Physicochemical Properties
| Molecular Formula | C26H30CLNO4 |
| Molecular Weight | 455.97 |
| Exact Mass | 455.186 |
| Elemental Analysis | C, 68.49; H, 6.63; Cl, 7.77; N, 3.07; O, 14.03 |
| CAS # | 282728-47-6 |
| PubChem CID | 10389320 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 533.6±50.0 °C at 760 mmHg |
| Flash Point | 276.5±30.1 °C |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.565 |
| LogP | 6.75 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 32 |
| Complexity | 607 |
| Defined Atom Stereocenter Count | 0 |
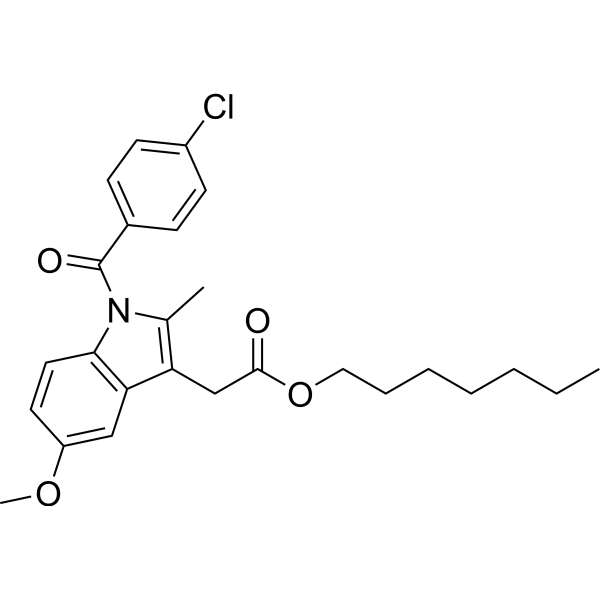
| SMILES | CCCCCCCOC(=O)CC1=C(C)N(C2=C1C=C(C=C2)OC)C(=O)C3=CC=C(C=C3)Cl |
| InChi Key | PYBCHCVNKGZCOH-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C26H30ClNO4/c1-4-5-6-7-8-15-32-25(29)17-22-18(2)28(24-14-13-21(31-3)16-23(22)24)26(30)19-9-11-20(27)12-10-19/h9-14,16H,4-8,15,17H2,1-3H3 |
| Chemical Name | heptyl 2-[1-(4-chlorobenzoyl)-5-methoxy-2-methylindol-3-yl]acetate |
| Synonyms | Indomethacin heptyl ester; 282728-47-6; INDOMETHACIN ESTER, N-HEPTYL-; heptyl 2-[1-(4-chlorobenzoyl)-5-methoxy-2-methylindol-3-yl]acetate; CHEMBL330194; CHEBI:184056; HMS3649K19; BDBM50090775; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | COX-2 (IC50 = 40 nM) |
| ln Vitro | Recent studies from our laboratory have shown that derivatization of the carboxylate moiety in substrate analogue inhibitors, such as 5,8,11,14-eicosatetraynoic acid, and in nonsteroidal antiinflammatory drugs (NSAIDs), such as indomethacin and meclofenamic acid, results in the generation of potent and selective cyclooxygenase-2 (COX-2) inhibitors (Kalgutkar et al. Proc. Natl. Acad. Sci. U.S.A. 2000, 97, 925-930). This paper summarizes details of the structure-activity studies involved in the transformation of the arylacetic acid NSAID, indomethacin, into a COX-2-selective inhibitor. Many of the structurally diverse indomethacin esters and amides inhibited purified human COX-2 with ICo5 values in the low-nanomolar range but did not inhibit ovine COX-1 activity at concentrations as high as 66 microM. Primary and secondary amide analogues of indomethacin were more potent as COX-2 inhibitors than the corresponding tertiary amides. Replacement of the 4-chlorobenzoyl group in indomethacin esters or amides with the 4-bromobenzyl functionality or hydrogen afforded inactive compounds. Likewise, exchanging the 2-methyl group on the indole ring in the ester and amide series with a hydrogen also generated inactive compounds. Inhibition kinetics revealed that indomethacin amides behave as slow, tight-binding inhibitors of COX-2 and that selectivity is a function of the time-dependent step. Conversion of indomethacin into ester and amide derivatives provides a facile strategy for generating highly selective COX-2 inhibitors and eliminating the gastrointestinal side effects of the parent compound.[1] |
| Enzyme Assay |
Enzymology. [1] COX-1 was purified from ram seminal vesicles as described earlier.36 The specific activity of the protein was 20 (μMO2/min)/mg, and the percentage of holoprotein was 13.5%. ApoCOX-1 was prepared as described earlier.37 Apoenzyme was reconstituted by the addition of hematin to the assay mixtures. Human COX-2 was expressed in SF-9 insect cells by means of the pVL 1393 expression vector and purified by ion-exchange and gel filtration chromatography. All of the purified proteins were shown by densitometric scanning of a 7.5% SDS PAGE gel to be >80% pure. Time- and Concentration-Dependent Inhibition of Ovine COX-1 and Human COX-2 Using Thin Layer Chromatography (TLC) Assay. [1] Cyclooxygenase activity of ovine COX-1 (44 nM) or human COX-2 (66 nM) was assayed by TLC. Reaction mixtures of 200 μL consisted of hematin-reconstituted protein in 100 mM Tris-HCl, pH 8.0, 500 μM phenol, and [1-14C]arachidonic acid (50 μM, ∼55−57 mCi/mmol). For the time-dependent inhibition assay, hematin-reconstituted COX-1 (44 nM) or COX-2 (66 nM) was preincubated at room temperature for 20 min with varying inhibitor concentrations in DMSO followed by the addition of [1-14C]arachidonic acid (50 μM) for 30 s at 37 °C. Reactions were terminated by solvent extraction in Et2O/CH3OH/1 M citrate, pH 4.0 (30:4:1). The phases were separated by centrifugation at 2000g for 2 min and the organic phase was spotted on a TLC plate. The plate was developed in EtOAc/CH2Cl2/glacial AcOH (75:25:1) at 4 °C. Radiolabeled prostanoid products were quantitated with a radioactivity scanner. The percentage of total products observed at different inhibitor concentrations was divided by the percentage of products observed for protein samples preincubated for the same time with DMSO. |
| Cell Assay |
Inhibition of COX-2 Activity in Activated RAW264.7. [1] Protocols for COX-2 inhibition in RAW264.7 cells have been previously described.23 Briefly, cells (6.2 × 106 cells/T25 flask) were activated with lipopolysaccharide (1 μg/mL) and γ-interferon (10 U/mL) in serum-free DMEM for 7 h and then treated with inhibitor (0−2 μM) for 30 min at 37 °C. Exogenous arachidonate metabolism was determined by adding [1-14C]arachidonic acid (20 μM) for 15 min at 37 °C. IC50 values are the average of two independent determinations. |
| References | [1]. Ester and amide derivatives of the nonsteroidal antiinflammatory drug, indomethacin, as selective cyclooxygenase-2 inhibitors. J Med Chem. 2000 Jul 27;43(15):2860-70. |
| Additional Infomation | Indomethacin heptyl ester is a N-acylindole. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1931 mL | 10.9656 mL | 21.9313 mL | |
| 5 mM | 0.4386 mL | 2.1931 mL | 4.3863 mL | |
| 10 mM | 0.2193 mL | 1.0966 mL | 2.1931 mL |