Physicochemical Properties
| Molecular Formula | C59H89N19O13S |
| Molecular Weight | 1304.5225 |
| Exact Mass | 1303.66 |
| Elemental Analysis | C, 54.32; H, 6.88; N, 20.40; O, 15.94; S, 2.46 |
| CAS # | 130308-48-4 |
| Related CAS # | Icatibant acetate; 138614-30-9 |
| PubChem CID | 6918173 |
| Appearance | White to off-white solid powder |
| Density | 1.6±0.1 g/cm3 |
| Index of Refraction | 1.742 |
| LogP | -2.63 |
| Hydrogen Bond Donor Count | 15 |
| Hydrogen Bond Acceptor Count | 18 |
| Rotatable Bond Count | 30 |
| Heavy Atom Count | 92 |
| Complexity | 2720 |
| Defined Atom Stereocenter Count | 12 |
| SMILES | S1C([H])=C([H])C([H])=C1C([H])([H])[C@@]([H])(C(N([H])[C@@]([H])(C([H])([H])O[H])C(N1C([H])([H])C2=C([H])C([H])=C([H])C([H])=C2C([H])([H])[C@]1([H])C(N1C([H])(C(N([H])[C@]([H])(C(=O)O[H])C([H])([H])C([H])([H])C([H])([H])/N=C(\N([H])[H])/N([H])[H])=O)C([H])([H])[C@]2([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[C@]12[H])=O)=O)=O)N([H])C(C([H])([H])N([H])C([C@]1([H])C([H])([H])[C@]([H])(C([H])([H])N1C([C@]1([H])C([H])([H])C([H])([H])C([H])([H])N1C([C@]([H])(C([H])([H])C([H])([H])C([H])([H])/N=C(\N([H])[H])/N([H])[H])N([H])C([C@@]([H])(C([H])([H])C([H])([H])C([H])([H])/N=C(\N([H])[H])/N([H])[H])N([H])[H])=O)=O)=O)O[H])=O)=O |
| InChi Key | QURWXBZNHXJZBE-SKXRKSCCSA-N |
| InChi Code | InChI=1S/C59H89N19O13S/c60-37(14-5-19-67-57(61)62)48(82)72-38(15-6-20-68-58(63)64)52(86)75-22-8-18-43(75)54(88)77-30-35(80)26-44(77)50(84)70-28-47(81)71-40(27-36-13-9-23-92-36)49(83)74-41(31-79)53(87)76-29-34-12-2-1-10-32(34)24-46(76)55(89)78-42-17-4-3-11-33(42)25-45(78)51(85)73-39(56(90)91)16-7-21-69-59(65)66/h1-2,9-10,12-13,23,33,35,37-46,79-80H,3-8,11,14-22,24-31,60H2,(H,70,84)(H,71,81)(H,72,82)(H,73,85)(H,74,83)(H,90,91)(H4,61,62,67)(H4,63,64,68)(H4,65,66,69)/t33-,35+,37+,38-,39-,40-,41-,42-,43-,44-,45-,46+/m0/s1 |
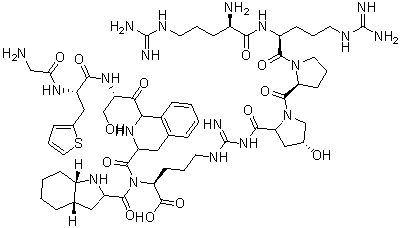
| Chemical Name | (2S)-2-[[(2S,3aS,7aS)-1-[(3R)-2-[(2S)-2-[[(2S)-2-[[2-[[(2S,4R)-1-[(2S)-1-[(2S)-2-[[(2R)-2-amino-5-(diaminomethylideneamino)pentanoyl]amino]-5-(diaminomethylideneamino)pentanoyl]pyrrolidine-2-carbonyl]-4-hydroxypyrrolidine-2-carbonyl]amino]acetyl]amino]-3-thiophen-2-ylpropanoyl]amino]-3-hydroxypropanoyl]-3,4-dihydro-1H-isoquinoline-3-carbonyl]-2,3,3a,4,5,6,7,7a-octahydroindole-2-carbonyl]amino]-5-(diaminomethylideneamino)pentanoic acid |
| Synonyms | Firazyr; Icatibant |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Bradykinin B2 Receptor (B2R) |
| ln Vitro | Icatibant (10–30 μM) increases the effects of angiotensin III, but not II (contraction mediated by angiotensin AT1 receptors), and Lys-des-Arg9-bradykinin, but not des-Arg9-bradykinin (effects mediated by the bradykinin B1 receptors) (see reference 3). |
| ln Vivo | Icatibant (0.3, or 1.5 mg/kg, subcutaneous administration twice daily in mice) significantly reduces the risk of ulcerative colitis[2]. |
| Animal Protocol |
Female mice of the CBA/J (H-2k) strain 0.06, 0.3, or 1.5 mg/kg. Subcutaneous administration twice daily |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion The absolute bioavailability of icatibant following a 30 mg subcutaneous dose is approximately 97%. Maximum plasma concentrations (Cmax) of 974 ± 280 ng/mL were reached when a single subcutaneous dose of 30 mg was administered. The AUC was 2165 ± 568 ng∙hr/mL. Icatibant did not accumulate following multiple doses. The pharmacokinetics of icatibant have been characterized in studies using both intravenous and subcutaneous administration to healthy subjects and patients. The pharmacokinetic profile of icatibant in patients with HAE is similar to that in healthy subjects. The absolute bioavailability of icatibant following a 30 mg subcutaneous dose is approximately 97%. Following subcutaneous administration of a single 30 mg dose of icatibant to healthy subjects (N=96), a mean (± standard deviation) maximum plasma concentration (Cmax) of 974 ± 280 ng/mL was observed after approximately 0.75 hours. The mean area under the concentration-time curve (AUC0-∞) after a single 30 mg dose was 2165 ± 568 ng·hr/mL, with no evidence of accumulation of icatibant following three 30 mg doses administered 6 hours apart. Icatibant's inactive metabolites are primarily excreted in the urine, with less than 10% of the dose eliminated as unchanged drug. Following subcutaneous administration, d volume of distribution at steady state (Vss) was 29.0 ± 8.7 L. Following subcutaneous administration, plasma clearance was 245 ± 58 mL/min. Metabolism / Metabolites Icatibant is metabolized by proteolytic enzymes into inactive metabolites. The cytochrome P450 enzyme system is not involved with the metabolism of icatibant. Biological Half-Life Following subcutaneous administration, mean elimination half-life was 1.4 ± 0.4 hours. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the excretion of icatibant into breastmilk. Because icatibant is a protein molecule with a molecular weight of 1305 Da, the amount in milk is likely to be very low. It is also likely to be partially destroyed in the infant's gastrointestinal tract and absorption by the infant is probably minimal. One patient reportedly used the drug safely during breastfeeding. Waiting 6 hours after a dose before breastfeeding should minimize the amount of drug excreted into breastmilk. ◉ Effects in Breastfed Infants A woman with hereditary angioedema began using icatibant 30 mg subcutaneously as needed for hereditary angioedema attacks when her breastfed infant was 4 months of age and continued breastfeeding for 1 year while taking icatibant. Doses were injected at night before the infant’s longest sleep period and breastfeeding was not resumed until at least 6 hours after a dose. In cases of swelling of the face and neck and abdominal pain, icatibant was immediately self-administered, and formula was given instead of breastmilk. In her second pregnancy 2 years later, she used C1 esterase inhibitor until the infant was 1 month of age, when she resumed icatibant therapy. . ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding No information is available on the protein binding of icatibant |
| References |
[1]. Hoe 140 a new potent and long acting bradykinin-antagonist: in vitro studies. Br J Pharmacol. 1991 Mar;102(3):769-73. [2]. Effect of Icatibant, a Bradykinin B2 Receptor Antagonist, on the Development of Experimental Ulcerative Colitis in Mice. Dig Dis Sci. 1999 Apr;44(4):845-51. [3]. The Bradykinin B2 Receptor Antagonist Icatibant (Hoe 140) Blocks Aminopeptidase N at Micromolar Concentrations: Off-Target Alterations of Signaling Mediated by the Bradykinin B1 and Angiotensin Receptors. Eur J Pharmacol. 2006 Dec 3;551(1-3):108-11. |
| Additional Infomation |
Icatibant is a synthetic decapeptide with 5 nonproteinogenic amino acid antagonist targeting the B2 receptors with a similar affinity to bradykinin. It is resistant to bradykinin-cleaving enzyme degradation and has a potency of 2-3 times higher than earlier B2 receptors antagonists, thus representing a new class of medication. It was investigated as a potential treatment of hereditary angioedema (HAE) as bradykinin was implicated in HAE swelling; specifically, mice lacking B2 receptors showed reduced swelling, thus demonstrating bradykinin involvement in the disease pathophysiology. Icatibant was approved by the FDA on August 25, 2011, and by the EMA in 2008 as a treatment for hereditary angioedema. The FDA approval was based on positive results obtained from 3 double-blind, randomized, controlled clinical trials known as FAST 1, 2, and 3, where a median time to almost complete symptom relief was observed to be 8 hours compared to 36 hours for the placebo treatment. Icatibant is a Bradykinin B2 Receptor Antagonist. The mechanism of action of icatibant is as a Bradykinin B2 Receptor Antagonist. Icatibant is an antagonist of the human bradykinin B2 receptor (B2R), that can be used for the treatment of hereditary angioedema (HAE). Upon administration, icatibant targets and binds to B2R, thereby preventing bradykinin from binding to the B2R. This may prevent bradykinin/B2R-mediated vasodilation, the resulting increase in vascular permeability, and the swelling, inflammation, and pain associated with HAE. This may also prevent or improve pulmonary edema not associated with HAE and improve the associated decrease in blood oxygen levels. See also: Icatibant Acetate (has salt form). Drug Indication Icatibant is indicated for the treatment of acute attacks of hereditary angioedema (HAE) in adults 18 years of age and older. FDA Label Firazyr is indicated for symptomatic treatment of acute attacks of hereditary angioedema (HAE) in adults (with C1-esterase-inhibitor deficiency). Icatibant Accord is indicated for symptomatic treatment of acute attacks of hereditary angioedema (HAE) in adults, adolescents and children aged 2 years and older, with C1 esterase inhibitor deficiency. Treatment of ACE inhibitor-induced angioedema Mechanism of Action Icatibant is a competitive antagonist selective for the bradykinin B2 receptor, with an affinity similar to bradykinin. Hereditary angioedema is caused by an absence or dysfunction of C1-esterase-inhibitor, a key regulator of the Factor XII/kallikrein proteolytic cascade that leads to bradykinin production. Bradykinin is a vasodilator thought to be responsible for the characteristic HAE symptoms of localized swelling, inflammation, and pain. Icatibant inhibits bradykinin from binding to the B2 receptor, thereby treating the clinical symptoms of an acute, episodic attack of HAE. Pharmacodynamics Following bradykinin challenge, intravenous administration of icatibant caused dose and time-dependent inhibition of the development of bradykinin-induced hypotension, vasodilation, and reflex tachycardia in healthy young subjects. Icatibant intravenous doses of 0.4 and 0.8 mg/kg infused over 4 hours inhibited response to bradykinin challenge for 6 to 8 hours following completion of the infusion. Based on exposure-response analysis, a subcutaneous dose of 30 mg icatibant is predicted to be effective against bradykinin challenge for at least 6 hours. The clinical significance of these findings is unknown. The effect of icatibant 30 and 90 mg following a single subcutaneous injection on QTc interval was evaluated in a randomized, placebo-, and active-controlled (moxifloxacin 400 mg) four-period crossover thorough QT study in 72 healthy subjects. In a study with demonstrated ability to detect small effects, the upper bound of the one-sided 95% confidence interval for the largest placebo-adjusted, baseline-corrected QTc based on individual correction method (QTcI) was below 10 ms, the threshold for regulatory concern. The dose of 90 mg is adequate to represent the high-exposure clinical scenario. |
Solubility Data
| Solubility (In Vitro) | H2O: ~100 mg/mL (~76.7 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 50 mg/mL (38.33 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.7666 mL | 3.8328 mL | 7.6657 mL | |
| 5 mM | 0.1533 mL | 0.7666 mL | 1.5331 mL | |
| 10 mM | 0.0767 mL | 0.3833 mL | 0.7666 mL |