Physicochemical Properties
| Molecular Formula | C19H22CL2N2S |
| Molecular Weight | 381.36 |
| Exact Mass | 380.088 |
| Elemental Analysis | C, 59.84; H, 5.81; Cl, 18.59; N, 7.35; S, 8.41 |
| CAS # | 1071544-43-8 |
| Related CAS # | 500282-33-7;1071544-43-8 (HCl); |
| PubChem CID | 5351674 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 24 |
| Complexity | 380 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | IORPXUDDKDSURS-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C19H21ClN2S.ClH/c1-13-16(9-10-21)17-11-15(23-2)7-8-19(17)22(13)12-14-5-3-4-6-18(14)20;/h3-8,11H,9-10,12,21H2,1-2H3;1H |
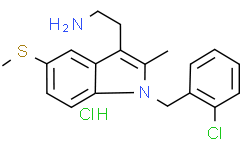
| Chemical Name | 2-[1-[(2-chlorophenyl)methyl]-2-methyl-5-methylsulfanylindol-3-yl]ethylazanium;chloride |
| Synonyms | K103; 1071544-43-8; 2-[1-[(2-chlorophenyl)methyl]-2-methyl-5-methylsulfanylindol-3-yl]ethylazanium;chloride; NSC17383; K103 Hydrochloride; 2PIQ |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | SHIP |
| ln Vitro | K103 showed inhibition of both SHIP paralogs, marking it as a pan-SHIP1/2 inhibitor, but the molecule showed no effect on OCRL, another 5′ inositol phosphatase. In line with the “Two PIP Hypothesis” the molecule demonstrated significant antitumor effects on several cell lines, especially on breast cancer cells. Additional studies on K103 showed that inhibition of SHIP1/2 in multiple myeloma cells resulted in a G2/M cell cycle arrest, followed by caspase cascade activation leading to extensive apoptosis. Tryptamine 1 is compliant with commonly used measures of druglike small molecule properties, but during the course of this work, it was discovered that 1 induced a psychotropic effect in mice which limited the usefulness of the molecule in vivo. Therefore, some synthetic studies on this tryptamine were initiated to define what features of the molecule were needed to maintain pan-SHIP1/2 inhibition so that an inhibitor with good pharmacodynamic properties and an improved side effect profile could be designed. [1] |
| Enzyme Assay |
Malachite Green Phosphatase Release Assays [1] Malachite green assays to monitor phosphatase release were performed with recombinant human truncated SHIP1 (tSHIP1) and SHIP2. Serial dilutions of the compounds dissolved in DMSO were added to the recombinant enzymes diluted in reaction buffer Rx (50 mM Hepes pH 7.4, 150 mM NaCl, 1 mM MgCl2, 0.25 mM EDTA) in triplicate reaction wells in 96-well plates. After incubation for 2 min at room temperature, 2.5 µL of 1 mM phosphatidylinositol 3,4,5-trisphosphate diC8 (PI(3,4,5)P3-diC8) was added to each reaction to a final concentration of 100 µM in a final volume of 25 µL /well. Following a 20 min incubation at 37 °C, 100 µL of Malachite Green solution was added to each well and the plates were incubated at room temperature in the dark for 15 min. Plates were then read at 620 nm on a plate reader. In addition, 3α-Aminocholestane, a known SHIP1 inhibitor, and AS1949490, a known SHIP2 inhibitor, were used as positive controls. Fluorescence Polarization (FP) Assays for the Detection of PI(3,4)P2 [1] The FP Assay for the detection of PI(3,4)P2 was performed using tSHIP1 with serial dilutions of the tryptamines in DMSO according to the manufacturer’s recommendation as previously described. Briefly, serial dilution of the compounds and control with 2.5% solvent only (0 µM) is added to the enzyme in a volume of 40 µL of enzyme-specific reaction buffer (Rx) (S1 Reaction buffer for tSHIP1: 20 mM Tris pH 7.5, 150 mM NaCl, 0.05% Tween 20, 10 mM MgCl2), (S2 reaction buffer for tSHIP1 or SHIP2-Enz: 50 mM Hepes pH 7.4, 150 mM NaCl, 1mM MgCl2, 0.25 mM EDTA). 40 µL of 4 µM PIP3 (PI(3,4,5)P3-diC8) in Rx buffer is added to each reaction and incubated at 37 °C for 20 min, followed by heat inactivation for 3 min at 95 °C. Reactions were spun down briefly and 10 µL of each reaction was added in 6 replicate wells to a black 384-well plate provided with the FP kit. 10 µL of 1X reconstituted Detector (a phosphoinositide binding protein) in PBS is then added to each well, followed by 5 µL of 1X freshly diluted Probe (in PBS, a fluorophore-labeled phosphoinositide) protected from light. Assay plates are gently tapped to mix, the plate is sealed and spun down briefly and the reaction is allowed to equilibrate for 60 min at room temperature protected from light. Control wells are included with each assay (no enzyme control (NE): 5µL Rx buffer with 2.5% solvent + 5 µL PIP3 (4 µM), 10 µL Detector, 5 µL Probe, Probe Alone (PA) control (10 µL enzyme buffer, 10 µL PBS, 5 µL Probe). Fluorescence polarization is then read on a Bio-Tek Synergy 2 plate reader and is expressed in milli Polarization (mP) units. 3α-Aminocholestane, a known SHIP1 inhibitor, and AS1949490, a known SHIP2 inhibitor, were used as positive controls. |
| Cell Assay |
MTT Cell Viability Assay [1] Cells were treated in triplicate or more with increasing concentrations of the compound. Cell viability was determined with a Cell Counting Kit per the manufacturer’s instructions. The odds density (OD) of compound-treated cells was divided by the OD of their vehicle control, and the viability was expressed as a percentage of untreated cells. Results are expressed as mean ± standard error of the mean (SEM) of three individual experiments. HSB2 (ATCC CCL-120.1), K562 (ATCC CCL-243), MBA-MD-231 (ATCC HTB-26), and MCF-7 (ATCC HTB-22) cell lines were obtained from the ATCC, while the 697 Pre-B (ACC 42), NB-4 (ACC 207), and OPM-2 (ACC-50) cell lines were obtained from the DSMZ. Both the ATCC and DSMZ perform genetic validation on deposited cell lines. |
| References | [1]. N1-Benzyl Tryptamine Pan-SHIP1/2 Inhibitors: Synthesis and Preliminary Biological Evaluation as Anti-Tumor Agents. Molecules. 2022 Dec 2;27(23):8451. |
| Additional Infomation |
Inhibition of phosphatidylinositol 3,4,5-trisphosphate 5-phosphatase (SHIP) with small molecule inhibitors leads to apoptosis in tumor cells. Inhibitors that target both SHIP1 and SHIP2 (pan-SHIP1/2 inhibitors) may have benefits in these areas since paralog compensation is not possible when both SHIP paralogs are being inhibited. A series of tryptamine-based pan-SHIP1/2 inhibitors have been synthesized and evaluated for their ability to inhibit the SHIP paralogs. The most active compounds were also evaluated for their effects on cancer cell lines.[1] In summary, an initial evaluation of some tryptamine pan-SHIP1/2 inhibitors has been conducted. The parent tryptamine and some analogous tryptamines have been synthesized and evaluated for activity against both SHIP paralogs in the Malachite Green assay. Several of these analogs showed significant activity against these inositol phosphatase enzymes. The most active analogs all contained chlorinated benzyl groups on the indole N1 nitrogen and a thiomethyl group at the 5-position on the indole. Some alkyl substitution was tolerated on the primary amine of the tryptamine, but disubstitution with alkyl groups or conversion to an amide or sulfonamide led significantly to reduced SHIP inhibition. The most active analogs showed good activity against a number of cancer cell lines. The analog that was best at killing cancer cells was not the most potent SHIP inhibitor, but this may be due to the lipophilicity of the compounds, as the most lipophilic compound was the most potent on cells and SHIP is known to be recruited the cell membrane. Future work will include efforts to further differentiate the structure from the tryptamine scaffold to avoid any psychotropic side effects.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6222 mL | 13.1110 mL | 26.2219 mL | |
| 5 mM | 0.5244 mL | 2.6222 mL | 5.2444 mL | |
| 10 mM | 0.2622 mL | 1.3111 mL | 2.6222 mL |