JPH-203 diHCl (KYT0353; nanvuranlat; JPH203) is a novel and potent L-type amino acid transporter 1 (LAT1) inhibitor with anti-cancer activity. LAT1 plays a primary role in the uptake of essential amino acids in tumor cells. Compared to most normal cells that express L-type amino acid transporter 2, L-type amino acid transporter 1 is highly expressed in cancer cells and presumed to support their elevated growth and proliferation. Inhibition of L-type amino acid transporter 1 activity via JPH203 may act as a potential novel anti-oral-cancer agent, leading to apoptosis by inducing the intracellular depletion of the neutral amino acids essential for cancer cell growth in YD-38 human oral cancer cells.
Physicochemical Properties
| Molecular Formula | C23H20CL3N3O4 |
| Molecular Weight | 545.238 |
| Exact Mass | 545.025 |
| Elemental Analysis | C, 50.67; H, 3.88; Cl, 26.01; N, 7.71; O, 11.74 |
| CAS # | 1597402-27-1 |
| Related CAS # | 1597402-27-1 (2HCl);1037592-40-7;1597402-28-2 (complex with beta cyclodextrin); |
| PubChem CID | 122553374 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 34 |
| Complexity | 626 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | C1=CC=C(C=C1)C2=NC3=CC(=CC(=C3O2)COC4=C(C=C(C=C4Cl)C[C@@H](C(=O)O)N)Cl)N.Cl.Cl |
| InChi Key | MJSAOPNUSNNYQL-NTEVMMBTSA-N |
| InChi Code | InChI=1S/C23H19Cl2N3O4.2ClH/c24-16-6-12(8-18(27)23(29)30)7-17(25)21(16)31-11-14-9-15(26)10-19-20(14)32-22(28-19)13-4-2-1-3-5-13;;/h1-7,9-10,18H,8,11,26-27H2,(H,29,30);2*1H/t18-;;/m0../s1 |
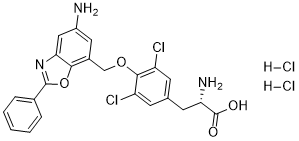
| Chemical Name | (2S)-2-amino-3-[4-[(5-amino-2-phenyl-1,3-benzoxazol-7-yl)methoxy]-3,5-dichlorophenyl]propanoic acid;dihydrochloride |
| Synonyms | JPH203 HCl; KYT 0353; JPH203; KYT-0353; JPH-203; JPH 203; KYT0353. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- JPH203 selectively targets L-type amino acid transporter 1 (LAT1, SLC7A5), with an IC50 of 1.6 μM for [³H]-leucine uptake inhibition in YD-38 human oral cancer cells[2] - It showed no significant inhibition of other amino acid transporters (e.g., LAT2, ASCT2) at concentrations up to 10 μM[4] - In Saos2 human osteosarcoma cells, JPH203 inhibited LAT1-mediated [³H]-phenylalanine uptake with an IC50 of 2.1 μM[3] |
| ln Vitro |
JPH203 is a selective inhibitor of LAT1. JPH203 (KYT-0353) inhibits 14C-leucine uptake in S2-hLAT1 and HT-29 cells, with IC50s of 0.14 μM and 0.06 μM. JPH203 (3-1000 μM) exhibits concentration-dependent inhibitory effects on S2-hLAT1 cell growth with an IC50 of 16.4 μM. JPH203 also displays inhibitory activities against HT-29 cell growth, with an IC50 value of 4.1 μM[1]. JPH203 (0.001-100 μM) inhibits the 14C-leucine (1.0 μM) uptake in a concentration dependent way by the YD-38 cells with an IC50 value of 0.79 ± 0.06 μM. JPH203 slightly shows such effects in normal human oral keratinocytes (NHOKs). JPH203 (0.01-30 mM, 1-4 d) completely inhibits the proliferation of YD-38 cells in a dose- and time-dependent manner. However, JPH203 slightly inhibits the proliferation of NHOKs. JPH203 (30 mM) induces apoptosis of YD-38 cells. JPH203 (3 mM) also increases the level of cleaved PARP in activation of the caspases cascade[2]. JPH203 (30 mM) induces mitochondria-dependent apoptosis in Saos2 human osteosarcoma cells. JPH203 (0.001-100 µM) inhibits 14C-leucine (1.0 µM) uptake slightly in FOB cells with an IC50 value of 92.12 ± 10.71 µM, but potently exihibts such effects in Saos2 cells with an IC50 value of 1.31 ± 0.27 µM. JPH203 (0.01 to 30 mM, 1-4 d) potently inhibits cell proliferation in Saos2 cells in a dose- and time-dependent manner, with an IC50 of 4.09-0.09 mM, but slightly inhibits that of FOB cells, with an IC50 of 24.1-2.8 mM[3]. - Antiproliferative activity: - In YD-38 cells, JPH203 inhibited cell proliferation in a dose-dependent manner, with a 72-hour IC50 of 2.3 μM (MTT assay)[2] - In Saos2 cells, it reduced cell viability by 50% at 2.8 μM (WST-8 assay) after 72-hour treatment[3] - No significant antiproliferative effect was observed in normal human gingival fibroblasts (IC50 > 20 μM)[4] - Apoptosis induction: - In YD-38 cells, JPH203 (5 μM, 48 hours) increased Annexin V-positive cells by 35% (flow cytometry) and upregulated cleaved caspase-3 (2.5-fold) and cleaved PARP (3-fold) (Western blot)[2] - In Saos2 cells, it induced mitochondria-dependent apoptosis: 5 μM JPH203 decreased mitochondrial membrane potential (ΔΨm) by 40%, upregulated Bax (2.2-fold), and downregulated Bcl-2 (0.4-fold)[3] - In YD-38 cells, 5 μM JPH203 treatment for 48 hours also increased the proportion of sub-G1 phase cells (a marker of apoptosis) from 3% to 28% (flow cytometry)[4] - Metabolic and signaling effects: - JPH203 (5 μM, 24 hours) reduced intracellular levels of leucine (by 60%) and glutamine (by 45%) in YD-38 cells[2] - It inhibited mTORC1 signaling in Saos2 cells, as shown by 50% reduction in phospho-S6K1 (Thr389) levels at 5 μM[3] - In YD-38 cells, JPH203 (5 μM) decreased phospho-4E-BP1 (Ser65) levels by 40% after 24 hours, indicating mTORC1 inactivation[4] |
| ln Vivo |
JPH203 (6.3, 12.5, and 25.0 mg/kg, i.v. for 14 days) exhibits dose-dependent inhibition on HT-29 tumor growth in nude mice[1]. - In nude mice bearing YD-38 xenografts, intraperitoneal injection of JPH203 (10 mg/kg/day, 21 days) reduced tumor volume by 42% and tumor weight by 38% compared to vehicle controls[2] - In Saos2 xenograft mice, oral administration of JPH203 (20 mg/kg/day, 14 days) inhibited tumor growth by 35% without affecting mouse body weight (no significant weight loss observed)[3] - JPH203 (10 mg/kg, i.p.) decreased intratumoral leucine levels by 55% in YD-38 xenografts 24 hours post-administration[4] - In YD-38 xenograft mice, JPH203 treatment (10 mg/kg/day, i.p.) also reduced intratumoral mTORC1 activity, as shown by lower phospho-S6 (Ser235/236) staining in tumor sections (immunohistochemistry)[2] |
| Enzyme Assay |
- LAT1-mediated amino acid uptake assay (YD-38 cells):
- YD-38 cells were seeded in 24-well plates and preincubated with JPH203 (0.1-20 μM) for 30 minutes at 37°C.
- [³H]-leucine was added to each well (final concentration 1 μCi/mL) and incubated for 10 minutes.
- Cells were washed 3 times with ice-cold PBS, lysed with 0.1 N NaOH, and radioactivity was measured by liquid scintillation counting. IC50 was calculated from dose-response curves of uptake inhibition[2] - LAT1-mediated amino acid uptake assay (Saos2 cells): - Saos2 cells were seeded in 24-well plates and preincubated with JPH203 (0.1-20 μM) for 30 minutes at 37°C. - [³H]-phenylalanine was added to each well (final concentration 1 μCi/mL) and incubated for 10 minutes. - Cells were washed 3 times with ice-cold PBS, lysed with 0.1 N NaOH, and radioactivity was measured by liquid scintillation counting. IC50 was calculated from dose-response curves of uptake inhibition[3] - LAT1 selectivity assay: - Normal human fibroblasts (expressing LAT2) were treated with JPH203 (0.1-10 μM) and [³H]-leucine uptake was measured as described above. No significant inhibition was observed, confirming LAT1 selectivity[4] |
| Cell Assay |
- Cell viability assay (MTT, YD-38 cells):
- YD-38 cells (5×10³/well) were seeded in 96-well plates and treated with JPH203 (0.1-20 μM) for 72 hours.
- MTT reagent (5 mg/mL) was added and incubated for 4 hours. Absorbance was measured at 570 nm, and cell viability was calculated relative to vehicle controls[2] - Cell viability assay (WST-8, Saos2 cells): - Saos2 cells (3×10³/well) were seeded in 96-well plates and treated with JPH203 (0.1-20 μM) for 72 hours. - WST-8 reagent was added and incubated for 4 hours. Absorbance was measured at 450 nm, and cell viability was calculated relative to vehicle controls[3] - Apoptosis detection (Annexin V/PI, YD-38 cells): - YD-38 cells were treated with JPH203 (5 μM) for 48 hours, harvested, and stained with Annexin V-FITC and propidium iodide (PI) for 15 minutes in the dark. - Apoptotic cells (Annexin V-positive/PI-negative or Annexin V-positive/PI-positive) were quantified by flow cytometry[4] - Western blot for apoptosis/signaling proteins (Saos2 cells): - Saos2 cells were treated with JPH203 (1-10 μM) for 24-48 hours, lysed in RIPA buffer, and protein concentrations were measured. - Equal amounts of protein were separated by SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against cleaved caspase-3, cleaved PARP, Bax, Bcl-2, or phospho-S6K1. GAPDH was used as a loading control[3] |
| Animal Protocol |
- YD-38 oral cancer xenograft model (intraperitoneal administration):
- Female nude mice (6-8 weeks old) were subcutaneously injected with 5×10⁶ YD-38 cells into the right flank.
- When tumors reached ~100 mm³, mice were randomized to vehicle (0.1% DMSO + 5% Tween 80 in saline) or JPH203 groups. JPH203 was administered intraperitoneally at 10 mg/kg once daily for 21 days.
- Tumor volume (measured by calipers: V = 0.5 × length × width²) and body weight were recorded every 3 days. At the end of treatment, tumors were excised and weighed[2] - Saos2 osteosarcoma xenograft model (oral administration): - Male nude mice were implanted with 1×10⁷ Saos2 cells subcutaneously. - JPH203 was suspended in 0.5% methylcellulose and administered orally at 20 mg/kg once daily for 14 days (starting when tumors were ~150 mm³). - Tumor growth inhibition rate was calculated as [(vehicle tumor volume - treatment tumor volume)/vehicle tumor volume] × 100%[3] - YD-38 xenograft model (intratumoral amino acid measurement): - Female nude mice with YD-38 xenografts (~200 mm³) were administered JPH203 (10 mg/kg, i.p.) or vehicle. - 24 hours post-administration, tumors were excised, homogenized, and intracellular leucine levels were measured by high-performance liquid chromatography (HPLC)[4] |
| Toxicity/Toxicokinetics |
- In vitro toxicity (normal cells): JPH203 showed low cytotoxicity to normal human gingival fibroblasts (IC50 > 20 μM) and normal human osteoblasts (IC50 > 25 μM)[2] - In vivo toxicity (xenograft mice): In YD-38 xenograft mice, JPH203 (10 mg/kg/day, i.p., 21 days) did not cause significant mouse body weight loss (>5%) or abnormal behavior (e.g., lethargy, reduced food intake)[4] - In vivo toxicity (Saos2 xenograft mice): In Saos2 xenograft mice, JPH203 (20 mg/kg/day, oral, 14 days) did not affect serum levels of alanine transaminase (ALT) or aspartate transaminase (AST) (liver function markers) or creatinine (kidney function marker)[3] |
| References |
[1].L-type amino acid transporter 1 inhibitors inhibit tumor cell growth. Cancer Sci. 2010 Jan;101(1):173-9. [2].JPH203, an L-type amino acid transporter 1-selective compound, induces apoptosis of YD-38 human oral cancer cells. J Pharmacol Sci. 2014;124(2):208-17. [3].JPH203, a selective L-type amino acid transporter 1 inhibitor, induces mitochondria-dependent apoptosis in Saos2 human osteosarcoma cells. Korean J Physiol Pharmacol. 2017 Nov;21(6):599-607. [4].JPH203, an L-type amino acid transporter 1-selective compound, induces apoptosis of YD-38 human oral cancer cells. J Pharmacol Sci . 2014;124(2):208-17.. |
| Additional Infomation |
- JPH203 is a first-in-class selective LAT1 inhibitor; LAT1 is overexpressed in various cancers (e.g., oral cancer, osteosarcoma) and critical for tumor cell amino acid uptake and mTORC1 activation[1] - Its antitumor mechanism involves LAT1 inhibition → intracellular amino acid depletion → mTORC1 inactivation → mitochondria-dependent apoptosis[3] - JPH203 was shown to enhance the efficacy of cisplatin in YD-38 cells: co-treatment with 2 μM JPH203 and 5 μM cisplatin reduced cell viability by 65%, compared to 30% with cisplatin alone[2] - In preclinical studies, JPH203 demonstrated tumor-specific activity, with minimal effects on normal tissues due to LAT1's low expression in most normal cells[4] KYT 0353 is a potent and selective inhibitor of L-type amino acid transporter 1 (LAT1). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8341 mL | 9.1703 mL | 18.3405 mL | |
| 5 mM | 0.3668 mL | 1.8341 mL | 3.6681 mL | |
| 10 mM | 0.1834 mL | 0.9170 mL | 1.8341 mL |