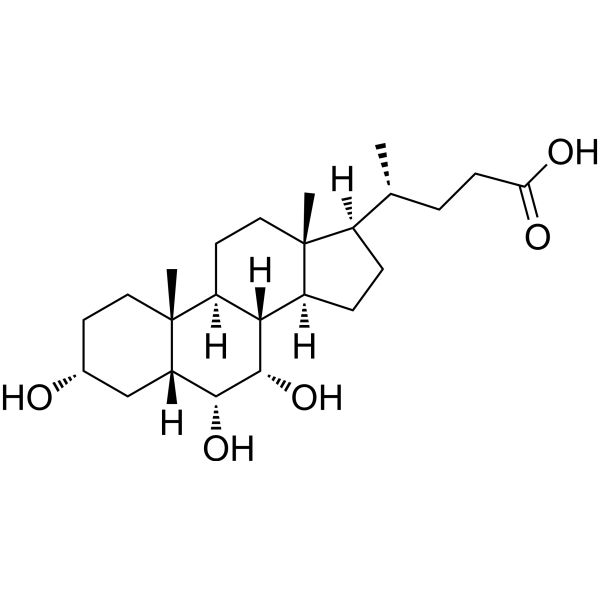
Hyocholic Acid (3α,6α,7α-trihydroxy-5β-cholan-24-oic acid) is a biliary acid found mainly in pig as well as in human urine at low concentrations, in particular, in urine samples from patients with cholestasis. Hyocholic Acid can promote GLP-1 secretion via activating TGR5 and inhibiting FXR in enteroendocrine cells. It is known for its exceptional resistance to type 2 diabetes . Hyocholic acid differs from the primary bile acids found in humans by having a third hydroxyl group in the α-conformation at the 6-position, unlike cholic acid, which has a 12-hydroxyl, and chenodeoxycholic acid which has neither a 6- or 12-hydroxyl. It also differs from the muricholic acids found in rodents, as they are 6β-hydroxylated, and can have the 7-hydroxyl in either the α- or β- positions, forming α- or β-muricholic acids.
Physicochemical Properties
| Molecular Formula | C24H40O5 |
| Molecular Weight | 408.5714 |
| Exact Mass | 408.287 |
| CAS # | 547-75-1 |
| Related CAS # | Hyocholic Acid-d4;Hyocholic Acid-d5 |
| PubChem CID | 92805 |
| Appearance | White to light yellow solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 565.7±40.0 °C at 760 mmHg |
| Melting Point | 188-189ºC |
| Flash Point | 310.0±23.8 °C |
| Vapour Pressure | 0.0±3.5 mmHg at 25°C |
| Index of Refraction | 1.558 |
| LogP | 3.82 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 29 |
| Complexity | 637 |
| Defined Atom Stereocenter Count | 11 |
| SMILES | C[C@H](CCC(=O)O)[C@H]1CC[C@H]2[C@H]3[C@H](CC[C@]12C)[C@@]4(C)CC[C@H](C[C@H]4[C@H]([C@H]3O)O)O |
| InChi Key | DKPMWHFRUGMUKF-KWXDGCAGSA-N |
| InChi Code | InChI=1S/C24H40O5/c1-13(4-7-19(26)27)15-5-6-16-20-17(9-11-23(15,16)2)24(3)10-8-14(25)12-18(24)21(28)22(20)29/h13-18,20-22,25,28-29H,4-12H2,1-3H3,(H,26,27)/t13-,14-,15-,16+,17+,18+,20+,21-,22+,23-,24-/m1/s1 |
| Chemical Name | (4R)-4-[(3R,5R,6R,7S,8S,9S,10R,13R,14S,17R)-3,6,7-trihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl]pentanoic acid |
| Synonyms | gamma-Muricholic acid; Iocholic acid; 3alpha,6alpha,7alpha-Trihydroxy-5beta-cholan-24-oic acid; (4R)-4-[(3R,5R,6R,7S,8S,9S,10R,13R,14S,17R)-3,6,7-trihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl]pentanoic acid; 2H5H0Q47FL; CHEBI:81244; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | TGR5; FXR |
| ln Vitro | In STC-1 and NCI-H716 cells, HCA (25 and 50 μM, 24 h) increases GLP-1 protein secretion[3]. In STC-1 and NCI-H716 cells, HCA (25 and 50 μM, 24 h) increases the transcription of the proglucagon gene[3]. |
| ln Vivo |
Pigs' blood glucose rise brought on by BA depletion is suppressed by hyocholic acid (20 mg/kg, po)[3]. In diabetic mouse models, hydroxycholic acid (100 mg/kg/day, po) enhances serum fasting GLP-1 secretion and glucose homeostasis [3]. Hyocholic acid (HCA) and its derivatives are found in trace amounts in human blood but constitute approximately 76% of the bile acid (BA) pool in pigs, a species known for its exceptional resistance to type 2 diabetes. Here, we show that BA depletion in pigs suppressed secretion of glucagon-like peptide-1 (GLP-1) and increased blood glucose levels. HCA administration in diabetic mouse models improved serum fasting GLP-1 secretion and glucose homeostasis to a greater extent than tauroursodeoxycholic acid. HCA upregulated GLP-1 production and secretion in enteroendocrine cells via simultaneously activating G-protein-coupled BA receptor, TGR5, and inhibiting farnesoid X receptor (FXR), a unique mechanism that is not found in other BA species. We verified the findings in TGR5 knockout, intestinal FXR activation, and GLP-1 receptor inhibition mouse models. Finally, we confirmed in a clinical cohort, that lower serum concentrations of HCA species were associated with diabetes and closely related to glycemic markers.[3] |
| Cell Assay |
Western Blot Analysis[3] Cell Types: NCI-H716 cells Tested Concentrations: 50 μM Incubation Duration: 48 h Experimental Results: Inhibited the high expression of SHP (small heterodimer partner) induced by the FXR agonist. |
| Animal Protocol |
Animal/Disease Models: BA depletion pigs[3] Doses: 20 mg/kg Route of Administration: Oral administration (po) Experimental Results: Attenuated the increased blood glucose levels corresponding with GLP-1 decrease. Animal/Disease Models: db/db model, and the high-fat diet and streptozotocin (HFD+STZ) induced diabetic model[3] Doses: 100 mg/kg/day Route of Administration: Oral administration (po) Experimental Results: Improved oral glucose tolerances shown by lower glucose levels. Increased circulating active GLP- 1 levels and fasting insulin levels. |
| ADME/Pharmacokinetics |
Metabolism / Metabolites Hyocholic acid has known human metabolites that include hyocholic acid 24-O-glucuronide and hyocholic acid 6-O-glucuronide. |
| References |
[1]. Hyocholic acid species as novel biomarkers for metabolic disorders. Nat Commun. 2021 Mar 5;12(1):1487. [2]. Sulphated and unsulphated bile acids in serum, bile, and urine of patients with cholestasis. Gut 17(11), 861-869 (1976). [3]. Hyocholic acid species improve glucose homeostasis through a distinct TGR5 and FXR signaling mechanism. Cell Metab. 2021 Apr 6;33(4):791-803.e7. |
| Additional Infomation |
Hyocholic acid is a trihydroxy-5beta-cholanic acid in which the three hydroxy substituents are located at the 3alpha-, 6alpha- and 7alpha-positions. It has a role as a mouse metabolite, a human urinary metabolite and a rat metabolite. It is a C24-steroid, a 6alpha-hydroxy steroid, a 7alpha-hydroxy steroid and a member of muricholic acids. It is a conjugate acid of a hyocholate. Hyocholic acid has been reported in Homo sapiens with data available. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~62.5 mg/mL (~152.97 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.12 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.12 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.12 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4476 mL | 12.2378 mL | 24.4756 mL | |
| 5 mM | 0.4895 mL | 2.4476 mL | 4.8951 mL | |
| 10 mM | 0.2448 mL | 1.2238 mL | 2.4476 mL |