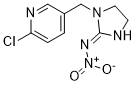
Imidacloprid is a novel and potent insect neurotoxin
Physicochemical Properties
| Molecular Formula | C9H10CLN5O2 |
| Molecular Weight | 255.66 |
| Exact Mass | 255.052 |
| CAS # | 105827-78-9 |
| PubChem CID | 86418 |
| Appearance | White to off-white solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 442.3±55.0 °C at 760 mmHg |
| Melting Point | 136-144ºC |
| Flash Point | 221.3±31.5 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.706 |
| LogP | -0.43 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 17 |
| Complexity | 319 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | YWTYJOPNNQFBPC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C9H10ClN5O2/c10-8-2-1-7(5-12-8)6-14-4-3-11-9(14)13-15(16)17/h1-2,5H,3-4,6H2,(H,11,13) |
| Chemical Name | N-[1-[(6-chloropyridin-3-yl)methyl]-4,5-dihydroimidazol-2-yl]nitramide |
| Synonyms | Confidor Admire Imidacloprid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Metabolism / Metabolites Two main routes of metabolism responsible for the degradation of imidacloprid were identified. The first is oxidative cleavage, yielding 6-chloronicotinic acid, which is conjugated with glycine to form a hippuric acid-type conjugate. These two metabolites together represented most of the identified metabolites, or about 30% of the recovered radiolabel. Of minor importance in terms of quantity is dechlorination of the pyridinyl moiety, producing the 6-hydroxy nicotinic acid and its methylmercapturic acid derivative, probably as a degradation product of a glutathione conjugate. The 6-methylmercapto nicotinic acid conjugated with glycine, and the glycine conjugate constituted 5.6% of the recovered radiolabel. The second important biodegradation step starts with hydroxylation of the imidazolidine ring at the 4 or 5 position, and about 16% of the recovered radiolabel was identified as the sum of 4- and 5-hydroxy imidacloprid. The loss of water yields the olefinic compound. These biotransformation products and the unchanged parent compound were excreted in urine and feces, while the guanidine compound was a less important metabolite and was eliminated only in feces (A623). |
| Toxicity/Toxicokinetics |
Toxicity Summary Imidacloprid acts on the nicotinic acetylcholine receptor; the chlorination inhibits degradation by acetylcholine-esterase (L1130). Toxicity Data LD50: 450 mg/kg (Oral, Rat) (L1130) LD50: 131 mg/kg (Oral, Mouse) (L1130) LD50: >5000 mg/kg (Dermal, Rat) (L1130) LD50: 69 mg/m3 (Inhalation (aerosol), Rat) (L1130) LD50: 5 323 mg/m3 (Inhalation (dust), Rat) (L1130) |
| References |
[1]. Imidacloprid, a neonicotinoid insecticide, induces insulin resistance. J Toxicol Sci. 2013;38(5):655-60. [2]. Insecticide imidacloprid influences cognitive functions and alters learning performance and related gene expression in a rat model. Int J Exp Pathol. 2015 Oct;96(5):332-7. [3]. Neurobehavioral impairments caused by developmental imidacloprid exposure in zebrafish. Neurotoxicol Teratol. 2015 May-Jun;49:81-90. [4]. A 90 days oral toxicity of imidacloprid in female rats: morphological, biochemical and histopathological evaluations. Food Chem Toxicol. 2010 May;48(5):1185-90. [5]. Effect of imidacloprid on antioxidant enzymes and lipid peroxidation in female rats to derive its No Observed Effect Level (NOEL). J Toxicol Sci. 2010 Aug;35(4):577-81. [6]. Immunotoxic effects of imidacloprid following 28 days of oral exposure in BALB/c mice. Environ Toxicol Pharmacol. 2013 May;35(3):408-18. |
| Additional Infomation |
2-Imidazolidinimine, 1-[(6-chloro-3-pyridinyl)methyl]-N-nitro- has been reported in Streptomyces canus and Ganoderma lucidum with data available. Imidacloprid is a neonicotinoid insecticide, which is a class of neuro-active insecticides modeled after nicotine. Nicotine was identified and used as an insecticide and rat poison as early as the 1600’s. Its effectiveness as an insecticide spurred a search for insecticidal compounds that have selectively less effect on mammals, which led to the discovery of neonicotinoids. Neonicotinoids, like nicotine, bind to nicotinic acetylcholine receptors of a cell. In mammals, nicotinic acetylcholine receptors are located in cells of both the central and peripheral nervous systems. In insects these receptors are limited to the CNS. While low to moderate activation of these receptors causes nervous stimulation, high levels overstimulate and block the receptors causing paralysis and death. Nicotinic acetylcholine receptors are activated by the neurotransmitter acetylcholine. Acetylcholine is broken down by acetylcholinesterase to terminate signals from these receptors. However, acetylcholinesterase cannot break down neonicotinoids and the binding is irreversible. Because most neonicotinoids bind much more strongly to insect neuron receptors than to mammal neuron receptors, these insecticides are selectively more toxic to insects than mammals. The low mammalian toxicity of neonicotinoids can be explained in large part by their lack of a charged nitrogen atom at physiological pH. The uncharged molecule can penetrate the insect blood–brain barrier, while the mammalian blood–brain barrier filters it. However, Some neonicotinoid breakdown products are toxic to humans, especially if they have become charged. Because of their low toxicity and other favorable features, neonicotinoids are among the most widely used insecticides in the world. Most neonicotinoids are water-soluble and break down slowly in the environment, so they can be taken up by the plant and provide protection from insects as the plant grows. Neonicotinoids are currently used on corn, canola, cotton, sorghum, sugar beets and soybeans. They are also used on the vast majority of fruit and vegetable crops, including apples, cherries, peaches, oranges, berries, leafy greens, tomatoes, and potatoes. The use of neonicotinoids has been linked in a range of studies to adverse ecological effects, including honey-bee colony collapse disorder (CCD) and loss of birds due to a reduction in insect populations. This has led to moratoriums and bans on their use in Europe. See also: Imidacloprid (preferred); Imidacloprid; Moxidectin (component of); Imidacloprid; Ivermectin (component of). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.9114 mL | 19.5572 mL | 39.1144 mL | |
| 5 mM | 0.7823 mL | 3.9114 mL | 7.8229 mL | |
| 10 mM | 0.3911 mL | 1.9557 mL | 3.9114 mL |