ID-8 is a novel and potent small molecule DYRK (Dual-specificity tyrosine phosphorylation-regulated kinase) inhibitor that sustains embryonic stem cell self-renewal in long-term culture. ID-8 (0.5 μM) increased hESC survival by 1.1% in HES2 cells. Wnt3a and ID-8 together totally blocked Wnt-induced morphological differentiation and raised survival by 1.7%. Additionally, ID-8 dramatically decreased the expression of Wnt-induced differentiation marker genes GATA6, GSC, SOX17, and CDX2. By inhibiting DYRKs, ID-8 promoted Wnt-mediated hESC proliferation and survival in hESCs. Additionally, ID-8 directly targets the DYRK family.
Physicochemical Properties
| Molecular Formula | C16H14N2O4 | |
| Molecular Weight | 298.29 | |
| Exact Mass | 298.095 | |
| Elemental Analysis | C, 64.42; H, 4.73; N, 9.39; O, 21.45 | |
| CAS # | 147591-46-6 | |
| Related CAS # |
|
|
| PubChem CID | 791637 | |
| Appearance | Light yellow to yellow solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 418.3±45.0 °C at 760 mmHg | |
| Melting Point | 266 °C(dec.) | |
| Flash Point | 206.8±28.7 °C | |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C | |
| Index of Refraction | 1.638 | |
| LogP | 4.13 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 2 | |
| Heavy Atom Count | 22 | |
| Complexity | 405 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O([H])C1C([H])=C([H])C2C(=C(C([H])([H])[H])N(C3C([H])=C([H])C(=C([H])C=3[H])OC([H])([H])[H])C=2C=1[H])[N+](=O)[O-] |
|
| InChi Key | VVZNWYXIOADGSW-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C16H14N2O4/c1-10-16(18(20)21)14-8-5-12(19)9-15(14)17(10)11-3-6-13(22-2)7-4-11/h3-9,19H,1-2H3 | |
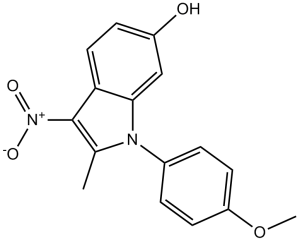
| Chemical Name | 1-(4-methoxyphenyl)-2-methyl-3-nitroindol-6-ol | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
DYRK ID-8 exhibits binding affinity to estrogen receptor β (ERβ) (Ki = 0.34 μM) and weak affinity to estrogen receptor α (ERα) (Ki = 12.5 μM) [1] ID-8 targets cellular oxidative stress pathways by modulating antioxidant enzyme activity [1,2] |
| ln Vitro |
ID-8 sustains Nanog gene expression through Sox2-Oct3/4 activation, reversibly maintaining the long-term culture of ESCs.[1] ID-8 inhibits DYRKs to improve Wnt-mediated hESC survival and proliferation. Based on the mechanism study, ID-8 enhances the CBP/β-catenin association in hESCs and modulates Wnt/β-catenin signaling to maintain the undifferentiated state in the presence of Wnt.[2] Treatment of RAW264.7 macrophages with ID-8 (1–50 μM) dose-dependently inhibited lipopolysaccharide (LPS)-induced nitric oxide (NO) production, with maximum inhibition (~70%) at 50 μM. It also reduced the expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) at the mRNA and protein levels via suppressing NF-κB activation [1] - ID-8 (10–100 μM) scavenged DPPH and ABTS free radicals in vitro, with scavenging rates of 52.3% and 68.7% at 100 μM, respectively. It also increased the activity of superoxide dismutase (SOD) and catalase (CAT) in H2O2-treated HepG2 cells, reducing intracellular reactive oxygen species (ROS) levels by ~45% at 50 μM [1] - In mouse bone marrow-derived mesenchymal stem cells (BMSCs), ID-8 (0.1–10 μM) promoted cell proliferation in a dose-dependent manner, with a 1.8-fold increase in cell number at 10 μM after 72 hours. It inhibited H2O2-induced apoptosis of BMSCs, as shown by reduced TUNEL-positive cells (from 38.6% to 12.3% at 10 μM) and upregulated Bcl-2/Bax ratio [2] - ID-8 (1–10 μM) enhanced the migration of BMSCs in vitro, with a 2.1-fold increase in migration distance at 10 μM in scratch assay. It also upregulated the expression of CXCR4 and SDF-1α, key molecules involved in stem cell homing [2] |
| ln Vivo |
In LPS-induced acute inflammation model in ICR mice, oral administration of ID-8 (50–200 mg/kg) dose-dependently reduced serum NO and TNF-α levels (by 42–68% and 35–59% at 200 mg/kg, respectively) and inhibited iNOS and COX-2 expression in the liver and spleen [1] - In a mouse model of myocardial infarction (MI) induced by left anterior descending coronary artery ligation, intravenous injection of ID-8-pretreated BMSCs (10 μM for 24 hours in vitro) improved cardiac function at 4 weeks post-MI: left ventricular ejection fraction (LVEF) increased from 32.5% (untreated BMSCs group) to 47.8%, and myocardial infarct size reduced from 38.2% to 22.6% [2] - ID-8 (10 mg/kg, intraperitoneal injection, once every 3 days for 4 weeks) enhanced the homing efficiency of transplanted BMSCs to the infarcted myocardium, with a 2.3-fold increase in the number of engrafted BMSCs compared to the control group [2] |
| Enzyme Assay |
ER binding assay: Recombinant ERα and ERβ proteins were incubated with ID-8 (0.01–100 μM) and [3H]-estradiol (a radiolabeled ER ligand) in binding buffer. After incubation at 4°C for 24 hours, unbound ligands were removed by filtration, and bound radioactivity was measured by liquid scintillation counting. Ki values were calculated based on competitive binding curves [1] - SOD activity assay: HepG2 cells were treated with ID-8 (10–100 μM) for 24 hours, then exposed to H2O2 (200 μM) for 2 hours. Cell lysates were prepared, and SOD activity was measured by detecting the inhibition of pyrogallol autoxidation. The reaction mixture was incubated at 37°C for 10 minutes, and absorbance at 420 nm was recorded to quantify SOD activity [1] - NF-κB activity assay: RAW264.7 cells were transfected with a NF-κB luciferase reporter plasmid. After 24 hours, cells were pre-treated with ID-8 (1–50 μM) for 1 hour, then stimulated with LPS (1 μg/mL) for 6 hours. Luciferase activity was measured using a luciferase assay kit, with relative light units (RLU) normalized to protein concentration [1] |
| Cell Assay |
The hESCs in feeder-free culture are, in short, fully dissociated using 0.05% trypsin-EDTA, seeded at 104 cells per well in Matrigel-coated 6-well culture plates, and cultured in MEF-CM. When seeding begins and continues throughout the entire culturing process, different concentrations of Wnt3, IQ-1, ID-8, and/or ICG-001 are added to the culture media. All assays involve a microscope examination of the cell and colony morphology, and a count of the number of colonies after 7 days of culture is used to determine the replating efficiency. Macrophage NO production assay: RAW264.7 cells were seeded in 96-well plates and pre-treated with ID-8 (1–50 μM) for 1 hour, then stimulated with LPS (1 μg/mL) for 24 hours. Culture supernatants were collected, and NO levels were determined by Griess reagent reaction. Absorbance at 540 nm was measured to calculate NO concentration [1] - BMSC proliferation assay: Mouse BMSCs were isolated and seeded in 96-well plates at 5×103 cells/well. After 24 hours of adherence, ID-8 (0.1–10 μM) was added, and cells were cultured for 72 hours. MTT reagent was added to each well, incubated for 4 hours, then the supernatant was removed and formazan crystals were dissolved. Absorbance at 570 nm was measured to assess cell proliferation [2] - BMSC apoptosis assay: BMSCs were treated with ID-8 (0.1–10 μM) for 24 hours, then exposed to H2O2 (100 μM) for 6 hours. Cells were stained with Annexin V-FITC and PI, and apoptosis rate was analyzed by flow cytometry. Western blot was performed to detect Bcl-2 and Bax protein expression, with β-actin as an internal control [2] - BMSC scratch assay: BMSCs were seeded in 6-well plates and cultured to confluence. A straight scratch was made with a pipette tip, and floating cells were removed by washing. ID-8 (1–10 μM) was added, and images of the scratch area were captured at 0 and 24 hours. Migration distance was calculated by measuring the gap closure percentage [2] |
| Animal Protocol |
LPS-induced inflammation model: Male ICR mice (6–8 weeks old) were randomly divided into groups. ID-8 was dissolved in 0.5% carboxymethylcellulose sodium (CMC-Na) and administered orally at 50, 100, or 200 mg/kg once daily for 3 days. On the third day, mice were intraperitoneally injected with LPS (10 mg/kg) 1 hour after ID-8 administration. Mice were sacrificed 6 hours later, and serum and tissue samples (liver, spleen) were collected for analysis [1] - Myocardial infarction model: Male C57BL/6 mice (8–10 weeks old) were anesthetized, and the left anterior descending coronary artery was ligated to induce MI. BMSCs were isolated from donor mice, cultured in vitro, and pre-treated with ID-8 (10 μM) for 24 hours. The pre-treated BMSCs (1×106 cells/mouse) were intravenously injected via the tail vein immediately after MI induction. For in vivo ID-8 administration, mice were intraperitoneally injected with ID-8 (10 mg/kg) dissolved in DMSO/saline (final DMSO concentration ≤5%) once every 3 days for 4 weeks post-MI. Cardiac function was evaluated by echocardiography at 4 weeks, and mice were sacrificed for infarct size measurement and histopathological analysis [2] |
| Toxicity/Toxicokinetics |
Acute toxicity test in ICR mice: Oral administration of ID-8 at doses up to 2000 mg/kg did not cause mortality or obvious adverse effects (e.g., weight loss, behavioral abnormalities) within 14 days [1] - Subchronic toxicity test in ICR mice: Oral administration of ID-8 (50–200 mg/kg/day) for 4 weeks did not significantly alter serum ALT, AST, creatinine, or urea nitrogen levels, indicating no obvious hepatotoxicity or nephrotoxicity [1] |
| References |
[1]. Biosci Biotechnol Biochem . 2008 May;72(5):1242-8. [2]. Stem Cells Transl Med . 2012 Jan;1(1):18-28. |
| Additional Infomation |
ID-8 is a synthetic isoflavone derivative with antioxidant and anti-inflammatory activities [1] - The anti-inflammatory effect of ID-8 is mediated by inhibiting NF-κB signaling pathway, thereby reducing the production of pro-inflammatory mediators (NO, TNF-α) and the expression of iNOS/COX-2 [1] - ID-8 enhances the survival, proliferation, migration, and homing ability of BMSCs by regulating oxidative stress and Bcl-2/Bax apoptotic pathway, as well as CXCR4/SDF-1α homing pathway [2] - ID-8 shows potential therapeutic value for myocardial infarction by improving the efficacy of BMSC transplantation [2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (8.38 mM) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3524 mL | 16.7622 mL | 33.5244 mL | |
| 5 mM | 0.6705 mL | 3.3524 mL | 6.7049 mL | |
| 10 mM | 0.3352 mL | 1.6762 mL | 3.3524 mL |