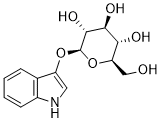
Indican (Indoxyl-β-D-glucoside) is a naturally occurring plantsglycoside of indoxyl and a colourless organic compound found in Indigofera. It is a precursor of the dyesindigo and indirubin.
Physicochemical Properties
| Molecular Formula | C14H17NO6 |
| Molecular Weight | 295.29 |
| Exact Mass | 295.105 |
| CAS # | 487-60-5 |
| Related CAS # | 1328-73-0 (trihydrate);487-60-5; |
| PubChem CID | 441564 |
| Appearance | Off-white to light yellow solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 606.7±55.0 °C at 760 mmHg |
| Melting Point | 57-58ºC |
| Flash Point | 320.7±31.5 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.715 |
| LogP | -0.75 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 21 |
| Complexity | 356 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | C1=CC=C2C(=C1)C(=CN2)O[C@H]3[C@@H]([C@H]([C@@H]([C@H](O3)CO)O)O)O |
| InChi Key | XVARCVCWNFACQC-RKQHYHRCSA-N |
| InChi Code | InChI=1S/C14H17NO6/c16-6-10-11(17)12(18)13(19)14(21-10)20-9-5-15-8-4-2-1-3-7(8)9/h1-5,10-19H,6H2/t10-,11-,12+,13-,14-/m1/s1 |
| Chemical Name | (2R,3S,4S,5R,6S)-2-(Hydroxymethyl)-6-(1H-indol-3-yloxy)oxane-3,4,5-triol |
| Synonyms | NSC87517 IndicanNSC 87517 NSC-87517 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo | Indican ((Indolyl-beta-D-glucoside; Oral; 20, 40 mg/kg; Single dose) increases MTX by significant metabolite inhibition of numerous anion transporters, including OAT 1, OAT 3, and MRP 4 Systemic exposure and MRT IS[1]. |
| Animal Protocol |
Animal/Disease Models: Male Spraguee Dawley rat, body weight 330-400 g[1] Doses: 20, 40 mg/kg Route of Administration: Oral Experimental Results: Methotrexate (MTX) Increased systemic exposure and multidrug resistance-related protein (MRP; 5.0 mg/kg). |
| ADME/Pharmacokinetics |
Metabolism / Metabolites Uremic toxins tend to accumulate in the blood either through dietary excess or through poor filtration by the kidneys. Most uremic toxins are metabolic waste products and are normally excreted in the urine or feces. |
| Toxicity/Toxicokinetics |
Toxicity Summary Uremic toxins such as indican are actively transported into the kidneys via organic ion transporters (especially OAT3). Increased levels of uremic toxins can stimulate the production of reactive oxygen species. This seems to be mediated by the direct binding or inhibition by uremic toxins of the enzyme NADPH oxidase (especially NOX4 which is abundant in the kidneys and heart) (A7868). Reactive oxygen species can induce several different DNA methyltransferases (DNMTs) which are involved in the silencing of a protein known as KLOTHO. KLOTHO has been identified as having important roles in anti-aging, mineral metabolism, and vitamin D metabolism. A number of studies have indicated that KLOTHO mRNA and protein levels are reduced during acute or chronic kidney diseases in response to high local levels of reactive oxygen species (A7869). |
| References |
[1]. Transporter-mediated interaction of indican and methotrexate in rats. J Food Drug Anal. 2018 Apr;26(2S):S133-S140. [2]. Indoxyl sulfate, a uremic toxin, is biotransformed from indoxyl-beta-D-glucoside (indican) in rats. Toxicon. 2008 Sep 1;52(3):440-4. |
| Additional Infomation |
Indican is an indolyl carbohydrate, a beta-D-glucoside and an exopolysaccharide. Indican has been reported in Strobilanthes cusia, Calanthe discolor, and other organisms with data available. Indican is a toxic metabolite derived from dietary proteins and tryptophan. In the intestine, proteins and tryptophan are converted to indole by tryptophanase-expressing organisms. In the liver, indole is hydroxylated to form indoxyl and indoxyl is sufated to produce indican. Overproduction of indican is associated with glomerular sclerosis, interstitial fibrosis and renal failure. Indican is a uremic toxin. Uremic toxins can be subdivided into three major groups based upon their chemical and physical characteristics: 1) small, water-soluble, non-protein-bound compounds, such as urea; 2) small, lipid-soluble and/or protein-bound compounds, such as the phenols and 3) larger so-called middle-molecules, such as beta2-microglobulin. Chronic exposure of uremic toxins can lead to a number of conditions including renal damage, chronic kidney disease and cardiovascular disease. It is a colourless organic compound, soluble in water, naturally occurring in Indigofera plants. It is a precursor of indigo dye. Indican interferes with many commercial procedures for measuring total bilirubin[6] which can be a problem for renal failure patients where blood indican levels are raised. It can cause gastrointestinal symptoms in patients where protein absorption is reduced - like Hartnup's disease, allowing for greater bacterial decomposition of the Tryptophan to indole and its conversion to indican. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~338.65 mM) MEthanol : ~83.33 mg/mL (~282.20 mM) H2O : ~20 mg/mL (~67.73 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.47 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.47 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (8.47 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 14.29 mg/mL (48.39 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3865 mL | 16.9325 mL | 33.8650 mL | |
| 5 mM | 0.6773 mL | 3.3865 mL | 6.7730 mL | |
| 10 mM | 0.3387 mL | 1.6933 mL | 3.3865 mL |