Physicochemical Properties
| Molecular Formula | C12H13NO2S |
| Molecular Weight | 235.302 |
| Exact Mass | 235.067 |
| CAS # | 1169755-45-6 |
| Related CAS # | 1169755-45-6; |
| PubChem CID | 44136031 |
| Appearance | Off-white to light yellow solid powder |
| LogP | 2.078 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 16 |
| Complexity | 316 |
| Defined Atom Stereocenter Count | 0 |
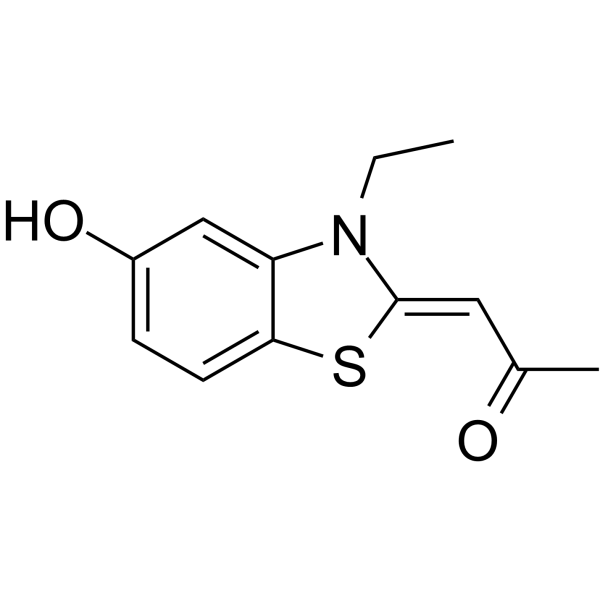
| SMILES | CCN\1C2=C(C=CC(=C2)O)S/C1=C\C(=O)C |
| InChi Key | GCSZJMUFYOAHFY-SDQBBNPISA-N |
| InChi Code | InChI=1S/C12H13NO2S/c1-3-13-10-7-9(15)4-5-11(10)16-12(13)6-8(2)14/h4-7,15H,3H2,1-2H3/b12-6- |
| Chemical Name | (1Z)-1-(3-ethyl-5-hydroxy-1,3-benzothiazol-2-ylidene)propan-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | At 3 μM and 30 μM, INDY (0.3-30 μM; 20 hours) virtually totally blocks tau phosphorylation [1]. Nuclear factor of activated T cells, or NFAT, signaling is blocked when acid-(Y)-phosphorylation regulator 1A is overexpressed [1]. |
| ln Vivo | ProINDY (2.5 μM) significantly restores synchronized normal development in Xenopus laevis [1]. |
| Cell Assay |
Western Blot Analysis[1] Cell Types: COS7 cells transfected with EGFP-Dyrk1A and EGFP-tau Tested Concentrations: 0.3, 1, 3, 10, 30 μM Incubation Duration: 20 hrs (hours) Experimental Results: Mild inhibition of tau phosphorylation at 3 μM , almost complete inhibition at 30 μM. |
| References |
[1]. Development of a novel selective inhibitor of the Down syndrome-related kinase Dyrk1A. Nat Commun. 2010 Oct 5;1:86. [2]. Inhibition of DYRK1A destabilizes EGFR and reduces EGFR-dependent glioblastomagrowth. J Clin Invest. 2013 Jun;123(6):2475-87. |
| Additional Infomation | INDY is a member of the class of benzothiazoles that is 2,3-dihydro-1,3-benzothiazole substituted by 2-oxopropylidene, ethyl, and hydroxy groups at positions 2, 3 and 5, respectively. It is an ATP-competitive inhibitor of Dyrk1A and Dyrk1B (IC50 of 0.24 muM and 0.23 muM, respectively). It has a role as an antineoplastic agent, a drug metabolite and an EC 2.7.12.1 (dual-specificity kinase) inhibitor. It is a member of benzothiazoles, an organic hydroxy compound and an enone. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 33.33 mg/mL (~141.65 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (10.62 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.2499 mL | 21.2495 mL | 42.4989 mL | |
| 5 mM | 0.8500 mL | 4.2499 mL | 8.4998 mL | |
| 10 mM | 0.4250 mL | 2.1249 mL | 4.2499 mL |