Physicochemical Properties
| Molecular Formula | C26H27NO9 |
| Exact Mass | 497.168 |
| CAS # | 58957-92-9 |
| Related CAS # | Idarubicin hydrochloride;57852-57-0 |
| PubChem CID | 42890 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 725.4±60.0 °C at 760 mmHg |
| Flash Point | 392.5±32.9 °C |
| Vapour Pressure | 0.0±2.5 mmHg at 25°C |
| Index of Refraction | 1.706 |
| LogP | 2.95 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 36 |
| Complexity | 912 |
| Defined Atom Stereocenter Count | 6 |
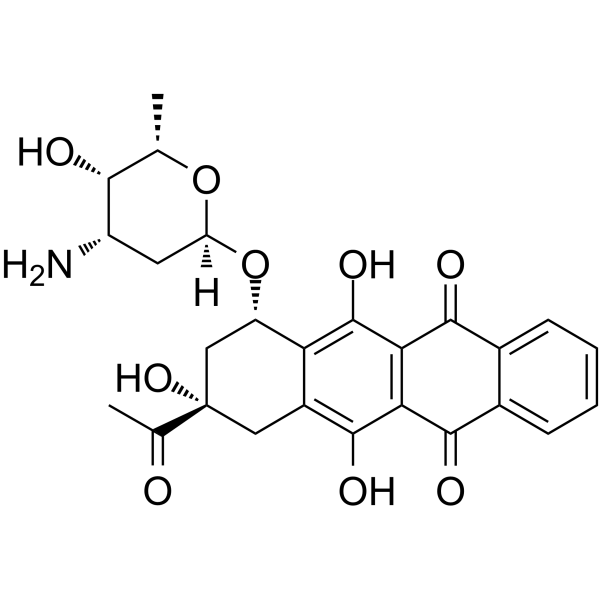
| SMILES | C[C@H]1[C@H]([C@H](C[C@@H](O1)O[C@H]2C[C@@](CC3=C(C4=C(C(=O)C5=CC=CC=C5C4=O)C(=C32)O)O)(C(=O)C)O)N)O |
| InChi Key | XDXDZDZNSLXDNA-TZNDIEGXSA-N |
| InChi Code | InChI=1S/C26H27NO9/c1-10-21(29)15(27)7-17(35-10)36-16-9-26(34,11(2)28)8-14-18(16)25(33)20-19(24(14)32)22(30)12-5-3-4-6-13(12)23(20)31/h3-6,10,15-17,21,29,32-34H,7-9,27H2,1-2H3/t10-,15-,16-,17-,21+,26-/m0/s1 |
| Chemical Name | (7S,9S)-9-acetyl-7-[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-8,10-dihydro-7H-tetracene-5,12-dione |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Idarubicin's half-life (IC50) in MCF-7 cell monolayers is 3.3 ± 0.4 ng/mL, but in multicellular spheroids, it is 7.9 ± 1.1 ng/mL [1]. In a number of in vitro systems, it was found that idarubicin was more cytotoxic than either daunorubicin or doxorubicin. This is explained by idarubicin's superior capacity to trigger topoisomerase II-mediated DNA break formation [2]. Idarubicin's activity is roughly 25 times higher than that of epirubicin and doxorubicin's, respectively, at 57.5 times [3]. With an IC50 of roughly 0.01 μM, idarubicin inhibits MCF-7 cell proliferation in a concentration-dependent manner. Idarubicin inhibits c-myc expression in a concentration-dependent manner as well as in a time- and concentration-dependent manner [4]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion The drug is eliminated predominately by biliary and to a lesser extent by renal excretion, mostly in the form of idarubicinol. Biological Half-Life 22 hours |
| Toxicity/Toxicokinetics |
Protein Binding 97% |
| References |
[1]. Idarubicin and idarubicinol effects on breast cancer multicellular spheroids. J Chemother. 2005 Dec;17(6):663-7. [2]. Robert J. Clinical pharmacokinetics of idarubicin. Clin Pharmacokinet. 1993 Apr;24(4):275-88. [3]. Enhanced in vitro cytotoxicity of idarubicin compared to epirubicin and doxorubicin in rat prostate carcinoma cells. Eur Urol. 1997;31(3):365-70. [4]. Induction of DNA damage, inhibition of DNA synthesis and suppression of c-myc expression by the anthracycline analog, idarubicin (4-demethoxy-daunorubicin) in the MCF-7 breast tumor cell line. Cancer Chemother Pharmacol. 1998;41(5):361-. [5]. Idarubicin inhibits the growth of bacteria and yeasts in an automated blood culture system. Eur J Clin Microbiol Infect Dis. 2009 Mar;28(3):301-3. |
| Additional Infomation |
Idarubicin is a monosaccharide derivative, an anthracycline antibiotic and a deoxy hexoside. It derives from a hydride of a tetracene. An orally administered anthracycline antineoplastic. The compound has shown activity against breast cancer, lymphomas and leukemias, together with the potential for reduced cardiac toxicity. Idarubicin is an Anthracycline Topoisomerase Inhibitor. The mechanism of action of idarubicin is as a Topoisomerase Inhibitor. Idarubicin is a semisynthetic 4-demethoxy analogue of the antineoplastic anthracycline antibiotic daunorubicin. Idarubicin intercalates into DNA and interferes with the activity of topoisomerase II, thereby inhibiting DNA replication, RNA transcription and protein synthesis. Due to its high lipophilicity, idarubicin penetrates cell membranes more efficiently than other anthracycline antibiotic compounds. An orally administered anthracycline antineoplastic. The compound has shown activity against BREAST NEOPLASMS; LYMPHOMA; and LEUKEMIA. See also: Idarubicin Hydrochloride (has salt form). Drug Indication For the treatment of acute myeloid leukemia (AML) in adults. This includes French-American-British (FAB) classifications M1 through M7. Mechanism of Action Idarubicin has antimitotic and cytotoxic activity through a number of proposed mechanisms of action: Idarubicin forms complexes with DNA by intercalation between base pairs, and it inhibits topoisomerase II activity by stabilizing the DNA-topoisomerase II complex, preventing the religation portion of the ligation-religation reaction that topoisomerase II catalyzes. Pharmacodynamics Idarubicin is an antineoplastic in the anthracycline class. General properties of drugs in this class include: interaction with DNA in a variety of different ways including intercalation (squeezing between the base pairs), DNA strand breakage and inhibition with the enzyme topoisomerase II. Most of these compounds have been isolated from natural sources and antibiotics. However, they lack the specificity of the antimicrobial antibiotics and thus produce significant toxicity. The anthracyclines are among the most important antitumor drugs available. Doxorubicin is widely used for the treatment of several solid tumors while daunorubicin and idarubicin are used exclusively for the treatment of leukemia. Idarubicin may also inhibit polymerase activity, affect regulation of gene expression, and produce free radical damage to DNA. Idarubicin possesses an antitumor effect against a wide spectrum of tumors, either grafted or spontaneous. The anthracyclines are cell cycle-nonspecific. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |