hours, RAW264.7 cells are serum starved in DMEM with 0.1% FCS. The cells are then treated with serial dilutions of Ki20227 and allowed to incubate for an hour. AVC 264. For four minutes, seven cells are stimulated with 50 ng/mL of recombinant mouse M-CSF. The RAW264 protein contains c-Fms. An ice-cold lysis buffer is used to prepare the 7 cell lysate. |
| Animal Protocol |
Athymic rats (F344/NJcl-rnu), Sprague-Dawley rats 20 mg/kg Oral gavage |
| References |
[1]. Mol Cancer Ther (2006) 5 (11): 2634–2643. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 62.5~96 mg/mL (130.1~199.8 mM) Water: ˂1 mg/mL Ethanol: ~3 mg/mL (~6.2 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.17 mg/mL (4.52 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 21.7 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.17 mg/mL (4.52 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 21.7 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.17 mg/mL (4.52 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 21.7 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0810 mL | 10.4050 mL | 20.8099 mL | |
| 5 mM | 0.4162 mL | 2.0810 mL | 4.1620 mL | |
| 10 mM | 0.2081 mL | 1.0405 mL | 2.0810 mL |