Physicochemical Properties
| Molecular Formula | C11H16N8O8 |
| Molecular Weight | 388.2935 |
| Exact Mass | 388.109 |
| CAS # | 39236-46-9 |
| Related CAS # | 71042-94-9 (mono-hydrochloride salt) |
| PubChem CID | 38258 |
| Appearance | White to off-white solid powder |
| Density | 1.9±0.1 g/cm3 |
| Index of Refraction | 1.715 |
| LogP | -5.36 |
| Hydrogen Bond Donor Count | 8 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 27 |
| Complexity | 627 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | ZCTXEAQXZGPWFG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H16N8O8/c20-2-18-4(6(22)16-10(18)26)14-8(24)12-1-13-9(25)15-5-7(23)17-11(27)19(5)3-21/h4-5,20-21H,1-3H2,(H2,12,14,24)(H2,13,15,25)(H,16,22,26)(H,17,23,27) |
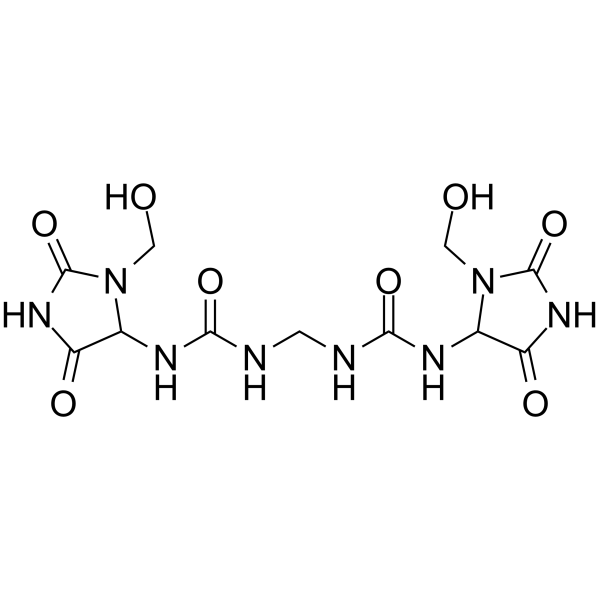
| Chemical Name | 1-[3-(hydroxymethyl)-2,5-dioxoimidazolidin-4-yl]-3-[[[3-(hydroxymethyl)-2,5-dioxoimidazolidin-4-yl]carbamoylamino]methyl]urea |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
1. Decomposition Experiment in Cosmetics and Patch Test Materials: Different cosmetic formulations (e.g., lotion, cream) and patch test materials containing Imidazolidinyl urea were stored at 4°C, 25°C, and 40°C for 0, 1, 3, 6, and 12 months. Samples were collected at each time point, and high-performance liquid chromatography (HPLC) was used to analyze the concentration of Imidazolidinyl urea and its decomposition products (formaldehyde, N-(2-hydroxyethyl)ethyleneurea, N-(2-hydroxyethyl)urea). The results showed that Imidazolidinyl urea decomposed gradually with increasing storage temperature and time; at 40°C for 12 months, its residual rate in lotions was only 35-45%, while formaldehyde content increased from initial 0.01-0.02% to 0.08-0.10% [1] 2. In Vitro Sensitization-Related Detection: Extracts of Imidazolidinyl urea-containing patch test materials were prepared using physiological saline. The extracts were tested for formaldehyde release using a formaldehyde-specific colorimetric kit. The data indicated that the amount of formaldehyde released from Imidazolidinyl urea was positively correlated with the storage time and temperature of the patch test materials; fresh materials released 0.03-0.05 μg/mg formaldehyde, while those stored at 40°C for 6 months released 0.12-0.15 μg/mg formaldehyde [1] |
| ln Vivo |
1. Human Patch Test for Sensitization: A total of 120 volunteers with a history of cosmetic contact dermatitis were selected for the patch test. The patch test materials contained Imidazolidinyl urea at concentrations of 0.5%, 1.0%, and 2.0% (dissolved in petrolatum). The patches were applied to the volar forearm of volunteers and removed after 48 hours; skin reactions were evaluated at 48 hours and 72 hours after application according to the International Contact Dermatitis Research Group (ICDRG) criteria. The results showed that the positive reaction rate (erythema, edema, or vesicles) was 8.3% (10/120) for 2.0% Imidazolidinyl urea, 3.3% (4/120) for 1.0%, and 0% (0/120) for 0.5%. No severe adverse reactions (e.g., blistering, ulceration) were observed [1] |
| ADME/Pharmacokinetics |
Metabolism / Metabolites Formaldehyde may be absorbed following inhalation, oral, or dermal exposure. It is an essential metabolic intermediate in all cells and is produced during the normal metabolism of serine, glycine, methionine, and choline and also by the demethylation of N-, S-, and O-methyl compounds. Exogenous formaldehyde is metabolized to formate by the enzyme formaldehyde dehydrogenase at the initial site of contact. After oxidation of formaldehyde to formate, the carbon atom is further oxidized to carbon dioxide or incorporated into purines, thymidine, and amino acids via tetrahydrofolatedependent one-carbon biosynthetic pathways. Formaldehyde is not stored in the body and is excreted in the urine (primarily as formic acid), incorporated into other cellular molecules, or exhaled as carbon dioxide. Nitrosamines can also enter the body via ingestion, inhalation, or dermal contact. Once in the body, nitrosamines are metabolized by cytochrome P-450 enzymes, which essentially activates them into carcinogens. (L962, A2878, A2879) |
| Toxicity/Toxicokinetics |
Toxicity Summary Imidazolidinyl urea is a formaldehyde releaser. It is likely that formaldehyde toxicity occurs when intracellular levels saturate formaldehyde dehydrogenase activity, allowing the unmetabolized intact molecule to exert its effects. Formaldehyde is known to form cross links between protein and DNA and undergo metabolic incorporation into macromolecules (DNA, RNA, and proteins). Imidazolidinyl urea is also a nitrosating agent. Nitrosating agents may decompose and/or react to cause nitrosamine contamination. Nitrosamines are produced from secondary amines and amides in the presence of nitrite ions and are believed to be carcinogenic. Once in the body, nitrosamines are activated by cytochrome P-450 enzymes. They are then believed to induce their carcinogenic effects by forming DNA adducts at the N- and O-atoms. (L962, L1889, L1890, A2878, A2879, A2880, L1894) Toxicity Data LC50 (rat) > 5,000 mg/m3/1hr 1. Contact Sensitization Toxicity: The human patch test confirmed that Imidazolidinyl urea has potential contact sensitizing properties, especially at concentrations ≥1.0%. The sensitization mechanism is associated with its decomposition product formaldehyde, which can react with skin proteins to form haptens, triggering an immune response [1] 2. Decomposition Product-Related Toxicity: Imidazolidinyl urea decomposes to release formaldehyde, a known irritant and sensitizer. In the study, the formaldehyde content in Imidazolidinyl urea-containing samples exceeded the recommended safe limit (0.05%) after long-term storage at high temperatures (40°C for ≥6 months), increasing the risk of skin irritation and sensitization [1] |
| References |
[1]. Characterization of the decomposition of compounds derived from imidazolidinyl urea in cosmetics and patch test materials. Contact Dermatitis. 2012 Nov;67(5):284-92. |
| Additional Infomation |
Imidurea is a member of ureas. It has a role as an antimicrobial agent. Imidurea is an antimicrobial preservative used in cosmetics. It acts as a formaldehyde releaser. Imidurea is a Standardized Chemical Allergen. The physiologic effect of imidurea is by means of Increased Histamine Release, and Cell-mediated Immunity. Imidazolidinyl urea is an antimicrobial preservative used in cosmetics. It acts as a formaldehyde releaser. Imidazolidinyl urea may cause contact dermatitis. It's toxicity is also due to it's ability to react and release formaldehyde and nitrosamines, both of which are believed to be carcinogenic. (L1890, L1894) See also: Aloe vera leaf; imidurea; water (component of) ... View More ... 1. Background and Application: Imidazolidinyl urea is a common formaldehyde-releasing preservative widely used in cosmetics (lotion, cream, shampoo) and personal care products to inhibit the growth of bacteria and fungi. Its preservative effect is achieved by slowly releasing formaldehyde [1] 2. Decomposition Mechanism: Imidazolidinyl urea decomposes via hydrolysis under the influence of temperature, humidity, and pH. The hydrolysis process breaks the imidazolidine ring, generating formaldehyde and other by-products (N-(2-hydroxyethyl)ethyleneurea, N-(2-hydroxyethyl)urea). Higher temperatures and longer storage times accelerate this decomposition [1] 3. Regulatory and Safety Implications: Due to its potential sensitization risk (related to formaldehyde release), the use of Imidazolidinyl urea in cosmetics is restricted in some regions; for example, the European Union (EU) limits its maximum concentration to 1.0% in leave-on products. This study provides data support for optimizing the storage conditions (low temperature, short shelf life) of Imidazolidinyl urea-containing products to reduce safety risks [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ≥ 100 mg/mL (~257.54 mM) H2O : ~100 mg/mL (~257.54 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.44 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.44 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.44 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 100 mg/mL (257.54 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5754 mL | 12.8770 mL | 25.7539 mL | |
| 5 mM | 0.5151 mL | 2.5754 mL | 5.1508 mL | |
| 10 mM | 0.2575 mL | 1.2877 mL | 2.5754 mL |