Physicochemical Properties
| Molecular Formula | C19H15NO4CL-.NA+ |
| Molecular Weight | 379.7695 |
| Exact Mass | 379.058 |
| Elemental Analysis | C, 60.09; H, 3.98; Cl, 9.33; N, 3.69; Na, 6.05; O, 16.85 |
| CAS # | 7681-54-1 |
| Related CAS # | Indomethacin;53-86-1;Indomethacin sodium hydrate;74252-25-8 |
| PubChem CID | 23675763 |
| Appearance | Typically exists as solid at room temperature |
| Melting Point | 162ºC |
| LogP | 2.592 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 26 |
| Complexity | 512 |
| Defined Atom Stereocenter Count | 0 |
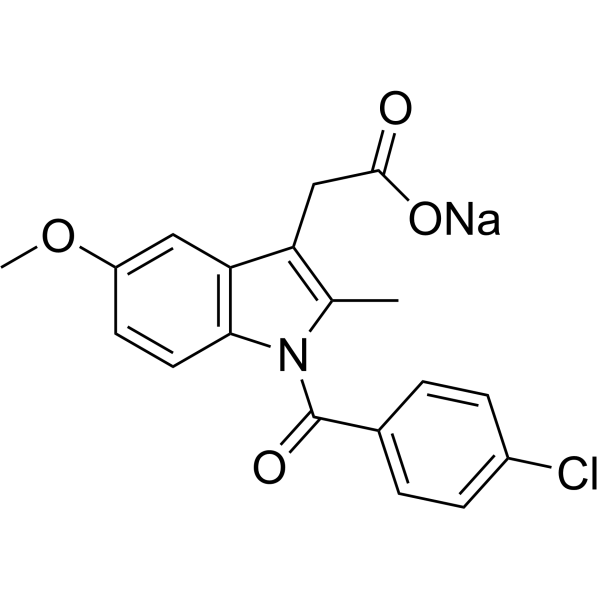
| SMILES | O=C([O-])CC1=C(C)N(C(C2=CC=C(Cl)C=C2)=O)C3=C1C=C(OC)C=C3.[Na+] |
| InChi Key | JMHRGKDWGWORNU-UHFFFAOYSA-M |
| InChi Code | InChI=1S/C19H16ClNO4.Na/c1-11-15(10-18(22)23)16-9-14(25-2)7-8-17(16)21(11)19(24)12-3-5-13(20)6-4-12;/h3-9H,10H2,1-2H3,(H,22,23);/q;+1/p-1 |
| Chemical Name | sodium;2-[1-(4-chlorobenzoyl)-5-methoxy-2-methylindol-3-yl]acetate |
| Synonyms | INDOMETHACIN SODIUM; 7681-54-1; Sodium indomethacin; Osmosin; Indomethacin (sodium); Indocin I.V.; EINECS 231-670-4; Indomethacin sodium anhydrous; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | hCOX-1 (IC50 = 18 nM in CHO cells); hCOX-2 (IC50 = 26 nM in CHO cells) |
| ln Vitro | In vitro anticancer activity of indomethacin sodium (0-150 μM; 24 hours; 3LL-D122 cells) has been reported [2]. Viral protein translation is halted and viral replication is inhibited when indomethacin sodium (0-1000 μM) activates PKR and phosphorylates eLF2α, shielding host cells from viral damage (IC50=2μM) [3]. |
| ln Vivo | In male Sprague-Dawley rats, indomethacin sodium (0.01–10 mg/kg; oral; 3 hours) causes hyperalgesia and paw edema, and it reverses carrageenan-induced hyperalgesia in a dose-dependent manner [1]. Tumor growth can be inhibited in vivo by indomethacin sodium (10 mg/mL); oral; once daily for 29 days; male C57BL/6J mice) [2]. |
| Enzyme Assay |
Determination of Ki and k2 values for the time-dependent inhibition of COX-2[1] Purified COX-2 (2.3 μg) was preincubated with inhibitor for 0–15 min in 180 μl of the reaction buffer described above, before the initiation of the reaction with a mixture of arachidonic acid and TMPD. The cyclo-oxygenase activity was determined by the spectrophotometric method as described above. For experiments performed without preincubation of the inhibitor, the reaction was initiated by addition of the assay mixture containing the enzyme to the inhibitor and arachidonic acid/TMPD ethanolic solution. The rate constants (kobs) for the time-dependent loss of activity at each inhibitor concentration were calculated by fitting of the data to a first order equation of the form y=a + b.exp(–kobst) by use of Sigmaplot software. Data were analysed in terms of the model developed by Rome and Lands (1975) for the time-dependent inhibition of ovine COX-1. In this model (Scheme 1), an initial reversible binding of enzyme and inhibitor (characterized by the dissociation constant Ki) is followed by a first order inactivation process (characterized by a first order rate constant k2). The rate of reversal of this process (k–2) is considered to be negligible. Determination of the stoichiometry of inhibitor binding[1] Aliquots of purified COX-2 (0.25 mg ml-1, concentration of subunit of 3.4 μm) were incubated in buffer (100 mm Tris-HCl, pH 8.0, 5 mm EDTA, 1 mm phenol) in the presence of varying concentrations of inhibitors (0–8 μm) for 15 or 30 min. An aliquot (20 μl) was then removed for determination of the remaining cyclo-oxygenase activity by oxygen uptake as described above. Enzyme concentration was determined by amino acid concentration following acid hydrolysis (Percival et al., 1994). Competition of time-dependent inhibition of COX-2 by arachidonic acid[1] Purified COX-2 (3.6 μg) was diluted into preincubation buffer (0.03 ml, 100 mm Tris-HCl, pH 8.0, 5 mm EDTA, 2 mm phenol) containing 60 mm diethyldithiocarbamic acid to prevent substrate oxygenation (Lands et al., 1974) and either 10 μm inhibitor, or 10 μm inhibitor plus 5 μm arachidonic acid, or 10 μm inhibitor plus 30 μm arachidonic acid. After a preincubation period of 0–4 min, the total enzyme was assayed for enzymatic activity by oxygen consumption at 30°C as described above. |
| Cell Assay |
Cell Viability Assay[2] Cell Types: 3LL-D122 cells (highly metastatic variant of mouse LL cancer cells) Tested Concentrations: 0, 20, 50, 100 and 150μM Incubation Duration: 24 hrs (hours) Experimental Results: Cell viability was inhibited at 20 mM , the inhibition rate reaches 50% at 60 nM. Cell cycle analysis[2] Cell Types: 3LL-D122 cells (highly metastatic variant of mouse LL cancer cells) Tested Concentrations: 0, 30 and 80μM Incubation Duration: 24 hrs (hours) Experimental Results: diminished percentage of cells in G2/M phase and increased in G1 phase Percentage of cells. |
| Animal Protocol |
Animal/Disease Models: Male SD (SD (Sprague-Dawley)) rat[1] Doses: 0.01-10 mg/kg Route of Administration: po (po (oral gavage)) 3 hrs (hrs (hours)) Experimental Results: Inhibited carrageenan-induced rat paw edema in a dose-dependent manner (ED50=2.0 mg/kg) and hyperalgesia (ED50=1.5mg/kg). Animal/Disease Models: Male C57BL/6J mice [2] Doses: 10 mg/mL Route of Administration: po (po (oral gavage)) one time/day for 29 days Experimental Results: Delayed the onset of tumor growth and the initial growth rate of footpad tumors. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Because of the low levels of indomethacin in breastmilk and therapeutic administration directly to infants, it is acceptable to use in nursing mothers. However, other agents with more published information on use during lactation may be preferable, especially while nursing a newborn or preterm infant. ◉ Effects in Breastfed Infants In one case report, a breastfeeding mother had been taking daily doses of indomethacin that increased to 200 mg (3 mg/kg) from the fourth to the sixth day postpartum. On the same day that indomethacin was stopped, the infant had a generalized seizure, followed by another on the next day. No metabolic findings could account for the convulsions and no indomethacin levels were measured in the mother or infant. This case was rated as indomethacin possibly causing the seizure; however, later studies and the established therapeutic use of indomethacin in newborns make this causality seem unlikely. In one study, 7 women breastfed their neonates while taking indomethacin. No adverse effects were noted in any of the infants. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. |
| References |
[1]. Biochemical and pharmacological profile of a tetrasubstituted furanone as a highly selective COX-2 inhibitor. Br J Pharmacol. 1997 May;121(1):105-17. [2]. Inhibition of viral protein translation by indomethacin in vesicular stomatitis virus infection: role of eIF2α kinase PKR. Cell Microbiol. 2015 Sep;17(9):1391-404. [3]. Comparative effects of indomethacin on cell proliferation and cell cycle progression in tumor cells grown in vitro and in vivo. Biochem Pharmacol. 2001 Mar 1;61(5):565-71. |
| Additional Infomation |
Indomethacin Sodium is the sodium salt of indomethacin, a methylated indole derivative with anti-inflammatory, analgesic-antipyretic and tocolytic effects. Indomethacin is a non-selective, reversible, and competitive inhibitor of cyclooxygenases 1 and 2, thereby blocking the conversion of arachidonic acid into prostaglandin precursors. Consequently, prostaglandin synthesis is decreased, and prostaglandin-mediated activities are prevented, including pain, inflammation, fever and uterine contraction. A non-steroidal anti-inflammatory agent (NSAID) that inhibits CYCLOOXYGENASE, which is necessary for the formation of PROSTAGLANDINS and other AUTACOIDS. It also inhibits the motility of POLYMORPHONUCLEAR LEUKOCYTES. See also: Indomethacin (has active moiety). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6332 mL | 13.1659 mL | 26.3317 mL | |
| 5 mM | 0.5266 mL | 2.6332 mL | 5.2663 mL | |
| 10 mM | 0.2633 mL | 1.3166 mL | 2.6332 mL |