Icilin (also known as AG-3-5 or AG 3-5) is a synthetic CMR1/TRPM8 (transient receptor potential M8) super-cooling agent/agonist that is 2.5-fold more efficacious and nearly 200-fold more potent than the reference cold thermosensory agonist, l-menthol. Icilin induces sensations of intense cold when applied orally in humans, and induces 'wet dog shakes', a behavioral marker of cold sensation, when given to rats. Icilin should serve as the reference cold nociceptive agonist For TRP-type ion channels in the future. Icilin down-regulated the expression of cell cycle signature genes and caused G1 arrest in PC-3 prostate cancer cells. Icilin inactivates a small regulatory module controlling the cell cycle in prostate cancer cells. This study might provide insight into the development of cell cycle-targeted cancer therapeutics.
Physicochemical Properties
| Molecular Formula | C16H13N3O4 | |
| Molecular Weight | 311.29 | |
| Exact Mass | 311.09 | |
| Elemental Analysis | C, 61.73; H, 4.21; N, 13.50; O, 20.56 | |
| CAS # | 36945-98-9 | |
| Related CAS # |
|
|
| PubChem CID | 161930 | |
| Appearance | Typically exists as Light yellow to yellow solids at room temperature | |
| Density | 1.4±0.1 g/cm3 | |
| Boiling Point | 594.8±50.0 °C at 760 mmHg | |
| Melting Point | 200-208ºC | |
| Flash Point | 313.5±30.1 °C | |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C | |
| Index of Refraction | 1.672 | |
| LogP | 2.73 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 2 | |
| Heavy Atom Count | 23 | |
| Complexity | 502 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O=C1N([H])C(C2C([H])=C([H])C([H])=C(C=2[H])[N+](=O)[O-])=C([H])C([H])([H])N1C1=C([H])C([H])=C([H])C([H])=C1O[H] |
|
| InChi Key | RCEFMOGVOYEGJN-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C16H13N3O4/c20-15-7-2-1-6-14(15)18-9-8-13(17-16(18)21)11-4-3-5-12(10-11)19(22)23/h1-8,10,20H,9H2,(H,17,21) | |
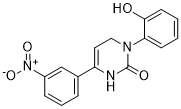
| Chemical Name | 3-(2-hydroxyphenyl)-6-(3-nitrophenyl)-1,4-dihydropyrimidin-2-one | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
TRPM8/transient receptor potential M8
Icilin is a synthetic super-cooling agent that activates TRPM8 (transient receptor potential melastatin 8) cation channel [1] Icilin exerts anti-inflammatory effects on CD4⁺ T cells independent of TRPM8 and TRPA1 (transient receptor potential ankyrin-1) channels[3] |
| ln Vitro |
Icilin-evoked TRPM8 currents demonstrate very varied latencies with substantial desensitization. They also have variable activation kinetics and Ca2+ dependence[1]. 1. Icilin activates TRPM8 with extremely variable latency followed by extensive desensitization in the presence of calcium; full efficacy of Icilin requires simultaneous elevation of cytosolic Ca²⁺ (either via permeation through TRPM8 channels or release from intracellular stores), while menthol/cold activate TRPM8 with rapid activation and moderate Ca²⁺-dependent adaptation [1] 2. Icilin potently inhibits the proliferation of murine and human CD4⁺ T cells in vitro; attenuation of TRPM8⁻/⁻ and TRPA1⁻/⁻ T-cell proliferation by Icilin is consistent with wild-type (WT) phenotype, indicating a TRPM8/TRPA1-independent mechanism [3] 3. Icilin alters the expressional profile of activated CD4⁺ T cells in vitro, leading to a phenotype indicative of restricted effector function and limited neuroinflammatory potential [3] |
| ln Vivo |
Rats and mice are behaviorally activated by iscilin, a transient receptor potential cation channel subfamily M (TRPM8) agonist. Mice are given the "Wet dog shake" when given icin (3 mg/kg; sc)[2]. 1. Icilin treatment protects both WT and TRPM8-deficient mice from disease progression in experimental autoimmune encephalomyelitis (EAE, a murine model of multiple sclerosis): delays in clinical onset and reductions in neuroinflammation were observed; peripheral expansion of autoantigen-restricted T cells was diminished by Icilin administration in mice [3] |
| Enzyme Assay |
TRPM8, a member of the transient receptor potential family of ion channels, depolarizes somatosensory neurons in response to cold. TRPM8 is also activated by the cooling agents menthol and icilin. When exposed to menthol or cold, TRPM8 behaves like many ligand-gated channels, exhibiting rapid activation followed by moderate Ca(2+)-dependent adaptation. In contrast, icilin activates TRPM8 with extremely variable latency followed by extensive desensitization, provided that calcium is present. Here, we show that, to achieve full efficacy, icilin requires simultaneous elevation of cytosolic Ca2+, either via permeation through TRPM8 channels or by release from intracellular stores. Thus, two stimuli must be paired to elicit full channel activation, illustrating the potential for coincidence detection by TRP channels. Determinants of icilin sensitivity map to a region of TRPM8 that corresponds to the capsaicin binding site on the noxious heat receptor TRPV1, suggesting a conserved molecular logic for gating of these thermosensitive channels by chemical agonists[1]. 1. TRPM8 channel activity assay (patch-clamp technique): TRPM8-expressing cells were exposed to Icilin, menthol, or cold stimuli; ion channel gating, activation latency, desensitization, and Ca²⁺ dependence were measured by recording membrane potentials and ion currents. The effects of cytosolic Ca²⁺ elevation (via TRPM8 permeation or intracellular store release) on Icilin-mediated TRPM8 activation were evaluated, and the sensitivity determinants of Icilin on TRPM8 were mapped to a region homologous to the capsaicin binding site on TRPV1 [1] |
| Cell Assay |
Cell Viability Assay[1] Cell Types: TRPM8-expressing oocytes or HEK293 cells Tested Concentrations: 10 μM Incubation Duration: 3 minutes for TRPM8-expressing oocytes; 1 minute for HEK293 cells Experimental Results: Activated membrane currents with variable delay of onset in voltage-clamped TRPM8-expressing oocytes or HEK293 cells. 1. TRPM8 channel functional assay: Cells expressing TRPM8 were treated with Icilin (concentration not specified), menthol, or cold; Ca²⁺ signaling, ion channel activation/desensitization kinetics were assessed using patch-clamp techniques and Ca²⁺ imaging. Chelating agents (e.g., EGTA) were used to manipulate cytosolic Ca²⁺ levels to evaluate the role of Ca²⁺ in Icilin-mediated TRPM8 activation [1] 2. T-cell proliferation assay: Murine and human CD4⁺ T cells (including TRPM8⁻/⁻ and TRPA1⁻/⁻ knockout cells) were isolated and activated in vitro; cells were treated with Icilin (concentration not specified), and proliferation was measured by standard methods (e.g., [³H]thymidine incorporation or CFSE dilution, method not explicitly stated). The proliferation rate of WT and knockout T cells was compared to determine the TRPM8/TRPA1 dependence of Icilin’s effect [3] 3. T-cell gene expression assay: Activated CD4⁺ T cells were treated with Icilin in vitro; RNA was extracted, and gene expression profiles were analyzed to identify changes in effector function-related genes [3] |
| Animal Protocol |
Animal/Disease Models: The C57BL/ 6 mice 9–10 weeks (adult, 26-30 g) or 24 months (aged, 35-42g)[2]. Doses: 3 mg/kg Route of Administration: Injected sc Experimental Results: Produced vivid and quantifiable shaking behaviors (“wet- dog shakes”), which were TRPM8-dependent. To determine if “compound 5” effectively inhibits TRPM8 activity in live mice, a WDS assay was performed. In this assay, a potent TRPM8 agonist (icilin 3 mg/kg s.c.) was injected in mice treated with “compound 5” or vehicle. Icilin injections in mice produce vivid and quantifiable shaking behaviors (“wet-dog shakes”), which are TRPM8-dependent (18–22)(2, 3). Accordingly, effective inhibition of TRPM8 is predicted to result in decreased number of icilin-induced behavioral events. “Compound 5” or vehicle was administered 60 minutes prior to icilin. The number of “wet-dog shakes” was counted over a 5-minute period starting 10 minutes after icilin injection.Experimental protocols (summarized in Table 1): In protocol 1, used to establish a method for long-lasting and sustained hypothermia through TRPV1 activation, DHC (2–4 mg/kg) or vehicle (20% DMSO in saline) was infused subcutaneously through the PE-10 line in a conscious mouse. The full dose was delivered in two bolus injections 30 minutes apart (Figure 1B) or as continuous infusion with or without an initial bolus injection (Figure 2). Core temperature was recorded by the thermocouple implanted in the abdomen; a second thermocouple recorded cage ambient temperature (22–24 °C). Temperature was measured at a frequency of 1 sample/min for up to 8 hours after the first infusion.[2] Protocol 3 was used to determine the effectiveness of TRPV1 agonists in aged subjects. Young (9–10 weeks) or aged (24 months) mice were injected with a single bolus of DHC (1.25 mg/kg s.c.). Temperature was measured by telemeter for 2 hours after injection (Figure 4). In protocol 4, used to determine the hypothermic effect of TRPM8 inhibition in mildly subneutral and cold environments, “compound 5” (20 mg/kg s.c.) or vehicle was administered to telemeter implanted mice (Figure 5). “Compound 5” is a selective TRPM8 inhibitor (see Drugs section). Core temperature was recorded for 30 minutes starting 30 minutes after the injection, at which point the mice were transferred to a cold room for 2 hours and measured at 20 minute intervals. The individual cages were fitted with a plastic mesh top to allow heat exchange with the atmosphere. Temperature in the cage averaged 8 °C.[2] Protocol 5 was used to determine if TRPM8 inhibition potentiates the hypothermic effect of TRPV1 agonists. TRPM8 antagonist “compound 5” (30 mg/kg i.p.) or vehicle was injected in mice with implanted telemeter (Figure 6). After 60 minutes, DHC (0.6, 1.25, or 2.5 mg/kg i.p.) was injected in both groups. Temperature was recorded for 5.5 hours (330 minutes) from the time of “compound 5” or vehicle injection, with a measurement taken every 10 minutes. The depth of hypothermia was determined by calculating the minimum value (nadir) of Tcore with each treatment. The duration of hypothermia was determined by measuring the time from DHC injection until recovery of Tcore to ≥34 °C to the nearest 10 minutes. The 0.6 mg/kg DHC dose failed to reliably drop Tcore below 34 °C during the experiment and was therefore excluded from Tcore recovery analysis. In the two groups treated with higher doses of DHC and pretreated with “compound 5”, the Tcore occasionally failed to return to 34 °C within the 330 minute measurement period. In these cases (6 out of 16 mice), the maximum time value (330 min) was assigned for the recovery time. All mice, however, fully recovered Tcore by the next morning. In addition, in a single mouse in the vehicle group treated with 2.5 mg/kg DHC, Tcore did not drop below 34 °C. This mouse was assigned the minimum time value (0 minutes) for the recovery time.[2] 1. EAE mouse model protocol: Female C57BL/6 mice (WT, TRPM8⁻/⁻) were induced with EAE (induction method not specified); Icilin was administered (route, dose, frequency not specified) during EAE progression. Clinical scores of EAE (disease onset and severity) were monitored, and neuroinflammation was evaluated by histological or molecular methods (not explicitly stated). Peripheral T-cell expansion (autoantigen-restricted) was assessed by flow cytometry or immunological assays (not explicitly stated) [3] |
| References |
[1]. The super-cooling agent icilin reveals a mechanism of coincidence detection by a temperature-sensitive TRP channel. Neuron. 2004 Sep 16;43(6):859-69. [2]. Transient receptor potential melastatin 8 channel inhibition potentiates the hypothermic response to transient receptor potential vanilloid 1 activation in the conscious mouse. Crit Care Med. 2014 May;42(5):e355-63. [3]. The cooling compound icilin attenuates autoimmune neuroinflammation through modulation of the T-cell response. FASEB J. 2018 Mar;32(3):1236-1249. |
| Additional Infomation |
3-(2-hydroxyphenyl)-6-(3-nitrophenyl)-1,4-dihydropyrimidin-2-one is a C-nitro compound. 1. Icilin is a synthetic super-cooling agent; its activation of TRPM8 requires coincidence detection of two stimuli (icilin binding and cytosolic Ca²⁺ elevation), illustrating a unique mechanism of TRP channel gating. The Icilin sensitivity region on TRPM8 is homologous to the capsaicin binding site on TRPV1, suggesting conserved molecular logic for thermosensitive TRP channel gating by chemical agonists [1] 2. Icilin exerts potent anti-inflammatory effects in lymphocyte-mediated neuroinflammation (EAE model), with pleiotropic effects beyond classic TRPM8 channel activation (TRPM8/TRPA1-independent mechanism in T-cell modulation) [3] 3. Icilin is a potential therapeutic agent for autoimmune neuroinflammatory disorders (e.g., multiple sclerosis) due to its ability to attenuate EAE progression by modulating CD4⁺ T-cell responses [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.03 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2124 mL | 16.0622 mL | 32.1244 mL | |
| 5 mM | 0.6425 mL | 3.2124 mL | 6.4249 mL | |
| 10 mM | 0.3212 mL | 1.6062 mL | 3.2124 mL |