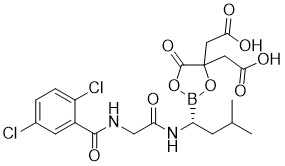
Ixazomib citrate (MLN9708; MLN-9708; MLN 9708), the citrate salt and orally bioavailable prodrug of Ixazomib (MMLN2238; MMLN-2238), is a selective inhibitor of 20S proteasome (IC50 = 3.4 nM) with potential anticancer activity. It is being studied in various countries as a treatment for lymphoma, amyloidosis, and multiple myeloma in a Phase II trial. The boron-containing compound ixazomib citrate needs to be hydrolyzed to yield the pharmacologically active compound MLN2238 (ixazomib). Preclinical studies have shown that MLN9708 has better antitumor activity, pharmacokinetics, and pharmacodynamics than bortezomib.
Physicochemical Properties
| Molecular Formula | C₂₀H₂₃BCL₂N₂O₉ |
| Molecular Weight | 517.12 |
| Exact Mass | 516.087 |
| Elemental Analysis | C, 46.45; H, 4.48; B, 2.09; Cl, 13.71; N, 5.42; O, 27.85 |
| CAS # | 1239908-20-3 |
| Related CAS # | Ixazomib;1072833-77-2 |
| PubChem CID | 56844015 |
| Appearance | White to off-white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Index of Refraction | 1.580 |
| LogP | 3.378 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 34 |
| Complexity | 797 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | ClC1C([H])=C([H])C(=C([H])C=1C(N([H])C([H])([H])C(N([H])[C@]([H])(B1OC(C(C([H])([H])C(=O)O[H])(C([H])([H])C(=O)O[H])O1)=O)C([H])([H])C([H])(C([H])([H])[H])C([H])([H])[H])=O)=O)Cl |
| InChi Key | MBOMYENWWXQSNW-AWEZNQCLSA-N |
| InChi Code | InChI=1S/C20H23BCl2N2O9/c1-10(2)5-14(21-33-19(32)20(34-21,7-16(27)28)8-17(29)30)25-15(26)9-24-18(31)12-6-11(22)3-4-13(12)23/h3-4,6,10,14H,5,7-9H2,1-2H3,(H,24,31)(H,25,26)(H,27,28)(H,29,30)/t14-/m0/s1 |
| Chemical Name | 2-[4-(carboxymethyl)-2-[(1R)-1-[[2-[(2,5-dichlorobenzoyl)amino]acetyl]amino]-3-methylbutyl]-5-oxo-1,3,2-dioxaborolan-4-yl]acetic acid |
| Synonyms | Ninlaro; MLN9708; MLN 9708; MLN-9708; ixazomib citrate; MMLN 2238-prodrug; MMLN-2238-prodrug; MMLN2238-prodrug; Ixazomib-prodrug |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
20S proteasome β5 (IC50 = 3.4 nM); 20S proteasome β1 (IC50 = 31 nM); 20S proteasome β2 (IC50 = 3500 nM) Ixazomib citrate (MLN9708) is a proteasome inhibitor. Its biologically active form, MLN2238, is generated upon hydrolysis in aqueous solutions or plasma. The primary target is the proteasome complex within the ubiquitin-proteasome system (UPS). [1] Ixazomib citrate (MLN9708/MLN2238) is a proteasome inhibitor. The active form, MLN2238, predominantly inhibits the chymotrypsin-like (CT-L) activity of the proteasome with an IC50 of 5 nM in MM.1S cells [2]. It also inhibits caspase-like (C-L) and trypsin-like (T-L) proteasome activities at higher concentrations [2]. |
| ln Vitro |
Ixazomib citrate (MLN9708; 0.20-3.20 μM) effectively suppresses the growth of both cell lines in a time- and dose-dependent manner. Cell cycle arrest is induced in MG-63 and Saos-2 cells by ixazomib. Ixazomib requires the activation of both caspase8 and caspase9 in order to induce apoptosis primarily through the caspases pathway. Treatment with ixazomib raises pro-apoptotic protein levels and decreases anti-apoptotic protein levels, which govern MOMP. Treatment with ixazomib causes the release of Cytc, Smac, and OMI from mitochondria and lowers XIAP protein levels. Ixazomib reduces MMP2/9 expression and secretion levels and inhibits the ability of MG-63 and Saos-2 cells to invade[1]. Ixazomib citrate (MLN9708; 12 nM) exhibits inhibitory action against the activities of the T-L and C-L proteasomes. Ixazomib treatment of H929 and MM.1S MM cells results in a notable increase in poly(ADP) ribose polymerase (PARP) proteolytic cleavage, a hallmark event during apoptosis. The upstream PARP activator caspase-3 is cleaved by isxazomib. Ixazomib increases the levels of CHOP/GADD153 and Bip protein as well as eIf2-α kinase activity. Ixazomib targets NF-κB, inhibits capillary tubule formation in vitro, and blocks MM cell proliferation induced by BMSCs[2]. MLN2238 inhibited the growth of human osteosarcoma (OS) cell lines MG-63 and Saos-2 in a time- and dose-dependent manner, as assessed by MTT assay. IC50 values for MG-63 and Saos-2 cells were determined at specific treatment times (e.g., for the 24h treatment, IC50 was 0.40 µM for MG-63 and 0.80 µM for Saos-2). MLN2238 induced significant apoptosis in MG-63 and Saos-2 cells, evidenced by increased Annexin V+/PI- and Annexin V+/PI+ populations in flow cytometry and the cleavage of caspase-3, caspase-8, caspase-9, and PARP in Western blot analysis. MLN2238 caused cell cycle arrest at S and G2/M phases in both OS cell lines, associated with upregulation of P21, p-Chk1 (Ser345), WEE1, p-CDK1 (Tyr15), E2F1, and APAF-1, and downregulation of BCL-2 and Rb (in MG-63). MLN2238 treatment promoted mitochondrial outer membrane permeabilization (MOMP), indicated by increased BAX/BCL-2 ratio, activation of BID, decreased p-BAD, and release of cytochrome c, Smac, and OMI from mitochondria to cytosol, along with decreased XIAP levels. MLN2238 attenuated the invasion ability of MG-63 and Saos-2 cells in a dose-dependent manner in a Transwell invasion assay, concomitant with decreased expression and secretion levels of MMP2 and MMP9. [1] MLN2238 treatment of multiple myeloma (MM) cell lines (MM.1S, INA-6, RPMI-8226, MM.1R, H929, OPM1, OPM2) resulted in a significant, concentration-dependent decrease in cell viability after 48 hours, as assessed by MTT assay [2]. MLN2238 induced apoptosis in MM cell lines (MM.1S, H929, OPM1, OPM2), evidenced by a significant increase in Annexin V+/PI+ apoptotic cell populations [2]. MLN2238 decreased the viability of purified CD138+ tumor cells from MM patients, including those refractory to bortezomib, lenalidomide, and dexamethasone [2]. MLN2238 overcame bortezomib resistance in isogenic cell lines (ANBL-6.WT vs. ANBL-6.BR), with a significantly lower IC50 resistance ratio compared to bortezomib [2]. MLN2238 at its IC50 for MM cells did not significantly affect the viability of normal peripheral blood mononuclear cells (PBMCs) from healthy donors [2]. MLN2238 induced the accumulation of ubiquitinated proteins in a time- and dose-dependent manner [2]. Mechanistic studies showed that MLN2238-induced apoptosis is associated with cleavage/activation of caspase-3, caspase-8, caspase-9, and PARP [2]; upregulation of p53, p21, Noxa, PUMA, and E2F [2]; downregulation of pRb, cyclin D1, and Cdk6 [2]; induction of endoplasmic reticulum (ER) stress proteins Bip, phospho-eIF2-α, and CHOP [2]; and inhibition of constitutive and TNF-α-induced NF-κB activation [2]. MLN2238 inhibited bone marrow stromal cell (BMSC)-induced proliferation of MM.1S cells [2]. MLN2238 (10 nM) inhibited in vitro capillary-like tube formation by human umbilical vein endothelial cells (HUVECs) without significant cytotoxicity, indicating anti-angiogenic activity [2]. Combination treatment of MLN2238 with lenalidomide, the HDAC inhibitor SAHA, or dexamethasone showed synergistic anti-MM activity in MM.1S cells, as determined by isobologram analysis (Combination Index <1.0) [2]. |
| ln Vivo |
Ixazomib citrate (MLN9708; 11 mg/kg) enhances survival in the human plasmacytoma MM.1S xenograft mouse model by substantially preventing the growth of MM tumors. Hemoglobin, bilirubin, and creatinine levels in the blood chemistry profiles of mice treated with ixazomib are normal. Cloned-caspase-3 positive cells in the xenograft model are significantly elevated by ixazomib[2]. In a human plasmacytoma xenograft model (MM.1S cells implanted in CB-17 SCID mice), intravenous administration of MLN2238 (11 mg/kg, twice weekly for 3 weeks) significantly inhibited tumor growth and prolonged survival compared to vehicle-treated controls [2]. Mice treated with MLN2238 survived longer than those treated with bortezomib (1 mg/kg) [2]. Oral administration of MLN2238 (8 mg/kg, twice weekly for 3 weeks) also significantly inhibited tumor growth and prolonged survival [2]. Immunohistochemical analysis of tumors from MLN2238-treated mice showed increased cleaved caspase-3 and TUNEL-positive cells (indicating apoptosis) [2], decreased Ki-67 staining (indicating reduced proliferation) [2], and decreased expression of angiogenesis markers VEGFR2 and Pecam [2]. Blood chemistry analysis of MLN2238-treated mice showed normal levels of creatinine, hemoglobin, and bilirubin, indicating good tolerability [2]. |
| Enzyme Assay |
Proteasome Activity Assay: MM.1S cell lysates were prepared using RIPA buffer [2]. Protein samples (20 µg) were incubated in a reaction buffer (20 mmol/L Hepes, pH 7.6, 0.5 mmol/L EDTA, 0.03% SDS) with fluorogenic peptide substrates: Suc-Leu-Leu-Val-Tyr-AMC (10 µM) for CT-L activity, Z-Leu-Leu-Glu-AMC (10 µM) for C-L activity, or Bz-Val-Gly-Arg-AMC (50 µM) for T-L activity [2]. The reaction was initiated by adding the substrate, and the release of free AMC was quantified by measuring fluorescence (excitation/emission 380/460 nm) [2]. HtrA2/Omi Protease Activity Assay: Recombinant human HtrA2 enzyme was incubated with its substrate β-casein in an assay buffer [2]. The cleavage of β-casein was assessed by SDS-PAGE followed by silver staining and quantification of the cleaved product [2]. |
| Cell Assay |
The MTT assay is used to determine cell viability. Trypsinized cells are seeded at 5000 per well in 96-well plates. Ixazomib or DMSO are added to basal medium and given to the cells at the prescribed times and doses. The viability of the cells is assessed in relation to control cells that were given the vehicle alone. Cell Proliferation Assay (MTT): MG-63, Saos-2, and normal osteoblast hFOB1.19 cells were seeded in 96-well plates. After adherence, cells were starved in basal medium for 12h, then treated with various concentrations of MLN2238 (0.20 µM to 3.20 µM) or vehicle (DMSO) dissolved in fresh basal medium for 12h, 24h, or 48h. MTT reagent was added, and cell viability was determined relative to vehicle-treated controls. [1] Apoptosis Assay (Flow Cytometry): Cells were treated with MLN2238 at indicated doses for 24h, collected, washed, resuspended in binding buffer, and stained with Annexin V-FITC and propidium iodide (PI) in the dark for 15 minutes. Apoptotic cells (Annexin V+/PI- and Annexin V+/PI+) were analyzed using a flow cytometer. In some experiments, cells were pre-treated for 1h with caspase inhibitors (Z-VAD-FMK pan-caspase inhibitor, IETD-FMK caspase-8 inhibitor, or LEHD-FMK caspase-9 inhibitor) prior to MLN2238 addition. [1] Cell Cycle Analysis (Flow Cytometry): Cells treated with MLN2238 for 6h, 12h, or 24h were harvested, washed, fixed, and stained with PI using a commercial cell cycle detection kit. DNA content was analyzed by flow cytometry to determine cell cycle distribution. [1] Western Blot Analysis: Cells were lysed with RIPA buffer containing protease inhibitors. Protein concentrations were determined using a BCA assay. Proteins were separated by SDS-PAGE, transferred to nitrocellulose membranes, blocked with non-fat milk, and incubated with primary antibodies overnight at 4°C. Membranes were then incubated with HRP-conjugated secondary antibodies, and protein bands were visualized using enhanced chemiluminescence substrate. [1] Subcellular Fractionation: Mitochondrial and cytosolic fractions were isolated from treated cells using a mitochondrial isolation kit. Cells were homogenized and centrifuged to separate fractions, which were then analyzed by Western blotting. [1] In Vitro Invasion Assay: Cells were seeded into the upper chamber of a Matrigel-coated Transwell insert with serum-free medium. Medium with 20% FBS was placed in the lower chamber as a chemoattractant. After 12h, cells were treated with MLN2238 or vehicle for 24h. Invaded cells on the lower membrane surface were stained with Calcein AM and counted under a fluorescence microscope. [1] Cell Viability Assay (MTT and CellTiter-Glo): MM cell lines or patient-derived cells were seeded in culture plates and treated with various concentrations of MLN2238 for 48 hours [2]. Cell viability was assessed using the MTT assay or the CellTiter-Glo luminescent cell viability assay according to the manufacturer's instructions [2]. Apoptosis Assay (Annexin V/PI Staining): Cells treated with MLN2238 were collected, washed, and stained with Annexin V-FITC and propidium iodide (PI) using a commercial kit [2]. Apoptotic cells (Annexin V+/PI- and Annexin V+/PI+) were analyzed by flow cytometry [2]. Proliferation Assay (Thymidine Incorporation): MM.1S cells were co-cultured with or without patient-derived bone marrow stromal cells (BMSCs) in the presence or absence of MLN2238 for 48 hours [2]. Proliferation was measured by the incorporation of tritiated thymidine into DNA [2]. Western Blot Analysis: Cells were lysed, and protein concentrations were determined [2]. Proteins were separated by SDS-PAGE, transferred to membranes, and probed with specific primary antibodies overnight at 4°C [2]. After incubation with HRP-conjugated secondary antibodies, protein bands were visualized using enhanced chemiluminescence [2]. NF-κB Activity Assay (ELISA): MM.1S cells were treated with MLN2238 for various times, harvested, and total cellular proteins were extracted [2]. NF-κB p65 and p52 activities were measured using a commercial transcription factor ELISA kit according to the manufacturer's instructions [2]. In Vitro Angiogenesis Assay (Tube Formation): Human umbilical vein endothelial cells (HUVECs) were pre-treated with vehicle or MLN2238 (10 nM) for 8 hours [2]. Cells were then washed, seeded onto Matrigel-coated 96-well plates, and incubated for an additional 4 hours to allow capillary-like tube formation [2]. Tube structures were visualized and photographed using an inverted microscope [2]. Cell viability was assessed in parallel using trypan blue exclusion [2]. |
| Animal Protocol |
Ixazomib is dissolved at a concentration of 2 mg/mL in 5% 2-hydroxypropyl-β-cyclodextrin. The test makes use of a human plasmacytoma xenograft tumor model. After receiving a subcutaneous inoculation of 5.0×106 MM.1S cells in 100 µL serum-free RPMI-1640 medium, 21 CB-17 SCID mice are randomly assigned to treatment groups once their tumors have grown to a size of 250–300 mm3. For three weeks, mice are given vehicle, bortezomib (1 mg/kg; i.v.) or ixazomib (11 mg/kg; i.v.) twice a week. When a tumor grows to be 2 cm3, the animal is put to death. Human MM Xenograft Model: CB-17 SCID mice (female, 6 weeks old) were subcutaneously inoculated with 5.0 × 10^6 MM.1S cells in 100 µL serum-free RPMI-1640 medium [2]. When tumors reached 250–300 mm³ (around day 28-30), mice were randomized into treatment groups [2]. MLN2238 was dissolved in 5% 2-hydroxypropyl-β-cyclodextrin at a concentration of 2 mg/mL [2]. For intravenous treatment, mice received MLN2238 (11 mg/kg) or vehicle twice weekly for 3 weeks [2]. A comparator group received bortezomib (1 mg/kg, i.v.) on the same schedule [2]. For oral treatment, a separate group of tumor-bearing mice received MLN2238 (8 mg/kg) or vehicle by oral gavage twice weekly for 3 weeks [2]. Tumor volume was measured regularly [2]. Animals were euthanized when tumors reached 2 cm³ [2]. Blood samples were collected for chemistry analysis [2]. Tumors were harvested, sectioned, and subjected to immunohistochemical staining for cleaved caspase-3, TUNEL, Ki-67, VEGFR2, and Pecam [2]. |
| ADME/Pharmacokinetics |
MLN9708 is described as an orally bioavailable citrate ester prodrug that is rapidly hydrolyzed to its active form, MLN2238, upon exposure to aqueous solutions or plasma. MLN2238 is noted to have an improved pharmacokinetic and pharmacodynamic profile and a shorter proteasome dissociation half-life compared to earlier proteasome inhibitors, leading to greater distribution and effects in tumor tissue than in blood. Specific PK parameters (e.g., half-life, bioavailability) are not provided. [1] MLN9708 is an orally bioavailable proteasome inhibitor [2]. Upon exposure to aqueous solutions or plasma, it rapidly hydrolyzes to its biologically active form, MLN2238 [2]. MLN2238 is noted to have a shorter proteasome dissociation half-life and improved pharmacokinetic and pharmacodynamic profiles compared to bortezomib, leading to greater distribution and effects in tumor tissue than in blood [2]. Specific PK parameters (e.g., half-life, oral bioavailability) are not provided in this study [2]. |
| Toxicity/Toxicokinetics |
In vitro, MLN2238 exhibited much lower cytotoxicity against normal human osteoblast cells (hFOB1.19) compared to osteosarcoma cells, even at the highest tested dose (3.20 µM) for up to 48h, suggesting a favorable therapeutic index. No other specific toxicity data (e.g., LD50, organ toxicity) are provided. [1] In vitro, MLN2238 at concentrations effective against MM cells did not significantly affect the viability of normal PBMCs [2]. In vivo, MLN2238 was well tolerated in mice [2]. Blood chemistry profiles (creatinine, hemoglobin, bilirubin) of treated mice remained within normal ranges [2]. No significant treatment-related toxicities or mortality were reported at the tested doses (11 mg/kg i.v., 8 mg/kg p.o.) [2]. |
| References |
[1]. A New Perspective for Osteosarcoma Therapy: Proteasome Inhibition by MLN9708/2238 Successfully Induces Apoptosis and Cell Cycle Arrest and Attenuates the Invasion Ability of Osteosarcoma Cells in Vitro. Cell Physiol Biochem. 2017 Jan 27;41(2 [2]. In vitro and in vivo selective antitumor activity of a novel orally bioavailable proteasome inhibitor MLN9708 against multiple myeloma cells. Clin Cancer Res. 2011 Aug 15;17(16):5311-21. [3]. Evaluation of the proteasome inhibitor MLN9708 in preclinical models of human cancer. Cancer Res. 2010 Mar 1;70(5):1970-80. |
| Additional Infomation |
Ixazomib citrate is a glycine derivative that is the amide obtained by formal condensation of the carboxy group of N-(2,5-dichlorobenzoyl)glycine with the amino group of 2,2'-{2-[(1R)-1-amino-3-methylbutyl]-5-oxo-1,3,2-dioxaborolane-4,4-diyl}diacetic acid. A prodrug for ixazomib that is used in combination therapy for treatment of multiple myeloma. It has a role as a prodrug, a proteasome inhibitor, an orphan drug, an antineoplastic agent and an apoptosis inducer. It is a glycine derivative, a member of benzamides, a dichlorobenzene, an oxo dicarboxylic acid and a 1,3,2-dioxaborolane. It is functionally related to an ixazomib. Ixazomib Citrate is the citrate salt form of ixazomib, an orally bioavailable second generation proteasome inhibitor (PI) with potential antineoplastic activity. Ixazomib inhibits the activity of the proteasome, blocking the targeted proteolysis normally performed by the proteasome, which results in an accumulation of unwanted or misfolded proteins; disruption of various cell signaling pathways may follow, resulting in the induction of apoptosis. Compared to first generation PIs, second generation PIs may have an improved pharmacokinetic profile with increased potency and less toxicity. Proteasomes are large protease complexes that degrade unneeded or damaged proteins that have been ubiquinated. See also: Ixazomib (has active moiety). Drug Indication Ninlaro in combination with lenalidomide and dexamethasone is indicated for the treatment of adult patients with multiple myeloma who have received at least one prior therapy. Treatment of systemic light chain amyloidosis Ixazomib citrate (MLN9708) is an investigational small-molecule proteasome inhibitor. It is highlighted as an orally bioavailable agent, which is a practical advantage. The study concludes that proteasome inhibition by MLN9708/2238 represents a novel biochemical target for osteosarcoma treatment in vitro, inducing apoptosis, cell cycle arrest, and reducing invasion. The clinical potential is suggested, as MLN9708 was under evaluation in phase I clinical trials at the time of the study. [1] Ixazomib citrate (MLN9708) is a second-generation, orally bioavailable small-molecule proteasome inhibitor, structurally distinct from bortezomib (a boronic acid analog) [2]. It is highlighted as a promising therapy for multiple myeloma (MM), including in settings of resistance to conventional therapies and bortezomib [2]. The study suggests that MLN2238 does not significantly inhibit the neuronal survival protease HtrA2/Omi, which may be linked to bortezomib-induced peripheral neuropathy, indicating a potentially improved safety profile [2]. The preclinical data support the clinical evaluation of MLN9708 as a single agent or in combination with lenalidomide, HDAC inhibitors, or dexamethasone for the treatment of MM [2]. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 100~250 mg/mL (193.4~483.5 mM) Ethanol: ~100 mg/mL (~193.4 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.02 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.02 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.02 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9338 mL | 9.6689 mL | 19.3379 mL | |
| 5 mM | 0.3868 mL | 1.9338 mL | 3.8676 mL | |
| 10 mM | 0.1934 mL | 0.9669 mL | 1.9338 mL |