Physicochemical Properties
| Molecular Formula | C25H27N7OS |
| Molecular Weight | 473.5931828022 |
| Exact Mass | 473.199 |
| CAS # | 2416844-79-4 |
| PubChem CID | 156760472 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 3.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 34 |
| Complexity | 715 |
| Defined Atom Stereocenter Count | 0 |
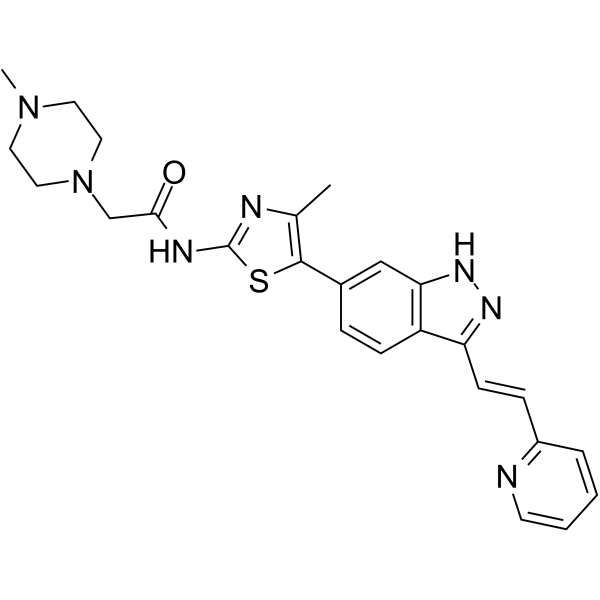
| SMILES | CC1=C(SC(=N1)NC(=O)CN2CCN(CC2)C)C3=CC4=C(C=C3)C(=NN4)/C=C/C5=CC=CC=N5 |
| InChi Key | CHLFFSKXUJQLQO-VQHVLOKHSA-N |
| InChi Code | InChI=1S/C25H27N7OS/c1-17-24(34-25(27-17)28-23(33)16-32-13-11-31(2)12-14-32)18-6-8-20-21(29-30-22(20)15-18)9-7-19-5-3-4-10-26-19/h3-10,15H,11-14,16H2,1-2H3,(H,29,30)(H,27,28,33)/b9-7+ |
| Chemical Name | 2-(4-methylpiperazin-1-yl)-N-[4-methyl-5-[3-[(E)-2-pyridin-2-ylethenyl]-1H-indazol-6-yl]-1,3-thiazol-2-yl]acetamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | IHMT-TRK-284 (Compound 34) exhibits antiproliferative effects on BaF3 cells, KM-12-LUC cells, and a panel of BaF3 cells that have undergone kinase transformation [1]. In KM-12-LUC cells, IHMT-TRK-284 (0-10 µM, 24 hours) can cause apoptosis and stop the cell cycle at the G0/G1 phase [1]. By specifically targeting TRK inhibition, IHMT-TRK-284 has an inhibitory effect on colon cancer cells [1]. Drug-resistant mutations such G667C/S in the DFG region and V573M and F589L in the ATP binding pocket can be defeated by IHMT-TRK-284 [1]. VEGFR2 kinase is selectively inhibited by IHMT-TRK-284 [1]. |
| ln Vivo | IHMT-TRK-284 (Compound 34) exhibits good in vivo PK and anti-tumor efficacious properties at oral doses of 40 and 80 mg/kg for ten days [1]. |
| Cell Assay |
Cell Proliferation Assay[1] Cell Types: BaF3 cells, BaF3 cells transformed with a panel of kinases, and KM-12-LUC Cells Tested Concentrations: 0-10 µM Incubation Duration: 72 hrs (hours) Experimental Results: Anti-proliferation demonstrated against BaF3 cells, a panel of kinases Kinase-transformed BaF3 cells and KM-12-LUC cells. The GI50 of KM-12-LUC cells is 0.002 µM. Antiproliferative effects of IHMT-TRK-284 on BaF3 cells transformed by a panel of kinases [1]. Target BaF3-TEL-ABL BaF3-TEL-CSF1R BaF3-TEL-KIT BaF3-TEL-PDGFRα BaF3-TEL-PDGFRβ BaF3-TEL-TRKA BaF3-TEL-TRKB BaF3-TEL-TRKC GI50 (nM) 411.1 4 923.2 1.7 1.4 8.5 8.2 27.3 Antiproliferative effect of IHMT-TRK-284 on a panel of BaF3 cells transformed with TRK wt/mutant (n = 3) [1]. Shows anti-proliferative effect on BaF3, BaF3-LMNA-, GI50 is 1.4 ± 0.011, 0.007 ± 0.001, 0.003 ± 0.001, 0.004 ± 0.001, 0.194 ± 0.013, 0.034 ± 0.002, 0.002 ± 0.001 μM TRKA, BaF3-LMNA- TRKA -V573M, BaF3-LMNA-TRKA-F589L, BaF3-LMNA-TRKA-G595R, BaF3-LMNA-TRKA-G667C, BaF3-LMNA-TRKA-G667S cell |
| Animal Protocol |
Animal/Disease Models: 4weeks old female nu/nu (nude) mice, 1 million BaF3-TELTRKA, BaF3-TEL-TRKB, BaF3-TEL-TRKC, BaF3-LMNA-TRKA-F589L, BaF3-LMNA-TRKA-G667S and 5 million A 1:1 mixture of KM-12-LUC cells and Matrigel in DMEM culture medium was prepared and injected into the right subcutaneousspace of nu/nu (nude) mice [1]. Doses: 40 mg/kg and 80 mg/kg. Method: Daily oral tube feeding for 10 days. Experimental Results: Dose-dependent inhibition of tumor progression of BaF3-TEL-TRKA, BaF3-TEL-TRKB and BaF3-TEL-TRKC, with TGI (tumor growth inhibition) of 68% and 93% respectively. and 58%. In a xenograft mouse model inoculated with KM-12-LUC cells, tumor progression was dose-dependently inhibited, with a TGI of 93% at 40 mg/kg/day and 95% at 80 mg/kg/day. At a dose of 80 mg/kg, it effectively inhibited tumor growth in BaF3-LMNA-TRKA-F589L and BaF3-LMNA-TRKA-G667S cells, with TGI values of 88% and 89% respectively. Animal/Disease Models: Mouse, Sprague Dawley Rat, and Beagle Dog [1] Doses: 1 mg/kg and 10 mg/kg Route of Administration: Intr |
| References |
[1]. Discovery of (E)-N-(4-methyl-5-(3-(2-(pyridin-2-yl)vinyl)-1H-indazol-6-yl)thiazol-2-yl)-2-(4-methylpiperazin-1-yl)acetamide (IHMT-TRK-284) as a novel orally available type II TRK kinase inhibitor capable of overcoming multiple resistan. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1115 mL | 10.5577 mL | 21.1153 mL | |
| 5 mM | 0.4223 mL | 2.1115 mL | 4.2231 mL | |
| 10 mM | 0.2112 mL | 1.0558 mL | 2.1115 mL |