Physicochemical Properties
| Molecular Formula | C26H19N3O5 |
| Molecular Weight | 453.45 |
| Exact Mass | 453.132 |
| Elemental Analysis | C, 68.87; H, 4.22; N, 9.27; O, 17.64 |
| CAS # | 99570-78-2 |
| PubChem CID | 9981344 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.8±0.1 g/cm3 |
| Boiling Point | 769.8±60.0 °C at 760 mmHg |
| Flash Point | 419.4±32.9 °C |
| Vapour Pressure | 0.0±2.8 mmHg at 25°C |
| Index of Refraction | 1.903 |
| LogP | 3.57 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 34 |
| Complexity | 961 |
| Defined Atom Stereocenter Count | 3 |
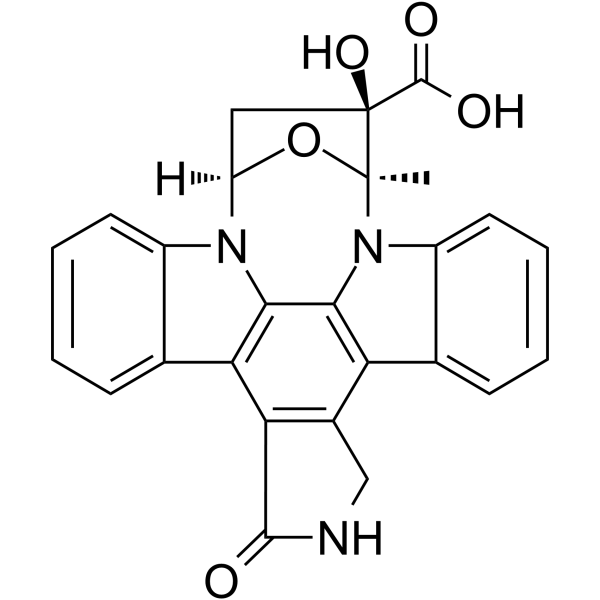
| SMILES | C[C@@]12[C@](C(O)=O)(O)C[C@@H](O1)N3C4=CC=CC=C4C5=C3C6=C(C7=C5C(NC7)=O)C8=CC=CC=C8N62 |
| InChi Key | AMSOPBXQXSAAAC-PLZPTFKGSA-N |
| InChi Code | InChI=1S/C26H19N3O5/c1-25-26(33,24(31)32)10-17(34-25)28-15-8-4-2-6-12(15)19-20-14(11-27-23(20)30)18-13-7-3-5-9-16(13)29(25)22(18)21(19)28/h2-9,17,33H,10-11H2,1H3,(H,27,30)(H,31,32)/t17-,25+,26+/m1/s1 |
| Chemical Name | (15S,16R,18R)-16-hydroxy-15-methyl-3-oxo-28-oxa-4,14,19-triazaoctacyclo[12.11.2.115,18.02,6.07,27.08,13.019,26.020,25]octacosa-1,6,8,10,12,20,22,24,26-nonaene-16-carboxylic acid |
| Synonyms | K-252-b; K 252 b; 9,12-Epoxy-1H-diindolo[1,2,3-fg:3',2',1'-kl]pyrrolo[3,4-i][1,6]benzodiazocine-10-carboxylicacid, 2,3,9,10,11,12-hexahydro-10-hydroxy-9-methyl-1-oxo-, (9S,10R,12R)-; K252b; (15S,16R,18R)-16-Hydroxy-15-methyl-3-oxo-28-oxa-4,14,19-triazaoctacyclo[12.11.2.115,18.02,6.07,27.08,13.019,26.020,25]octacosa-1,6,8,10,12,20,22,24,26-nonaene-16-carboxylic acid; CHEMBL4750633; DTXSID90433626; CHEBI:208814; K252b |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | PKC |
| ln Vitro |
The structures of four new protein kinase C inhibitors of microbial origin, K-252a, K252b, c and d were determined by spectral studies and chemical conversion.[1] These phosphorylation reactions were inhibited by a cell-impermeable ectokinase inhibitor, K252b, and cell surface phosphorylation was also inhibited by exogenously added OPN and BSP substrates, indicating competition for the ectokinase enzyme. However, phosphorylation of OPN and BSP, both of which can mediate cell attachment through Arg-Gly-Asp (RGD) motifs, was not inhibited by an RGD peptide, suggesting that binding of OPN and BSP to cell surface integrins is not required. In similar experiments, ectokinase-mediated phosphorylation of OPN and BSP was demonstrated during mineralized tissue formation by FRCCs in vitro. These studies demonstrate that OPN and BSP secreted by bone cells are phosphorylated by a casein kinase II-like ectokinase present on the surface of osteoblastic cells[3]. |
| ln Vivo | 1. ACh release from motor nerve endings in diaphragms of rats treated chronically with alpha-bungarotoxin (alpha-BuTX) is upregulated at the level of the individual endplate. Involvement of protein kinases in this mechanism of synaptic adaptation was investigated. 2. Miniature endplate potentials (MEPPs) and endplate potentials (EPPs) were recorded after mu-conotoxin treatment, which prevents muscle action potentials. The quantal content at endplates was calculated 'directly', i.e. by dividing the EPP amplitude by the MEPP amplitude. 3. Incubation of muscles from control and alpha-BuTX-treated rats with H-7, a protein kinase C (PKC) inhibitor, reduced MEPP amplitudes but had no clear effect on quantal contents. Polymyxin B, another PKC inhibitor, had a similar effect on muscles from alpha-BuTX-treated rats. 4. Incubation of muscles from alpha-BuTX-treated rats with K252a, a broad-spectrum protein kinase inhibitor of, amongst others, PKC, Ca(2+)-calmodulin-dependent protein kinase II (CaMKII) and neurotrophin receptor tyrosine kinases, resulted in a 30% decrease of the quantal content. However, K252a did not change the quantal content of controls. Incubations with the closely related compound K252b, which has an exclusively extracellular action, had a similar effect. 5. KN62, a specific inhibitor of CaMKII, decreased the mean quantal content of muscles from alpha-BuTX-treated rats by 18%. 6. Tyrphostin 51, a selective tyrosine kinase inhibitor, had no effect on quantal contents of muscles from alpha-BuTX-treated and control rats. However, it increased the frequency and amplitude of MEPPs in muscles from alpha-BuTX-treated rats, leaving those of controls unchanged. 7. The extent of reduction of quantal content, caused by K252a, K252b and KN62, varied between endplates of individual muscles from alpha-BuTX-treated rats; quantal contents at endplates with small MEPPs were more sensitive than those at endplates with large MEPPs. 8. It is concluded that PKC does not play a role in the mechanism of upregulation of ACh release at endplates of alpha-BuTX-treated rats. Instead, CaMKII and some tyrosine kinases in the presynaptic membrane, as well as in the cytoplasm, might be involved[2]. |
| References |
[1]. The structures of the novel protein kinase C inhibitors K-252a, b, c AND d Journal of Antibiotics 39(8), 1072-1078 (1986). [2]. Involvement of protein kinases in the upregulation of acetylcholine release at endplates of α-bungarotoxin-treated rats Journal of Physiology 493, 175-186 (1996). [3]. Evidence of ectokinase-mediated phosphorylation of osteopontin and bone sialoprotein by osteoblasts during bone formation in vitro Biochemistry Journal 323, 637-643 (1997). |
| Additional Infomation |
K-252b is an indolocarbazole. 9,12-Epoxy-1H-diindolo[1,2,3-fg:3',2',1'-kl]pyrrolo[3,4-i][1,6]benzodiazocine-10-carboxylicacid, 2,3,9,10,11,12-hexahydro-10-hydroxy-9-methyl-1-oxo-, (9S,10R,12R)- has been reported in Nocardiopsis with data available. Osteopontin (OPN) and bone sialoprotein (BSP) are phosphorylated glycoproteins that, together with osteonectin/secreted protein, acidic, rich in cysteine (SPARC) and osteocalcin, comprise the major non-collagen proteins of bone. Although phosphorylation of OPN and BSP, which is known to influence the biological properties of these proteins, has been shown to occur intracellularly, recent studies have demonstrated ectokinase activity in bone cell populations [Mikuni-Takagaki, Kakai, Satoyoshi, Kawano, Suzuki, Kawase and Saito (1995) J. Bone Miner. Res. 10, 231-241]. To determine whether OPN and BSP are phosphorylated by ectokinase activity we have used [gamma-32P]ATP and [gamma-32P]GTP as cell-impenetrable phosphate donors to analyse for ectokinase activity in osteoblastic UMR106.06 cells and fetal rat calvarial cells (FRCCs). By pulse-labelling confluent cells with radiolabelled nucleotides, the phosphorylation of endogenous and exogenously added OPN and BSP was demonstrated together with the labelling of a number of cell surface proteins.[3] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2053 mL | 11.0266 mL | 22.0531 mL | |
| 5 mM | 0.4411 mL | 2.2053 mL | 4.4106 mL | |
| 10 mM | 0.2205 mL | 1.1027 mL | 2.2053 mL |