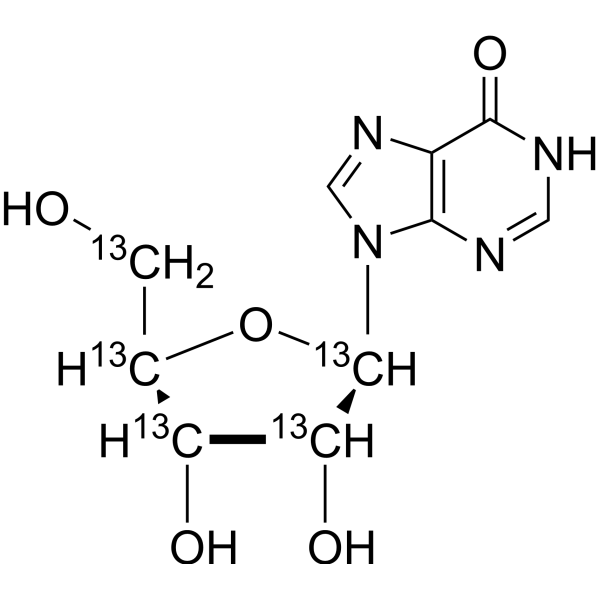
Physicochemical Properties
| Molecular Formula | C513C5H12N4O5 |
| Molecular Weight | 273.19 |
| Appearance | Solid powder |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Stable heavy isotopes of hydrogen, carbon, and other elements have been incorporated into drug molecules, largely as tracers for quantitation during the drug development process. Studies involving the human use of drugs with labeled deuterium suggest that these compounds may offer some advantages when compared with their nondeuterated counterparts. Deuteration has gained attention because of its potential to affect the pharmacokinetic and metabolic profiles of drugs. Deutetrabenazine is the first deuterated drug to receive Food and Drug Administration approval. This deuterated form of the drug tetrabenazine is indicated for the treatment of chorea associated with Huntington's disease as well as tardive dyskinesia. Ongoing clinical trials suggest that a number of other deuterated compounds are being evaluated for the treatment of human diseases and not merely as research tools. [1] |
| ln Vivo | Deuterated compounds may, in some cases, offer advantages over nondeuterated forms, often through alterations in clearance. Deuteration may also redirect metabolic pathways in directions that reduce toxicities. The approval of additional deuterated compounds may soon follow. Clinicians will need to be familiar with the dosing, efficacy, potential side effects, and unique metabolic profiles of these new entities. |
| References |
[1]. Impact of Deuterium Substitution on the Pharmacokinetics of Pharmaceuticals. Ann Pharmacother. 2019 Feb;53(2):211-239. |
Solubility Data
| Solubility (In Vitro) | DMSO : 10 mg/mL (36.60 mM; with sonication) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1 mg/mL (3.66 mM)(saturation unknown) in 10% DMSO 40% PEG300 5% Tween-80 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution This solution produces a clear solution of ≥ 1 mg/mL (saturation unknown). For example, if 1 mL of working solution, add 100 μL of 10.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix well; then add 50 μL Tween-80 to the above system and mix well; then add 450 μL saline to make up to 1 mL. *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1 mg/mL (3.66 mM)(saturation unknown) in 10% DMSO 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution This protocol will produce a clear solution of ≥ 1 mg/mL (saturation unknown). . For example, if 1 mL of working solution, add 100 μL of 10.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD in saline and mix. *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 1 mg/mL (3.66 mM)(Saturation unknown) in 10% DMSO 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution, add 100 μL of 10.0 mg/mL clear DMSO stock solution to 900 μL corn oil and mix well. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6605 mL | 18.3023 mL | 36.6046 mL | |
| 5 mM | 0.7321 mL | 3.6605 mL | 7.3209 mL | |
| 10 mM | 0.3660 mL | 1.8302 mL | 3.6605 mL |