Physicochemical Properties
| Molecular Formula | C32H40N10O7S |
| Molecular Weight | 708.79 |
| Exact Mass | 708.280 |
| PubChem CID | 172419005 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 3.2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 50 |
| Complexity | 1370 |
| Defined Atom Stereocenter Count | 2 |
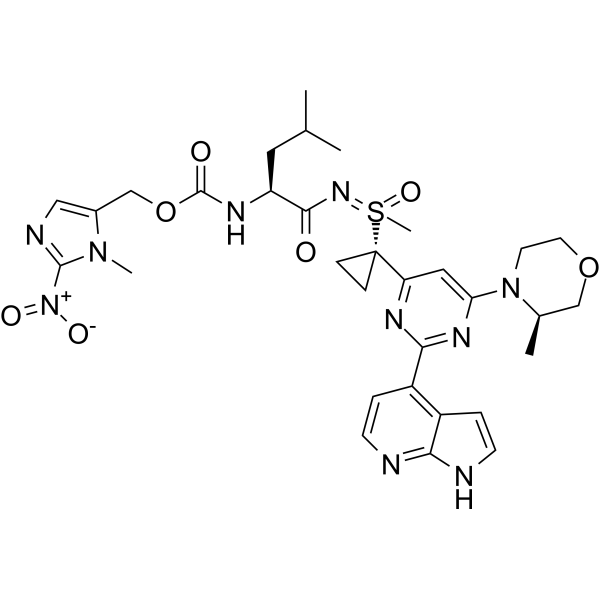
| SMILES | C[C@@H]1COCCN1C2=NC(=NC(=C2)C3(CC3)S(=NC(=O)[C@H](CC(C)C)NC(=O)OCC4=CN=C(N4C)[N+](=O)[O-])(=O)C)C5=C6C=CNC6=NC=C5 |
| InChi Key | ICQQVDQKEMQSJN-LEYGJJLKSA-N |
| InChi Code | InChI=1S/C32H40N10O7S/c1-19(2)14-24(36-31(44)49-18-21-16-35-30(40(21)4)42(45)46)29(43)39-50(5,47)32(8-9-32)25-15-26(41-12-13-48-17-20(41)3)38-28(37-25)23-7-11-34-27-22(23)6-10-33-27/h6-7,10-11,15-16,19-20,24H,8-9,12-14,17-18H2,1-5H3,(H,33,34)(H,36,44)/t20-,24+,50?/m1/s1 |
| Chemical Name | (3-methyl-2-nitroimidazol-4-yl)methyl N-[(2S)-4-methyl-1-[[methyl-[1-[6-[(3R)-3-methylmorpholin-4-yl]-2-(1H-pyrrolo[2,3-b]pyridin-4-yl)pyrimidin-4-yl]cyclopropyl]-oxo-lambda6-sulfanylidene]amino]-1-oxopentan-2-yl]carbamate |
| Synonyms | ICT10336; ICT-10336 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | ATR |
| ln Vitro | Targeted therapy remains the future of anti-cancer drug development, owing to the lack of specificity of current treatments which lead to damage in healthy normal tissues. ATR inhibitors have in recent times demonstrated promising clinical potential, and are currently being evaluated in the clinic. However, despite the considerable optimism for clinical success of these inhibitors, reports of associated normal tissues toxicities remain a concern and can compromise their utility. Here, ICT10336 is reported, a newly developed hypoxia-responsive prodrug of ATR inhibitor, AZD6738, which is hypoxia-activated and specifically releases AZD6738 only in hypoxic conditions, in vitro. This hypoxia-selective release of AZD6738 inhibited ATR activation (T1989 and S428 phosphorylation) and subsequently abrogated HIF1a-mediated adaptation of hypoxic cancers cells, thus selectively inducing cell death in 2D and 3D cancer models. Importantly, in normal tissues, ICT10336 is demonstrated to be metabolically stable and less toxic to normal cells than its active parent agent, AZD6738. In addition, ICT10336 exhibited a superior and efficient multicellular penetration ability in 3D tumor models, and selectively eradicated cells at the hypoxic core compared to AZD6738. In summary, the preclinical data demonstrate a new strategy of tumor-targeted delivery of ATR inhibitors with significant potential of enhancing the therapeutic index [1]. |
| Enzyme Assay |
In Vitro Metabolism [1] Cellular activation and metabolism of ICT10336 in normoxic or hypoxic conditions was conducted as described previously with slight modifications. Cells (2 × 106/mL) in complete media were seeded into 6‐well (1 per time point plus controls), and allowed attached over‐night. Pre‐conditioned media (2 mL) containing ICT10336 (10.0 µm) were added to each PBS washed cells. Controls contained either no cells or no drug. Samples were incubated for 6 and 24 h at 37 °C in either normoxia or hypoxic conditions. At each indicated time point, medium was removed and stored at −80 °C. Cells were washed (twice) with ice‐cold PBS, stripped and centrifuged (1000 g, 2 min), and the resulting cell pellets were solubilized in acetonitrile (200 µL), and then sonicated to disrupt the cells. Samples were centrifuged (10 000 g, 10 min), with the supernatant collected. Supernatants were then dried using a SP Genevac EZ‐PLUS evaporator for 30 min. The dried reaction was dissolved in a solution of 90% MeOH, 10% H2O, 0.1% formic acid (50 µL), which was then transferred into an HPLC vial for LC‐MS analysis. Ex Vivo Metabolism [1] Homogenized (1:4, w/v) human cancer cell‐derived tumors xenografts (CDX), normal mice tissues (liver and kidney, and plasma) were incubated with IC10336 (10 µm) in either hypoxic or normoxic conditions with reaction aliquots removed over a time‐period. Proteins were precipitated using acetonitrile and centrifuged for supernatant. Supernatants were dried for 30 min in a SP Genevac EZ‐PLUS evaporator. Dried metabolites were dissolved in 90% MeOH, 10% H2O, 0.1% formic acid (50 µL), and analyzed on LC‐MS. |
| Cell Assay |
Cell Cycle Analysis [1] MRC‐5 cells were seeded at a density of 1 × 106 cells and incubated for 24 h. Cells were treated with either AZD6738 or ICT10336 for 24 h. Treated cells and controls were trypsinised, washed with PBS, and fixed with 66.6% ethanol overnight. The fixed cells were rinsed with PBS, labeled with FxCycle PI/RNase staining solution following the manufacturer's instructions |
| References |
[1]. Hypoxia-Responsive Prodrug of ATR Inhibitor, AZD6738, Selectively Eradicates Treatment-Resistant Cancer Cells. Adv Sci (Weinh). 2024 Jul 8:e2403831. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4109 mL | 7.0543 mL | 14.1086 mL | |
| 5 mM | 0.2822 mL | 1.4109 mL | 2.8217 mL | |
| 10 mM | 0.1411 mL | 0.7054 mL | 1.4109 mL |