Physicochemical Properties
| Molecular Formula | C22H23CLO5 |
| Molecular Weight | 402.87 |
| Exact Mass | 402.123 |
| Elemental Analysis | C, 65.59; H, 5.75; Cl, 8.80; O, 19.86 |
| CAS # | 117621-64-4 |
| PubChem CID | 6438399 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.262 g/cm3 |
| Boiling Point | 540.1ºC at 760 mmHg |
| Flash Point | 280.4ºC |
| Vapour Pressure | 1.7E-12mmHg at 25°C |
| Index of Refraction | 1.583 |
| LogP | 5.259 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 28 |
| Complexity | 525 |
| Defined Atom Stereocenter Count | 3 |
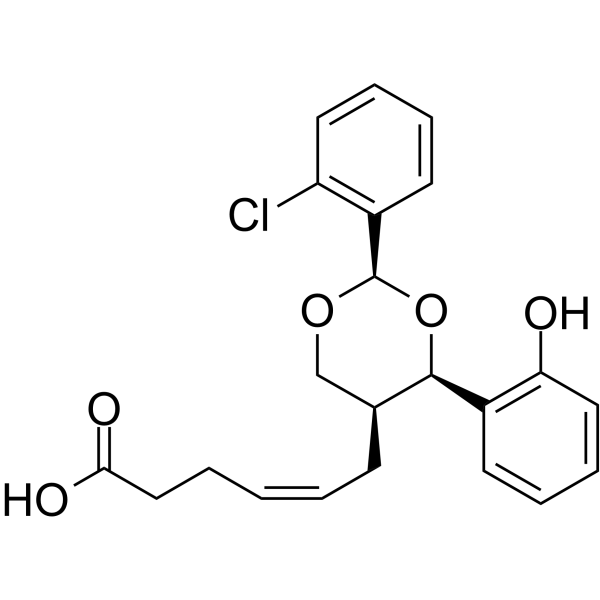
| SMILES | OC(CC/C=C\C[C@H]1CO[C@@H](C2=CC=CC=C2Cl)O[C@H]1C1=CC=CC=C1O)=O |
| InChi Key | WHUIENZXNGAHQI-YGPRPMEGSA-N |
| InChi Code | InChI=1S/C22H23ClO5/c23-18-11-6-4-9-16(18)22-27-14-15(8-2-1-3-13-20(25)26)21(28-22)17-10-5-7-12-19(17)24/h1-2,4-7,9-12,15,21-22,24H,3,8,13-14H2,(H,25,26)/b2-1-/t15-,21+,22+/m0/s1 |
| Chemical Name | (Z)-6-[(2R,4R,5S)-2-(2-chlorophenyl)-4-(2-hydroxyphenyl)-1,3-dioxan-5-yl]hex-4-enoic acid |
| Synonyms | ICI-192605 ; ICI192605; 117621-64-4; ICI 192605; 6-(2-(2-Chlorophenyl-4-hydroxyphenyl)-1,3-dioxan-5-yl)hexenoic acid; Ici-192605; (Z)-6-[(2R,4R,5S)-2-(2-chlorophenyl)-4-(2-hydroxyphenyl)-1,3-dioxan-5-yl]hex-4-enoic acid; ICI 192,605; FQ9418XA31; 4-Hexenoic acid, 6-(2-(2-chlorophenyl)-4-(2-hydroxyphenyl)-1,3-dioxan-5-yl)-, (2alpha,4alpha,5alpha(Z))-; ICI 192605 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | TXA2R |
| ln Vitro |
The synthesis and summary pharmacology of a novel thromboxane receptor antagonist 4(Z)-6-(2-o-chlorophenyl-4-o-hydroxyphenyl-1, 3-dioxan-cis-5-yl) hexenoic acid (ICI 192605) is reported. Compound 3 was competitive and selective with pA2 values of 8.0 +/- 0.1 (rabbit) and 8.4 +/- 0.05 (rat) on smooth muscle preparations and 8.16 +/- 0.01 on human platelets[1].
ICI 192605is a highly selective TP receptor blocker with pA2 of approximately 8 (Brewster et al., 1988; Brown et al., 1990). In the present study, 8-iso PGE2-evoked contractions were markedly and significantly reduced by 10−8 M ICI 192605, and abolished when the concentration of this blocker was increased 10-fold (Figures 3 and 4), indicating that they are likely directed through TP receptors.[2] |
| ln Vivo | In vivo activity of ICI 192605 was demonstrated in a Konzett Rossler guinea pig model at 0.01 mg/kg p.o[1]. |
| References |
[1]. The synthesis of a novel thromboxane receptor antagonist 4(Z)-6-(2-o-chlorophenyl-4-o-hydroxyphenyl-1,3-dioxan-cis-5-yl) hexenoic acid ICI 192605. Prostaglandins. 1988 Aug;36(2):173-8. [2].Vasoconstrictor responses, and underlying mechanisms, to isoprostanes in human and porcine bronchial arterial smooth muscle. Br J Pharmacol. 2003 Oct;140(4):759-63. |
| Additional Infomation |
The synthesis and summary pharmacology of a novel thromboxane receptor antagonist 4(Z)-6-(2-o-chlorophenyl-4-o-hydroxyphenyl-1, 3-dioxan-cis-5-yl) hexenoic acid (3) is reported. Compound 3 was competitive and selective with pA2 values of 8.0 +/- 0.1 (rabbit) and 8.4 +/- 0.05 (rat) on smooth muscle preparations and 8.16 +/- 0.01 on human platelets. In vivo activity of 3 was demonstrated in a Konzett Rossler guinea pig model at 0.01 mg/kg p.o. [1]
We investigated the effects of five different isoprostanes (8-iso PGE1, 8-iso PGE2, 8-iso PGF1α, 8-iso PGF2α and 8-iso PGF2β) on vasomotor tone in human and porcine bronchial arterial tissues. In the human bronchial arteries, 8-iso PGE2 and 8-iso PGF2α evoked powerful constrictions (magnitudes several fold greater than the responses to high millimolar KCl) with negative log concentration causing 50% excitation (EC50) values of 6.8 and 6.5, respectively; 8-iso PGE1 was less potent (EC50 not calculated, since a clear peak contraction was not obtained), while the other isoprostanes were largely ineffective. In the porcine arteries, on the other hand, all three F-ring isoprostanes as well as 8-iso PGE2 evoked constrictor responses, although the peak magnitudes were approximately 50% of the KCl-evoked response; 8-iso PGE2 and 8-iso PGF2α were the most potent, with negative log EC50 values of 6.5. We next sought to characterize the signaling pathways underlying the vasoconstrictor responses to 8-iso PGE2, since this was the most potent of the isoprostanes we tested. These responses were largely reversed by the thromboxane A2-selective (TP) prostanoid receptor antagonist ICI 192605 (10−8 M; 4(Z)-6-[(2,4,5 cis)2-(2-chlorophenyl)-4-(2-hydroxy phenyl)1,3-dioxan-5-yl]hexenoic acid) as well as by the nonspecific tyrosine kinase inhibitor genistein (10−5 and 10−4 M), and were reversed approximately 50% by the Rho-kinase inhibitor Y27632 (10−5 M; (+)-(R)-trans-4-(1-aminoethyl)-N-(pyridyl) cyclohexanecarboxamide dihydrochloride). We conclude, therefore, that 8-iso PGE2 constricts bronchial vasculature through the activation of TP receptors, which in turn trigger tyrosine kinase and Rho-kinase activities, resulting in powerful vasoconstriction. These findings are highly relevant to lung transplantation and to exercise-induced asthma. [2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4822 mL | 12.4110 mL | 24.8219 mL | |
| 5 mM | 0.4964 mL | 2.4822 mL | 4.9644 mL | |
| 10 mM | 0.2482 mL | 1.2411 mL | 2.4822 mL |