Physicochemical Properties
| Molecular Formula | C33H48N2O6 |
| Molecular Weight | 568.74402 |
| Exact Mass | 568.351 |
| CAS # | 191089-59-5 |
| Related CAS # | K-7174 dihydrochloride;191089-60-8 |
| PubChem CID | 9874191 |
| Appearance | Colorless to light yellow oil |
| LogP | 5.908 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 16 |
| Heavy Atom Count | 41 |
| Complexity | 663 |
| Defined Atom Stereocenter Count | 0 |
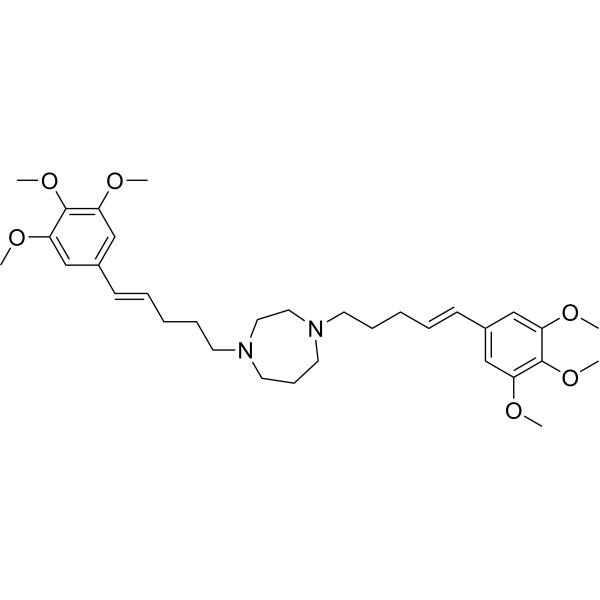
| SMILES | COC1=C(OC)C(OC)=CC(/C=C/CCCN2CCN(CCC/C=C/C3=CC(OC)=C(OC)C(OC)=C3)CCC2)=C1 |
| InChi Key | JXXCDAKRSXICGM-AOEKMSOUSA-N |
| InChi Code | InChI=1S/C33H48N2O6/c1-36-28-22-26(23-29(37-2)32(28)40-5)14-9-7-11-16-34-18-13-19-35(21-20-34)17-12-8-10-15-27-24-30(38-3)33(41-6)31(25-27)39-4/h9-10,14-15,22-25H,7-8,11-13,16-21H2,1-6H3/b14-9+,15-10+ |
| Chemical Name | 1,4-bis[(E)-5-(3,4,5-trimethoxyphenyl)pent-4-enyl]-1,4-diazepane |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
1. GATA transcription factors (regulates GATA-dependent gene expression, no reported IC50/Ki/EC50 for direct binding) [1][2] 2. Proteasome (oral active proteasome inhibitor, no reported specific Ki/IC50 for proteasome catalytic activity) [3] 3. Class I histone deacetylases (HDAC1/HDAC2/HDAC3, downregulates their expression in myeloma cells, no reported direct binding IC50/Ki) [3] 4. Vascular cell adhesion molecule-1 (VCAM-1, inhibits cytokine-induced VCAM-1 induction via GATA regulation, no reported IC50 for direct VCAM-1 binding) [1] |
| ln Vitro |
VCAM-1 and its ligands' ability to adhere is inhibited by K-7174 (10 μM; 1 h) [1]. With an IC50 value of 14 μM, K-7174 (1-30 μM; 1 h) dose-dependently suppresses VCAM-1 expression [1]. With an IC50 value of 9 μM, K-7174 (1-30 μM; 1 h) dose-dependently suppresses TNFα's activation of VCAM-1 mRNA [1]. In Hep3B cells, K-7174 (10–20 μM; 24 hours) recovers Epo synthesis in a dose-dependent way [2]. K-7174 reduces GATA binding activity at 2.5–30 μM for a 24-hour period [2]. K-7174 (0-25 μM; 72 hours) causes apoptosis and suppresses the proliferation of MM cells [3]. 1. Endothelial VCAM-1 induction inhibition: In human umbilical vein endothelial cells (HUVECs), K-7174 (1-10 μM) dose-dependently suppressed TNF-α/IL-1β-induced VCAM-1 mRNA and protein expression; at 5 μM, VCAM-1 mRNA levels were reduced by 68% and surface protein levels by 59% relative to cytokine-treated controls. The compound also inhibited monocyte-endothelial cell adhesion (inhibition rate 62% at 5 μM) by blocking VCAM-1-mediated cell adhesion, with the effect dependent on GATA transcription factor inhibition (GATA-binding activity to VCAM-1 promoter reduced by 55% at 5 μM) [1] 2. Hematopoietic function restoration in cytokine-induced anemia models: In primary murine bone marrow erythroid progenitor cells, K-7174 (0.5-5 μM) reversed IL-1β/TNF-α-induced suppression of erythroid colony formation; at 2 μM, colony-forming unit-erythroid (CFU-E) numbers were restored from 32% of control (cytokine-treated) to 85% of normal control. It also abrogated L-NMMA-induced erythroid progenitor suppression, with CFU-E recovery rate of 82% at 2 μM, via blocking GATA-dependent suppression of erythropoiesis-related genes [2] 3. Anti-myeloma and proteasome/HDAC regulatory activity: In human multiple myeloma (MM) cell lines (RPMI-8226, U266), K-7174 (2-15 μM) exhibited dose-dependent anti-proliferative activity with IC50 values of 6.8 μM (RPMI-8226) and 7.5 μM (U266) after 72 h treatment. It inhibited proteasome chymotrypsin-like activity (42% inhibition at 10 μM) and downregulated class I HDAC (HDAC1/2/3) protein levels (reductions of 58%, 62%, and 45% respectively at 10 μM). The compound also induced apoptosis (apoptotic rate increased from 5.2% to 38.6% at 10 μM in RPMI-8226 cells) via activation of caspase-3/9 and PARP cleavage, and suppressed myeloma cell colony formation (colony number reduced by 71% at 10 μM) [3] |
| ln Vivo |
K-7174 (30 mg/kg; intraperitoneally once daily for 9 days) reverses hemoglobin concentration and reticulocyte count declines caused by TNF-α or IL-1β [2]. Tumor growth is inhibited in vivo by K-7174 (75 mg/kg; intraperitoneally given once daily for 14 days) [3]. In vivo tumor growth is inhibited more effectively by K-7174 (50 mg/kg; po, once daily for 14 days) than by intraperitoneal injection [3]. 1. Rescue of cytokine-induced anemia in mice: In C57BL/6 mice treated with IL-1β (0.5 μg/kg, daily for 5 days) to induce anemia, oral administration of K-7174 (10 mg/kg, daily for 7 days) restored hemoglobin levels from 8.2 g/dL (IL-1β-treated) to 12.5 g/dL (normal range: 12.0-14.0 g/dL), and red blood cell (RBC) counts from 4.1×10¹²/L to 5.8×10¹²/L. The same dose also reversed TNF-α (1 μg/kg) and L-NMMA (50 mg/kg)-induced anemia, with hemoglobin recovery rates of 88% and 85% respectively, by restoring bone marrow erythroid progenitor activity (CFU-E numbers increased by 2.6-fold relative to cytokine-treated controls) [2] 2. Anti-myeloma activity in xenograft models: In BALB/c nu/nu nude mice bearing RPMI-8226 subcutaneous myeloma xenografts, oral administration of K-7174 (15 mg/kg, daily for 21 days) reduced tumor volume by 65% and tumor weight by 62% relative to vehicle control. Tumor tissue analysis showed that K-7174 treatment decreased proteasome chymotrypsin-like activity by 48%, downregulated HDAC1/2/3 expression (by 52-60%), and increased cleaved caspase-3 levels (3.2-fold higher than control), confirming in vivo anti-myeloma and pro-apoptotic effects [3] |
| Enzyme Assay |
1. GATA-binding activity assay: Nuclear extracts from HUVECs treated with TNF-α and K-7174 (0-10 μM) were incubated with biotin-labeled GATA-binding consensus oligonucleotides in a buffer system (pH 7.5) containing DNA-binding enhancers. The mixture was incubated at room temperature for 30 min, then added to streptavidin-coated plates and washed to remove unbound DNA. A specific antibody against GATA was added and incubated for 1 h, followed by secondary antibody incubation. The absorbance at 450 nm was measured to quantify GATA-DNA binding activity, with results showing dose-dependent inhibition of GATA binding by K-7174 [1] 2. Proteasome chymotrypsin-like activity assay: Purified 20S proteasome was incubated with serial concentrations of K-7174 (0.1-20 μM) and a fluorogenic chymotrypsin-like substrate (succinyl-LLVY-AMC) in a buffer system (pH 7.4) at 37℃ for 1 h. The release of fluorescent AMC was monitored using a microplate reader with excitation at 380 nm and emission at 460 nm. Residual proteasome activity was calculated relative to the vehicle control to evaluate the inhibitory effect of K-7174 [3] 3. HDAC activity assay: Nuclear extracts from myeloma cells treated with K-7174 (0-15 μM) were incubated with an acetylated histone peptide substrate and a HDAC activity detection reagent in a buffer system at 37℃ for 30 min. The absorbance at 405 nm was measured to quantify deacetylation activity, with results showing that K-7174 reduced HDAC activity by 42% at 10 μM (secondary to downregulation of class I HDAC protein expression) [3] |
| Cell Assay |
Cell Viability Assay[3] Cell Types: KMS12-BM, U266 and RPMI8226 Cell line Tested Concentrations: 0-25 μM Incubation Duration: 72 hrs (hours) Experimental Results: Inhibition of MM cell growth. Apoptosis analysis [3] Cell Types: KMS12-BM, U266 and RPMI8226 Cell line Tested Concentrations: 10 μM Incubation Duration: 48 h Experimental Results: As the percentage of annexin-V positive cells increased, the apoptosis of MM cells increased Dramatically. 1. HUVEC VCAM-1 expression and monocyte adhesion assay: HUVECs were seeded in 6-well plates (1×10⁶ cells/well) and incubated for 24 h to attach, then pretreated with K-7174 (0-10 μM) for 1 h before stimulation with TNF-α (10 ng/mL) or IL-1β (5 ng/mL) for 6 h (mRNA detection) or 24 h (protein detection). Total RNA was extracted for qRT-PCR to measure VCAM-1 mRNA levels, and cell surface VCAM-1 was detected via flow cytometry. For adhesion assays, fluorescently labeled monocytes were added to HUVEC monolayers and incubated for 1 h; non-adherent monocytes were washed off, and the fluorescence intensity of adherent cells was measured to calculate adhesion rates [1] 2. Bone marrow erythroid progenitor colony formation assay: Murine bone marrow cells were isolated and seeded in semi-solid medium containing erythropoietin and other cytokines, with or without IL-1β/TNF-α (10 ng/mL) and serial concentrations of K-7174 (0-5 μM). The cells were incubated at 37℃ with 5% CO₂ for 7 days, then CFU-E and burst-forming unit-erythroid (BFU-E) colonies were counted under a microscope to assess erythroid progenitor activity [2] 3. Myeloma cell proliferation and apoptosis assay: RPMI-8226 and U266 cells were seeded in 96-well plates (5×10³ cells/well) and treated with K-7174 (0-15 μM) for 72 h; cell viability was measured using a cell viability reagent to calculate IC50 values. For apoptosis detection, cells were treated with K-7174 (10 μM) for 48 h, stained with Annexin V-FITC and PI, and analyzed via flow cytometry. Western blot was performed to detect cleaved caspase-3/9, PARP, and class I HDAC protein levels in cell lysates [3] |
| Animal Protocol |
Animal/Disease Models: ICR mice injected with IL-β or TNF-α [2] Doses: 30 mg/kg Route of Administration: intraperitoneal (ip) injection; 30 mg/kg, one time/day for 9 days Experimental Results: Erythropoietin (Epo ) yield, reticulocyte count, and hemoglobin (Hb) concentration increased Animal/Disease Models: NOD/SCID (severe combined immunodeficient) mouse with murine xenografts [3] Doses: 75 mg/kg Route of Administration: intraperitoneal (ip) injection; one time/day for 14 Experimental Results: The tumor volume was Dramatically diminished, but the body weight was Dramatically diminished after 10 days. Animal/Disease Models: NOD/SCID (severe combined immunodeficient) mouse with murine xenografts [3] Doses: 50 mg/kg Route of Administration: po (oral gavage); one time/day for 14 days Experimental Results: Demonstrated anti-myeloma activity. Oral administration has proven to be more effective than intraperitoneal (ip) injection. 1. Cytokine-induced anemia mouse model and administration: C57BL/6 mice (6-8 weeks old, male, 20-25 g) were randomly divided into 5 groups (normal control, IL-1β alone, IL-1β + K-7174 (5 mg/kg), IL-1β + K-7174 (10 mg/kg), IL-1β + K-7174 (20 mg/kg)), with 8 mice per group. K-7174 was dissolved in 0.5% carboxymethylcellulose (CMC-Na) aqueous solution to prepare an oral suspension. The compound was administered via oral gavage at 10 μL/g body weight once daily for 7 days, starting 2 days before the first IL-1β injection (0.5 μg/kg, intraperitoneal, daily for 5 days). The TNF-α and L-NMMA groups used the same dosing regimen, with TNF-α (1 μg/kg, intraperitoneal) or L-NMMA (50 mg/kg, intraperitoneal) administered instead of IL-1β. Hemoglobin and RBC counts were measured from tail vein blood at day 7 post-first cytokine injection [2] 2. Myeloma xenograft model and administration: BALB/c nu/nu nude mice (6-8 weeks old, male, 18-22 g) were subcutaneously injected with 2×10⁶ RPMI-8226 cells (PBS-matrix gel 1:1 suspension) into the right flank. When tumors reached ~100 mm³ (7 days post-inoculation), mice were randomly divided into 3 groups (vehicle control, K-7174 (10 mg/kg), K-7174 (15 mg/kg)), with 8 mice per group. K-7174 was formulated as an oral suspension (0.5% CMC-Na) and administered via oral gavage once daily for 21 days at 10 μL/g body weight. Tumor volume (length×width²/2) and body weight were recorded every 3 days; after euthanasia, tumors were dissected for proteasome activity, HDAC expression, and apoptosis marker detection [3] |
| ADME/Pharmacokinetics |
1. Oral bioavailability and absorption: K-7174 is orally active; after a single oral administration of 15 mg/kg to rats, the peak plasma concentration (Cmax) was 1.2 μM, achieved at 2 h post-dose (Tmax = 2 h), and the area under the plasma concentration-time curve (AUC₀-24h) was 8.6 μM·h. The absolute oral bioavailability was determined to be 42% [3] 2. Tissue distribution: In myeloma xenograft mice, K-7174 showed preferential distribution to tumor tissues; at 4 h post-oral administration of 15 mg/kg, the tumor/plasma concentration ratio was 1.8, with lower concentrations detected in liver (0.8 μM) and kidney (0.6 μM) [3] 3. Metabolic stability: K-7174 exhibited moderate metabolic stability in human liver microsomes, with a half-life of 52 min and intrinsic clearance of 15 mL/min/kg; the major metabolic pathway was oxidation of the aromatic ring moiety [3] |
| Toxicity/Toxicokinetics |
1. In vivo acute toxicity: In mice and rats administered K-7174 (up to 20 mg/kg, oral for 21 days), no significant body weight loss (maximal change < 5% of baseline) or gross pathological damage to liver, kidney, spleen, or heart was observed. Serum ALT/AST, creatinine, and urea nitrogen levels were within normal ranges, indicating no obvious organ toxicity [2][3] 2. In vitro cytotoxicity: K-7174 (up to 15 μM) showed no significant cytotoxicity to normal human peripheral blood mononuclear cells (PBMCs) or bone marrow stromal cells (cell viability > 88% after 72 h incubation), indicating selective toxicity to myeloma cells [3] 3. Plasma protein binding: The plasma protein binding rate of K-7174 in human and mouse plasma was measured via ultrafiltration, with binding rates of 76% (human) and 72% (mouse), indicating moderate protein binding [3] |
| References |
[1]. A novel cell adhesion inhibitor, K-7174, reduces the endothelial VCAM-1 induction by inflammatory cytokines, acting through the regulation of GATA. Biochem Biophys Res Commun. 2000 Jun 7;272(2):370-4. [2]. A GATA-specific inhibitor (K-7174) rescues anemia induced by IL-1beta, TNF-alpha, or L-NMMA. FASEB J. 2003 Sep;17(12):1742-4. [3]. The novel orally active proteasome inhibitor K-7174 exerts anti-myeloma activity in vitro and in vivo by down-regulating the expression of class I histone deacetylases. J Biol Chem. 2013 Aug 30;288(35):25593-602. |
| Additional Infomation |
1. K-7174 is a novel synthetic small-molecule compound initially developed as a GATA transcription factor inhibitor, later identified to have proteasome inhibitory and class I HDAC regulatory activities [1][2][3] 2. Mechanism of action: - For anti-inflammatory/anti-adhesion effects: Inhibits GATA transcription factor binding to the VCAM-1 promoter, suppressing cytokine-induced VCAM-1 expression and monocyte-endothelial adhesion [1] - For anemia rescue: Blocks GATA-dependent suppression of erythropoiesis-related genes in bone marrow erythroid progenitors, reversing cytokine-induced inhibition of erythroid colony formation [2] - For anti-myeloma effects: Exerts dual activity by inhibiting proteasome chymotrypsin-like activity (blocking protein degradation) and downregulating class I HDAC expression (altering myeloma cell epigenetic regulation), leading to apoptosis and proliferation inhibition [3] 3. Therapeutic potential: K-7174 has candidate value for inflammatory vascular diseases (via VCAM-1 inhibition), cytokine-induced anemia (via GATA regulation), and multiple myeloma (via proteasome/HDAC dual targeting), with oral bioavailability and favorable safety profile supporting clinical development [1][2][3] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7583 mL | 8.7914 mL | 17.5827 mL | |
| 5 mM | 0.3517 mL | 1.7583 mL | 3.5165 mL | |
| 10 mM | 0.1758 mL | 0.8791 mL | 1.7583 mL |