Physicochemical Properties
| Molecular Formula | C55H71N13O12S2 |
| Molecular Weight | 1170.36 |
| Exact Mass | 1169.48 |
| Elemental Analysis | C, 56.44; H, 6.11; N, 15.56; O, 16.40; S, 5.48 |
| CAS # | 257943-19-4 |
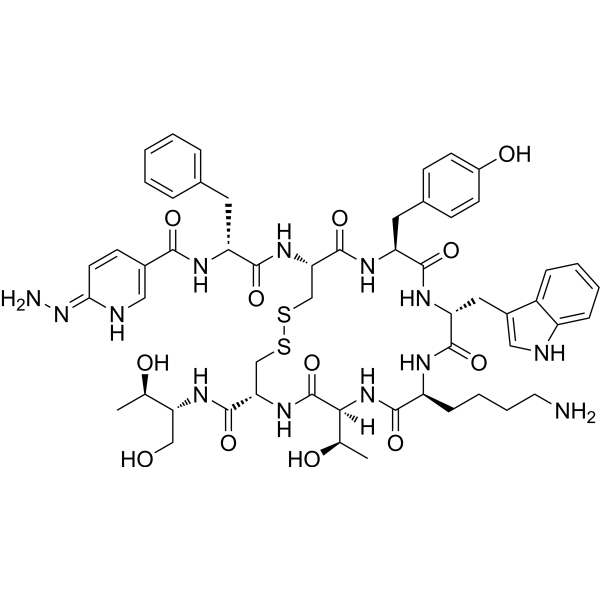
| Sequence | {Hynic}-{d-Phe}-Cys-Tyr-{d-Trp}-Lys-Thr-Cys-{Thr-ol} (Disulfidebridge:Cys2-Cys7); |
| SequenceShortening | FCYWKTCT; {Hynic}-{d-Phe}-CY-{d-Trp}-KTC-{Thr-ol} (Disulfidebridge:Cys2-Cys7) |
| Appearance | Typically exists as solid at room temperature |
| SMILES | S1C([H])([H])[C@@]([H])(C(N([H])[C@]([H])(C([H])([H])O[H])[C@@]([H])(C([H])([H])[H])O[H])=O)N([H])C([C@]([H])([C@@]([H])(C([H])([H])[H])O[H])N([H])C([C@]([H])(C([H])([H])C([H])([H])C([H])([H])C([H])([H])N([H])[H])N([H])C([C@@]([H])(C([H])([H])C2=C([H])N([H])C3=C([H])C([H])=C([H])C([H])=C23)N([H])C([C@@]([H])(C([H])([H])C2C([H])=C([H])C(=C([H])C=2[H])O[H])N([H])C([C@]([H])(C([H])([H])S1)N([H])C([C@@]([H])(C([H])([H])C1C([H])=C([H])C([H])=C([H])C=1[H])N([H])C(C1=C([H])N=C(C([H])=C1[H])N([H])N([H])[H])=O)=O)=O)=O)=O)=O)=O |
| Synonyms | HYNIC-Tyr3-octreotide; Hynic-toc; XBO8UII5SV; Hydrazinonicotinyl-Tyr3-octreotide; UNII-XBO8UII5SV; 913556-62-4; N-((6-Hydrazinyl-3-pyridinyl)carbonyl)-D-phenylalanyl-L-cysteinyl-L-tyrosyl-D-tryptophyl-L-lysyl-L-threonyl-N-((1R,2R)-2-hydroxy-1-(hydroxymethyl)propyl)-L-cysteinamide cyclic (2->7)-disulfide; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Radionuclide-Drug Conjugates (RDCs) |
| ln Vitro | [111In-diethylene triamine penta-acetic acid-D-Phe1]-octreotide (DTPA-octreotide) scintigraphy has gained widespread acceptance as a diagnostic clinical procedure in oncology for imaging somatostatin receptor-positive tumours. However, indium-111 as a radiolabel has several drawbacks, including limited availability, suboptimal gamma energy and high radiation burden to the patient. We have recently reported on the preclinical development of 99mTc-EDDA/HYNIC-TOC, a new octreotide derivative which showed promising results both in vitro [1]. |
| ln Vivo | We now report our initial clinical experiences with this new radiopharmaceutical in ten oncological patients. The clinical diagnoses were: carcinoid syndrome (n=5), thyroid cancer (n=3), pancreatic cancer (n=1) and pituitary tumour (n=1). The biodistribution and kinetics of 99mTc-EDDA/HYNIC-TOC were compared with those of 111In-DTPA-octreotide in six cases, and with those of 111In-DOTA-TOC in five cases. With the new tracer tumours were imaged within 15 min after injection and showed the highest target/non-target ratios 4 h after injection. Tumour uptake persisted up to 20 h p.i. The rate of blood clearance was similar to that of 111In-DTPA-octreotide but faster than that of 111In-DOTA-TOC, while urinary excretion was lower compared with the 111In derivatives. Semi-quantitative region of interest analysis showed that 99mTc-EDDA/HYNIC-TOC produced higher tumour/organ (target/non-target) ratios than the 111In derivatives, especially in relation to heart and muscle. Significantly more lesions could be detected in 99mTc images. We conclude that 99mTcEDDA/HYNIC-TOC shows better imaging properties for the identification of somatostatin receptor-positive tumour sites than currently available 111In-labelled octreotide derivatives [1]. |
| Animal Protocol | Patients: This clinical study was approved by the local ethical committee and all patients gave their informed consent prior to inclusion. Scintigraphy with 99mTc-EDDA/HYNIC-TOC was performed in ten patients, details of whom are given in Table 1. Comparative imaging with 111In-DTPA-octreotide alone was performed in five patients and with 111In-DOTA-TOC alone in four patients, while one patient underwent all three imaging modalities. The time between comparative studies normally ranged from 2 to 30 days, although in one patient the 111In study was performed on the day following the 99mTc study. Patients were not treated with cold somatostatin analogues within 1 month before the imaging studies. Imaging. Planar imaging was performed with a double-head camera (Elscint HELIX, Haifa, Israel). All patients were imaged at 4 h post injection. Seven patients were additionally imaged at 1–2 h, and in two patients additional imaging was performed at 15 min and 20 h. For 99mTc studies the camera was equipped with a lowenergy all-purpose parallel-hole collimator, window setting 140 keV, width 10%. 111In images were obtained using a high-energy parallel-hole collimator, with window setting over both 111In peaks at 172 and 246 keV and a window width of 20%. Single-photon emission tomography (SPET) imaging of areas of interest was performed 4 h post injection and in some patients additionally at 20 h. For 99mTc tomographic acquisition, the same double-head camera as described above was used. Acquisition parameters were: 60 projections, 25 s/projection, matrix 64×64, zoom 1. SPET for 111In studies was performed on a Siemens single-head camera (ZL3000, Siemens, Erlangen, Germany) equipped with a medium-energy parallel-hole collimator using 60 projections, 35 s/projection, matrix 64×64. This camera system was also used for SPET of brain and neck regions of interest (ROIs) for 99mTc studies using a low-energy parallel-hole collimator. In general, imaging parameters were chosen that produced comparable whole-body counts for both 111In and 99mTc studies. Total whole-body counts (arithmetic mean of anterior and posterior images ± SD) were 2056±244 kcounts for 111In studies and 1764±428 kcounts for 99mTc studies (decay-corrected to the time of injection). |
| References |
[1]. 99mTc-EDDA/HYNIC-TOC: a new 99mTc-labelled radiopharmaceutical for imaging somatostatin receptor-positive tumours; first clinical results and intra-patient comparison with 111In-labelled octreotide derivatives. Eur J Nucl Med. 2000 Sep;27(9):1318-25. |
| Additional Infomation | The imaging properties of 99mTc-EDDA/HYNIC-TOC appear to be advantageous for investigating somatostatin receptor-positive tumours in man. This new preparation has a rapid tumour uptake and a similar biodistribution to 111In-DTPA-octreotide but produced higher tumour to organ ratios and detected a greater number of lesions. Finally, the easy availability of 99mTc from generators and the low cost of this radionuclide make 99mTc-EDDA/ HYNIC-TOC a promising candidate to replace currently used 111In-octreotide derivatives in diagnostic nuclear medicine in oncology. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.8544 mL | 4.2722 mL | 8.5444 mL | |
| 5 mM | 0.1709 mL | 0.8544 mL | 1.7089 mL | |
| 10 mM | 0.0854 mL | 0.4272 mL | 0.8544 mL |